Scalable Copper-Catalyzed Synthesis of Cannabinol for Commercial Pharmaceutical Production
The pharmaceutical and fine chemical industries are witnessing a paradigm shift in the synthesis of high-value cannabinoids, driven by the urgent need for efficient, compliant, and scalable manufacturing processes. Patent CN114853711A introduces a groundbreaking methodology for the preparation of Cannabinol (CBN) utilizing a copper-catalyzed one-pot strategy. This innovation addresses critical bottlenecks in the current supply chain, specifically the challenge of converting abundant Cannabidiol (CBD) into the more valuable CBN without generating regulated psychoactive impurities like Tetrahydrocannabinol (THC). By leveraging earth-abundant transition metals, this technology offers a robust pathway for producing high-purity intermediates essential for next-generation therapeutic applications.
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the complex, multi-step total syntheses that have historically plagued cannabinoid production. The disclosed method transforms a readily available starting material into a premium product through a streamlined oxidative cyclization. This not only enhances the economic viability of CBN production but also ensures a cleaner impurity profile, which is paramount for meeting the stringent quality standards required by global regulatory bodies for pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthetic routes to Cannabinol have been fraught with inefficiencies and regulatory risks that hinder commercial adoption. Traditional strategies often rely on intermolecular aromatic nucleophilic substitutions or intramolecular cyclizations that require harsh conditions and multiple purification steps, leading to significant yield erosion. Furthermore, a prevalent method involves the derivatization of CBD using large excesses of elemental iodine to facilitate cyclization. This approach is not only economically burdensome due to the high cost of stoichiometric iodine but also generates substantial hazardous waste, complicating environmental compliance and increasing the overall cost of goods sold (COGS).
Moreover, the acid-catalyzed conversion of CBD, while common, suffers from a lack of selectivity, frequently resulting in the formation of Delta-9-THC and Delta-8-THC as unavoidable byproducts. Since THC is a strictly controlled substance in many jurisdictions, its presence necessitates expensive and technically challenging removal processes to ensure the final product remains non-psychoactive. These legacy methods create a fragile supply chain where yield, cost, and compliance are constantly at odds, making it difficult for manufacturers to secure a reliable cannabinol supplier capable of delivering consistent quality at scale.
The Novel Approach
In stark contrast, the technology disclosed in CN114853711A presents a sophisticated yet operationally simple solution by employing a copper-catalyzed oxidative system. This novel approach utilizes catalytic amounts of copper salts, such as CuCl2, CuBr2, or Cu(OAc)2, in conjunction with benign oxidants like oxygen, DDQ, or hydrogen peroxide. The reaction proceeds in a single pot, eliminating the need for intermediate isolation and significantly reducing solvent consumption and processing time. This one-pot design is a hallmark of modern green chemistry, aligning perfectly with the industry's drive towards sustainable manufacturing practices.
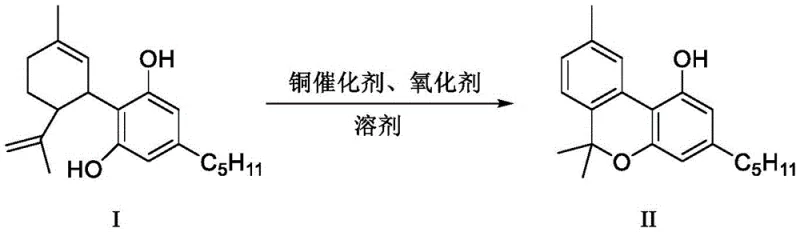
The core of this innovation is the selective oxidative aromatization of the CBD precursor. As illustrated in the reaction scheme, the copper catalyst facilitates the removal of hydrogen atoms to form the fully aromatic tricyclic structure of CBN without inducing the rearrangement reactions that lead to THC. This mechanistic divergence is crucial for maintaining product integrity and regulatory status. By operating under mild temperatures ranging from 40°C to 120°C and using common industrial solvents like toluene or acetonitrile, this method offers a practical and scalable alternative that dramatically simplifies the production workflow while maximizing output efficiency.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The success of this synthesis relies on the unique redox properties of the copper catalyst, which acts as a mediator for electron transfer during the oxidative cyclization process. In the presence of an oxidant, the copper species cycles between different oxidation states, activating the substrate for dehydrogenation. This catalytic cycle effectively promotes the formation of the new carbon-oxygen bond and the subsequent aromatization of the terpene moiety found in CBD. The choice of ligand, such as p-toluenesulfonic acid, further fine-tunes the acidity of the medium, ensuring that the reaction proceeds through the desired oxidative pathway rather than the acid-catalyzed isomerization pathway that generates THC.
From an impurity control perspective, this mechanism offers superior selectivity. The oxidative conditions are specifically tuned to target the allylic and benzylic positions necessary for CBN formation, leaving other sensitive functional groups intact. This high level of chemoselectivity results in a crude reaction mixture that is significantly cleaner than those obtained from iodine-mediated or acid-catalyzed methods. Consequently, the downstream purification burden is greatly reduced, allowing for the isolation of high-purity cannabinol with fewer chromatographic steps, which is a critical factor in reducing manufacturing costs and improving overall process throughput.
How to Synthesize Cannabinol Efficiently
The operational simplicity of this copper-catalyzed protocol makes it an attractive candidate for immediate technology transfer and pilot plant evaluation. The process begins with the precise mixing of CBD, the selected copper catalyst, and the oxidant in a reaction vessel equipped with heating and stirring capabilities. The reaction parameters, including temperature and time, are flexible, allowing process engineers to optimize the balance between reaction rate and energy consumption based on their specific facility constraints. Detailed standardized synthesis steps see the guide below.
- Mix Cannabidiol (CBD) with a copper catalyst (e.g., CuBr2, Cu(OAc)2) and an oxidant (e.g., DDQ, Oxygen) in a suitable solvent like toluene or acetonitrile.
- Heat the reaction mixture to a temperature between 40°C and 120°C and maintain stirring for 6 to 48 hours to ensure complete conversion.
- Quench the reaction with saturated ammonium chloride, extract the organic layer, dry, and purify via column chromatography to isolate high-purity Cannabinol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology represents a strategic opportunity to optimize the cost structure and reliability of the cannabinoid supply chain. By shifting away from expensive stoichiometric reagents like iodine and complex multi-step syntheses, manufacturers can achieve substantial cost savings in pharmaceutical intermediates manufacturing. The use of commodity chemicals such as copper salts and oxygen significantly lowers the raw material bill, while the one-pot nature of the reaction reduces utility consumption and labor costs associated with multiple unit operations.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants and the reduction in solvent usage directly translate to a lower cost of goods sold. Unlike traditional methods that require the disposal of large quantities of iodine waste, this catalytic process generates minimal hazardous byproducts, thereby reducing waste treatment expenses. Furthermore, the high yields reported in the patent examples, often exceeding 80%, ensure that raw material utilization is maximized, providing a clear economic advantage over legacy synthetic routes.
- Enhanced Supply Chain Reliability: Sourcing high-quality CBN has historically been challenging due to the complexity of its synthesis. This method utilizes widely available and stable starting materials, mitigating the risk of supply disruptions associated with specialty reagents. The robustness of the copper-catalyzed system allows for consistent batch-to-batch reproducibility, which is essential for maintaining long-term contracts with downstream pharmaceutical clients. This reliability ensures a steady flow of high-purity cannabinoids, reducing lead time for high-purity cannabinoids and stabilizing inventory levels.
- Scalability and Environmental Compliance: The commercial scale-up of complex cannabinoids is often hindered by safety concerns regarding exothermic reactions or hazardous reagents. This process operates under relatively mild conditions and uses oxygen or benign quinones as oxidants, presenting a much safer profile for large-scale production. Additionally, the simplified workup procedure facilitates easier compliance with environmental regulations regarding solvent emissions and waste disposal, positioning manufacturers as leaders in sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios. The answers provided are derived directly from the technical specifications and beneficial effects outlined in the patent documentation.
Q: How does this copper-catalyzed method avoid THC formation?
A: Unlike acid-catalyzed cyclization which often leads to psychoactive Tetrahydrocannabinol (THC) as a byproduct, this oxidative copper-catalyzed pathway selectively promotes aromatization to form Cannabinol (CBN) directly, ensuring regulatory compliance and higher product value.
Q: What are the cost advantages of using copper catalysts over traditional methods?
A: Traditional methods often require expensive stoichiometric amounts of elemental iodine or complex multi-step sequences. This method utilizes earth-abundant copper salts in catalytic amounts and common oxidants, drastically reducing raw material costs and simplifying the purification process.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent describes a robust one-pot procedure with yields consistently above 80%. The use of common solvents like toluene and mild reaction conditions makes it highly amenable to commercial scale-up without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cannabinol Supplier
As the global demand for non-psychoactive cannabinoids continues to surge, partnering with a technologically advanced CDMO is essential for securing a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Cannabinol meets the highest international standards for pharmaceutical and nutraceutical applications.
We invite you to collaborate with our technical team to explore how this copper-catalyzed technology can be tailored to your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this efficient manufacturing route. Contact our technical procurement team today to obtain specific COA data and route feasibility assessments, and let us help you streamline your supply chain for the future of cannabinoid therapeutics.
