Revolutionizing Polysubstituted Pyridine Production with Solvent-Free Iron Catalysis for Commercial Scale
Revolutionizing Polysubstituted Pyridine Production with Solvent-Free Iron Catalysis for Commercial Scale
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for more efficient, sustainable, and cost-effective synthetic routes for pharmaceutical intermediates. A groundbreaking development in this field is detailed in patent CN111138345A, which introduces a novel method for synthesizing polysubstituted pyridine derivatives. This technology leverages the catalytic power of inexpensive iron salts to facilitate a direct [3+3] cycloaddition between oxime esters and α-unsaturated ketones. Unlike traditional methodologies that often rely on toxic solvents and precious metal catalysts, this innovation operates under solvent-free heating conditions, marking a significant paradigm shift towards green chemistry in the production of complex nitrogen-containing heterocycles. For R&D directors and procurement managers alike, this represents a tangible opportunity to streamline supply chains and reduce the overall carbon footprint of API manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyridine core has been fraught with challenges related to substrate availability, catalyst cost, and environmental sustainability. Conventional strategies often necessitate the use of expensive transition metals such as manganese or copper, which not only inflate raw material costs but also introduce significant complications regarding heavy metal residue removal in final drug products. For instance, earlier methods utilizing cyclopropyl alcohol and vinyl azide required manganese catalysis in methanol, creating a workflow that was neither atom-economical nor environmentally benign. Furthermore, other established routes involving phenylacetaldehyde and azidotrimethylsilane demanded copper salt catalysis under oxygen atmospheres in mixed solvent systems like DMF and water, often requiring extended reaction times of up to 24 hours at elevated temperatures.
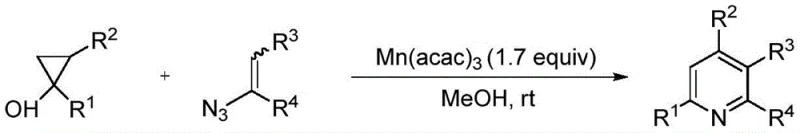
These legacy processes frequently suffer from limited substrate scope, often restricting the diversity of substituents that can be introduced at specific positions on the pyridine ring. Many existing protocols are confined to synthesizing symmetrically substituted pyridines or require pre-functionalized starting materials that are difficult and costly to prepare on a multi-kilogram scale. The reliance on organic solvents further exacerbates the issue, generating substantial volumes of hazardous waste that require expensive disposal protocols. Additionally, the use of oxidants like NHPI or specific atmospheric conditions adds layers of operational complexity, making these methods less attractive for robust, large-scale commercial production where simplicity and reliability are paramount.
The Novel Approach
In stark contrast to these cumbersome historical precedents, the methodology disclosed in CN111138345A offers a streamlined, one-step solution that directly addresses the pain points of modern chemical manufacturing. By utilizing readily available α-unsaturated ketones and ketoxime esters as starting materials, this process bypasses the need for complex substrate pre-modification. The reaction is catalyzed by ferric salts, specifically anhydrous ferric chloride, which are abundant, non-toxic, and significantly cheaper than their copper or manganese counterparts. The most transformative aspect of this technology is its operation under solvent-free conditions, where the reactants are simply heated together, drastically reducing the volume of chemical waste generated and eliminating the energy-intensive steps associated with solvent removal and recovery.
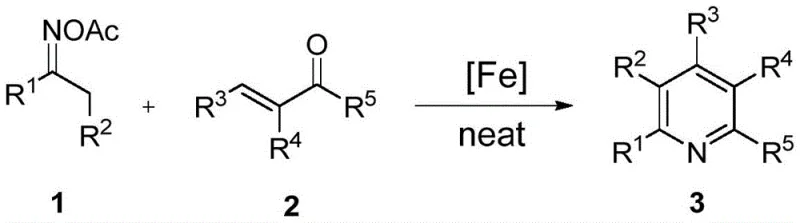
This novel approach enables the synthesis of highly selective unsymmetrical polysubstituted pyridines, a feat that many previous methods struggled to achieve without sacrificing yield or selectivity. The protocol allows for precise control over the substitution pattern on the pyridine ring by simply varying the structures of the ketene and oxime ester inputs. This flexibility is crucial for medicinal chemists who need to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies. Moreover, the reaction proceeds efficiently in air, removing the need for inert atmosphere gloveboxes or specialized equipment, thereby lowering the barrier to entry for adoption in standard manufacturing facilities and enhancing the overall safety profile of the operation.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this iron-catalyzed transformation is rooted in the principles of Michael addition followed by cyclization, a sequence that is both elegant and highly effective for carbon-carbon bond formation. The α-unsaturated ketone serves as an excellent Michael acceptor, activated by the Lewis acidic nature of the ferric salt catalyst. This activation facilitates the nucleophilic attack by the oxime ester, initiating a cascade that ultimately leads to the formation of the six-membered pyridine ring. The use of iron(III) chloride is particularly advantageous as it acts as a potent Lewis acid capable of coordinating with the carbonyl oxygen and the oxime nitrogen, thereby lowering the activation energy for the cycloaddition step. This catalytic cycle ensures high turnover numbers and allows the reaction to proceed to completion even with low catalyst loadings, typically around 0.05 equivalents.
From an impurity control perspective, the mechanism favors the formation of the desired polysubstituted product with high regioselectivity, minimizing the generation of isomeric byproducts that are common in less controlled radical processes. The solvent-free environment plays a critical role here as well; by increasing the effective concentration of the reactants, the rate of the desired bimolecular reaction is enhanced relative to potential side reactions. The robustness of the iron catalyst under aerobic conditions suggests that the mechanism does not rely on sensitive radical intermediates that would be quenched by oxygen, but rather proceeds through a stable coordination complex. This stability translates to a cleaner reaction profile, simplifying downstream purification and ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize Polysubstituted Pyridine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is remarkably straightforward, requiring minimal specialized equipment beyond standard heating and stirring apparatus. The process begins with the precise weighing of the oxime acetate and the α-unsaturated ketone, typically in a molar ratio favoring the ketone to drive the reaction to completion. These solids or liquids are combined directly in a reaction vessel along with the catalytic amount of anhydrous ferric chloride. The mixture is then heated to an optimized temperature of 140 °C, where it is maintained for a duration of approximately 3 hours. Detailed standardized synthesis steps for specific derivatives are provided in the guide below.
- Mix oxime acetate and alpha-unsaturated ketone in a molar ratio of 1: 2 with 0.05 equivalents of anhydrous ferric chloride.
- Heat the mixture to 140°C under solvent-free conditions in air for approximately 3 hours.
- Cool to room temperature, dissolve in ethyl acetate, wash with dilute ammonia and water, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology presents a compelling value proposition centered on cost efficiency and operational resilience. The elimination of organic solvents is perhaps the most significant economic driver, as it removes the recurring cost of purchasing, storing, and disposing of large volumes of volatile organic compounds. Furthermore, the work-up procedure is drastically simplified; instead of complex extraction and distillation sequences, the crude product can often be isolated by simple washing and filtration. This reduction in processing steps directly correlates to lower labor costs, reduced energy consumption, and shorter batch cycle times, all of which contribute to a leaner and more responsive manufacturing operation.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with earth-abundant iron salts results in a dramatic decrease in raw material expenditure. Copper and manganese catalysts not only carry a higher price tag but often require ligand systems that add further cost and complexity. By utilizing simple ferric chloride, the process achieves substantial cost savings without compromising on catalytic efficiency. Additionally, the solvent-free nature of the reaction means that reactors can be charged with a higher density of reactants, effectively increasing the throughput of existing infrastructure without the need for capital investment in larger vessels. This intensification of the process allows for greater production capacity within the same physical footprint.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, namely oxime esters and α-unsaturated ketones (chalcones), are commodity chemicals that are widely available from multiple global suppliers. This abundance mitigates the risk of supply disruptions that can occur with specialized or proprietary reagents required by other synthetic routes. The robustness of the reaction conditions, which tolerate air and moisture, further enhances supply chain reliability by reducing the need for stringent storage conditions or inert gas blanketing during transport and handling. This resilience ensures consistent production schedules and reliable delivery timelines for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the simplicity of this solvent-free method makes it inherently scalable. The absence of solvent removes the risk of thermal runaway associated with large volumes of flammable liquids, improving process safety. From an environmental compliance standpoint, the reduction in hazardous waste generation aligns perfectly with increasingly strict global regulations on industrial emissions and waste disposal. Companies adopting this technology can significantly improve their sustainability metrics, appealing to eco-conscious stakeholders and potentially qualifying for green manufacturing incentives, thereby future-proofing their operations against regulatory tightening.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing a reliable foundation for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into your existing production portfolio.
Q: What are the primary advantages of this iron-catalyzed method over traditional copper or manganese methods?
A: This method eliminates the need for expensive precious metal catalysts like copper or manganese and operates under solvent-free conditions, significantly reducing environmental impact and purification costs while maintaining high regioselectivity.
Q: Can this synthesis method accommodate diverse functional groups on the pyridine ring?
A: Yes, the process demonstrates excellent functional group tolerance, allowing for the introduction of nitro, methyl, methoxy, cyano, and halogen substituents, enabling the synthesis of a wide variety of unsymmetrical polysubstituted pyridines.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The solvent-free nature of the reaction simplifies the work-up procedure by removing the need for solvent recovery systems, and the use of earth-abundant iron salts makes it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iron-catalyzed synthesis of polysubstituted pyridines described in CN111138345A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite you to explore how this advanced synthetic route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us today to request specific COA data for our pyridine derivatives and to discuss route feasibility assessments for your next project. Let us partner with you to drive innovation and efficiency in your drug development pipeline.
