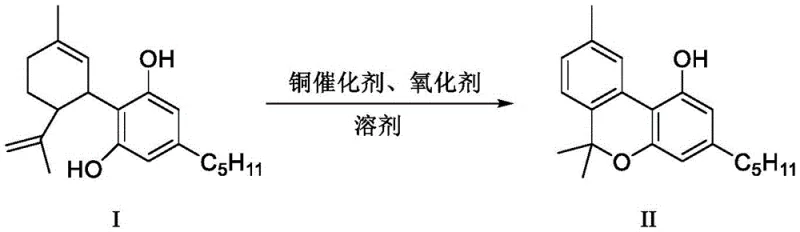
Advanced Copper-Catalyzed One-Pot Synthesis of Cannabinol for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust pathways to access high-value cannabinoid derivatives while navigating complex regulatory landscapes. Patent CN114853711A introduces a transformative approach to synthesizing Cannabinol (CBN), a compound renowned for its sedative and antibacterial properties, directly from the abundant precursor Cannabidiol (CBD). This innovation addresses a critical bottleneck in the supply chain: the difficulty of producing CBN without generating Tetrahydrocannabinol (THC), a strictly controlled psychotropic substance. By leveraging a novel copper-catalyzed oxidative cyclization strategy, this technology enables a direct, one-pot conversion that bypasses the regulatory and safety hurdles associated with THC formation. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable cannabinoid intermediate supplier capable of delivering high-purity materials through a streamlined, cost-effective process that is ready for immediate industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cannabinol has been plagued by inefficiencies and regulatory risks that hinder commercial viability. Traditional total synthesis routes often involve cumbersome multi-step sequences to construct the benzopyranone skeleton, resulting in low overall yields and excessive waste generation. Alternatively, semi-synthetic approaches deriving CBN from CBD frequently rely on the use of stoichiometric or large excess amounts of elemental iodine to facilitate the necessary oxidative cyclization. This reliance on iodine not only drives up raw material costs significantly but also creates substantial downstream processing challenges, as removing residual iodine and iodine-byproducts requires rigorous purification steps. Furthermore, many acid-catalyzed conversion methods carry the inherent risk of isomerizing CBD into THC, creating a compliance nightmare for manufacturers who must guarantee their products are free from controlled substances. These factors combined have prevented many existing methods from achieving true commercial scale-up, leaving the market underserved.
The Novel Approach
The methodology disclosed in CN114853711A fundamentally reimagines this transformation by replacing harsh halogenating agents with a sophisticated copper catalytic system. This new route utilizes inexpensive copper salts, such as copper(II) acetate or copper(II) bromide, in conjunction with benign oxidants like oxygen, hydrogen peroxide, or DDQ. The reaction proceeds efficiently in common organic solvents such as toluene or acetonitrile at moderate temperatures ranging from 40°C to 120°C. Crucially, this is a one-pot process, meaning the conversion from CBD to CBN occurs in a single reactor vessel without the need for isolating unstable intermediates. This simplification drastically reduces operational complexity and solvent consumption. By eliminating the need for elemental iodine and avoiding conditions that favor THC formation, this approach offers a cleaner, safer, and more economically attractive pathway for the production of high-purity CBN, positioning it as an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
From a mechanistic perspective, this transformation represents an elegant example of transition metal-catalyzed oxidative functionalization. The reaction likely initiates with the coordination of the copper catalyst to the phenolic hydroxyl groups of the Cannabidiol substrate, activating the aromatic ring towards nucleophilic attack or radical coupling. In the presence of the oxidant, the copper center undergoes redox cycling, facilitating the removal of hydrogen atoms and promoting the formation of the new carbon-oxygen bond required to close the pyran ring. The inclusion of a ligand, such as p-toluenesulfonic acid, further modulates the acidity of the medium and stabilizes the catalytic species, ensuring high turnover numbers. This precise control over the reaction environment allows for the selective aromatization of the cyclohexene ring in CBD to the aromatic ring found in CBN, a process that is thermodynamically favorable but kinetically challenging without the correct catalyst. The result is a highly selective transformation that preserves the integrity of the alkyl side chains while effecting the necessary skeletal rearrangement.
Impurity control is paramount in the synthesis of regulated substances, and this copper-catalyzed mechanism inherently suppresses the formation of unwanted byproducts. Unlike strong Brønsted acid catalysis which can induce carbocation rearrangements leading to THC, the oxidative nature of this copper system directs the reaction trajectory exclusively towards the aromatic CBN structure. The mild reaction conditions prevent the degradation of the sensitive terpene moieties often found in cannabinoid structures. Furthermore, the use of catalytic amounts of copper (typically 0.01 to 0.2 equivalents) ensures that heavy metal contamination in the final product is minimized, simplifying the purification protocol. For quality assurance teams, this means the resulting crude material has a cleaner impurity profile, reducing the burden on downstream chromatography or crystallization steps. This mechanistic elegance translates directly into higher process reliability and consistent batch-to-batch quality, which are essential metrics for any reliable cannabinoid intermediate supplier aiming to serve the global pharmaceutical market.
How to Synthesize Cannabinol Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent ratios and thermal management, although the procedure itself is remarkably straightforward. The general protocol involves dissolving the CBD starting material in a selected solvent, adding the copper catalyst and oxidant, and heating the mixture under reflux or controlled temperature conditions. The reaction progress can be easily monitored via TLC or HPLC, typically reaching completion within 6 to 48 hours depending on the specific oxidant and temperature chosen. Workup involves a standard aqueous quench, often using saturated ammonium chloride, followed by extraction and drying. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles optimized for maximum yield, are outlined in the guide below.
- Mix Cannabidiol (CBD) with a copper catalyst (e.g., Cu(OAc)2), an oxidant (e.g., DDQ), and optionally p-toluenesulfonic acid in a suitable solvent like toluene.
- Heat the reaction mixture to a temperature between 40°C and 120°C and maintain stirring for 6 to 48 hours to ensure complete conversion.
- Quench the reaction with saturated ammonium chloride, extract the organic layer, dry, and purify via column chromatography to obtain high-purity CBN.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology offers profound strategic advantages beyond mere technical feasibility. The shift from stoichiometric iodine reagents to catalytic copper systems fundamentally alters the cost structure of CBN production. Copper salts are commodity chemicals with stable pricing and widespread availability, contrasting sharply with the volatility and handling costs associated with large quantities of elemental iodine. Additionally, the one-pot nature of the reaction eliminates multiple unit operations, such as intermediate isolation and solvent swaps, which are major drivers of labor and utility costs in chemical manufacturing. This streamlining leads to substantial cost savings in production without compromising on the quality of the final active pharmaceutical ingredient. The ability to run the reaction at atmospheric pressure with oxygen or simple liquid oxidants further reduces the need for specialized high-pressure equipment, lowering capital expenditure requirements for scale-up.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic reduction in reagent costs and waste disposal fees. By utilizing low loadings of inexpensive copper catalysts instead of expensive stoichiometric oxidants, the direct material cost per kilogram of CBN is significantly lowered. Moreover, the simplified workup procedure reduces the consumption of auxiliary solvents and silica gel for purification, contributing to a leaner manufacturing budget. The elimination of hazardous iodine waste streams also mitigates environmental compliance costs, allowing for a more sustainable and financially efficient operation that maximizes margin potential for high-purity cannabinoid intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is critical for meeting the demands of the growing cannabinoid therapeutics market. This synthesis route relies on raw materials that are globally sourced and readily available, minimizing the risk of supply disruptions caused by geopolitical issues or single-source dependencies. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites without the need for highly specialized infrastructure. This flexibility ensures that lead times for high-purity cannabinoid intermediates can be kept short and predictable, providing downstream drug developers with the confidence they need to plan their clinical and commercial timelines effectively.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the simplicity of this one-pot method makes it inherently scalable from gram to multi-ton quantities. The reaction generates fewer byproducts and utilizes greener oxidants like oxygen or hydrogen peroxide in certain embodiments, aligning with modern green chemistry principles. This reduced environmental footprint simplifies the permitting process for new manufacturing facilities and ensures long-term operational sustainability. For supply chain leaders, this means investing in a technology that is future-proofed against tightening environmental regulations, securing the commercial scale-up of complex pharmaceutical intermediates for years to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios. We encourage technical teams to review these points closely to appreciate the full scope of the innovation.
Q: Why is copper catalysis preferred over traditional iodine-mediated methods for CBN synthesis?
A: Traditional methods often require large excesses of elemental iodine, which increases cost and complicates purification. The copper-catalyzed method described in patent CN114853711A utilizes inexpensive copper salts and common oxidants, offering a more economically viable and environmentally friendly pathway with yields exceeding 80%.
Q: Does this synthesis method risk forming Tetrahydrocannabinol (THC)?
A: No, a key advantage of this oxidative cyclization strategy is its selectivity. Unlike acid-catalyzed cyclizations that may inadvertently produce psychoactive THC, this copper-mediated oxidative pathway directly converts Cannabidiol (CBD) to Cannabinol (CBN), effectively bypassing the formation of regulated psychotropic substances.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed as a one-pot reaction operating under relatively mild temperatures (40-120°C) using commercially available solvents like toluene or acetonitrile. The simplicity of the workup procedure and the use of low catalyst loadings make it highly amenable to commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cannabinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this copper-catalyzed pathway to redefine the supply landscape for cannabinoid therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle the specific solvent and thermal requirements of this oxidative cyclization, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We are committed to delivering not just a chemical product, but a comprehensive supply solution that supports your regulatory filings and commercial launch strategies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this method can optimize your bottom line. Please contact us today to request specific COA data for our CBN batches and to discuss route feasibility assessments for your unique formulation needs. Let us be your partner in bringing high-quality, compliant cannabinoid medicines to patients worldwide.
