Scalable Synthesis of Novel Tetrahydrofuran Intermediates for Next-Generation Prostate Cancer Therapeutics
Scalable Synthesis of Novel Tetrahydrofuran Intermediates for Next-Generation Prostate Cancer Therapeutics
The landscape of oncology drug development, particularly for prostate cancer, has been revolutionized by androgen receptor inhibitors such as Enzalutamide (MDV3100) and Apalutamide (ARN-509). However, the manufacturing of these potent therapeutics often faces significant bottlenecks related to the synthesis of their complex spiro-hydantoin cores. Patent CN110669027B, filed in 2021, presents a transformative approach to this challenge by introducing novel substituted 3-(4-methylcarbamoyl-3-fluorophenylamino)tetrahydrofuran-3-carboxylic acid derivatives. These compounds serve as critical intermediates that streamline the production pathway, offering a robust alternative to traditional methods that rely heavily on costly chiral separations. By shifting the stereochemical control to an earlier stage of the synthesis, this technology enables more efficient access to high-purity active pharmaceutical ingredients.
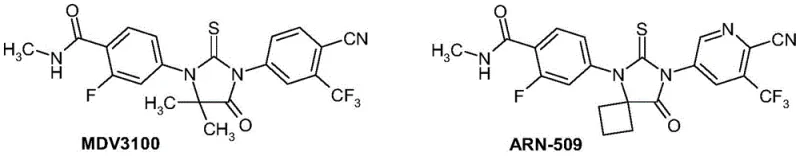
For R&D directors and process chemists, the structural integrity and purity of these intermediates are paramount. The patent details specific embodiments where the tetrahydrofuran ring is functionalized with ester groups (methyl or butyl) or protected amine groups, providing versatile handles for downstream processing. This level of molecular precision ensures that the final API meets the stringent regulatory standards required for global distribution. As a reliable pharmaceutical intermediate supplier, understanding these nuanced structural variations is key to delivering consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-[3-(3-trifluoromethyl-4-cyanophenyl)-4-oxo-2-thioxo-7-oxa-1,3-diazaspiro[4.4]non-1-yl]-2-fluoro-N-methylbenzamide (Compound A1) and its stereoisomers has been fraught with inefficiencies. As illustrated in the prior art schemes, the conventional route typically involves the direct interaction of 4-thioisocyanate-2-(trifluoromethyl)benzonitrile with a racemic amino-tetrahydrofuran derivative under harsh conditions, such as heating in dimethylformamide at 100°C for extended periods. The resulting product is a racemic mixture that necessitates separation into its optical isomers (A2 and A3) using high-pressure liquid chromatography (HPLC) with chiral columns. This reliance on preparative chiral HPLC is a major economic and operational burden, as it limits throughput, increases solvent consumption, and drastically escalates the cost of goods sold (COGS).
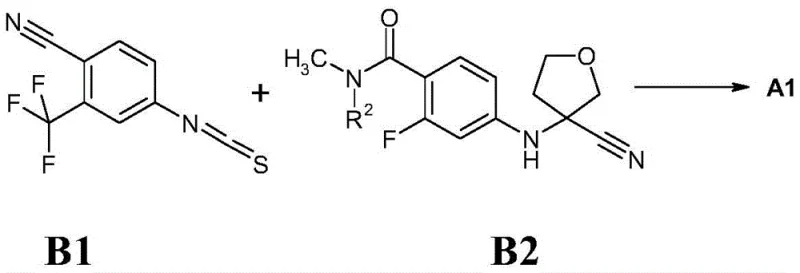
The Novel Approach
The innovation disclosed in CN110669027B circumvents these bottlenecks by introducing a pre-functionalized intermediate strategy. Instead of forming the complex spiro-cycle first and then resolving it, the new method focuses on synthesizing high-purity chiral intermediates of general formula 1. By utilizing chiral starting materials or optimized coupling conditions, the process generates the requisite stereochemistry before the final cyclization step. This strategic shift eliminates the need for difficult chiral chromatography on the final, highly lipophilic spiro-compound. Furthermore, the new route employs milder reaction conditions and more accessible reagents, facilitating cost reduction in API manufacturing. The ability to isolate intermediates like the methyl ester 1(2) or butyl ester 1(3) in high yields provides a stable platform for large-scale production.
Mechanistic Insights into Copper-Catalyzed C-N Bond Formation
The core of this synthetic breakthrough lies in the efficient construction of the C-N bond between the fluorobenzamide moiety and the tetrahydrofuran ring. The patent describes a copper-catalyzed coupling reaction, reminiscent of Ullmann-type condensation, which is pivotal for linking the aromatic system with the heterocyclic amine. Specifically, the reaction of 4-bromo-2-fluoro-N-methylbenzamide (Compound 2) with 3-aminotetrahydrofuran-3-carboxylic acid esters (Compound 3.2) is mediated by Copper(I) iodide. The presence of bases like potassium carbonate and ligands such as 2-acetylcyclohexanone plays a critical role in stabilizing the copper species and facilitating the oxidative addition and reductive elimination steps of the catalytic cycle. This mechanistic pathway ensures high conversion rates even with sterically hindered substrates.
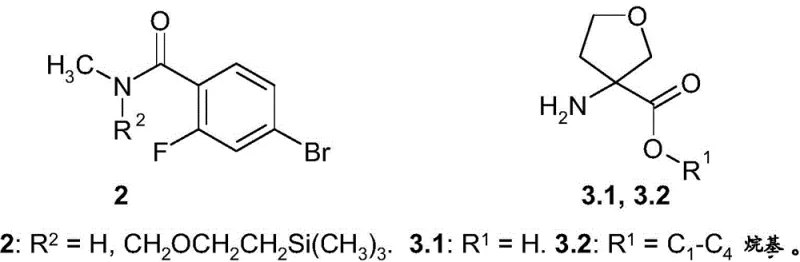
Impurity control is another significant advantage of this mechanism. In traditional routes, side reactions such as hydrolysis of the nitrile group or decomposition of the isothiocyanate can lead to complex impurity profiles that are difficult to purge. By isolating the intermediate of formula 1, manufacturers can implement rigorous quality control checks before proceeding to the final cyclization. The intermediate can be purified via simple crystallization or acid-base extraction, removing metal residues and unreacted starting materials effectively. This results in a cleaner reaction profile for the subsequent step, where the intermediate reacts with 4-isothiocyanato-2-trifluoromethylbenzonitrile to form the final hydantoin ring, thereby ensuring the final API meets strict impurity specifications.
How to Synthesize Substituted Tetrahydrofuran Intermediates Efficiently
The preparation of these high-value intermediates follows a logical sequence designed for scalability and reproducibility. The process begins with the coupling of the bromo-benzamide and the amino-tetrahydrofuran ester in a polar aprotic solvent system, followed by workup procedures that maximize recovery. The detailed standardized synthesis steps for producing these intermediates are outlined below.
- React 4-bromo-2-fluoro-N-methylbenzamide with 3-aminotetrahydrofuran-3-carboxylic acid esters using Copper(I) iodide catalyst in DMF/Water.
- Purify the resulting intermediate ester via acidification and filtration to achieve high purity without chromatography.
- Couple the purified intermediate with 4-isothiocyanato-2-trifluoromethylbenzonitrile in DMSO/Ethyl Acetate to form the final spiro-hydantoin structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible operational benefits. The primary advantage is the decoupling of the chiral resolution step from the final drug substance manufacturing. By securing the stereochemistry at the intermediate stage, the supply chain becomes more resilient against the bottlenecks associated with chiral column availability and capacity. This leads to enhanced supply chain reliability, ensuring that production schedules are met without unexpected delays caused by purification failures.
- Cost Reduction in Manufacturing: The elimination of preparative chiral HPLC for the final product represents a massive reduction in processing costs. Chromatographic separations on a multi-kilogram scale require expensive stationary phases and vast quantities of high-grade solvents. By replacing this with crystallization-based purifications of the intermediate, the overall manufacturing cost is significantly lowered. Additionally, the higher yields reported in the patent (up to 70-80% for intermediate formation) mean less raw material waste, further driving down the cost per kilogram of the final API.
- Enhanced Supply Chain Reliability: The reagents used in this novel pathway, such as copper iodide and common organic solvents like DMF and ethyl acetate, are commodity chemicals with stable global supply chains. Unlike specialized chiral catalysts or bespoke reagents that might have long lead times, these materials are readily available from multiple vendors. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages, providing a more secure sourcing strategy for long-term commercial contracts.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing water as a co-solvent in the coupling step and avoiding excessive use of chlorinated solvents where possible. The simplified workup procedures reduce the volume of hazardous waste generated per batch. This aligns with increasingly stringent environmental regulations and facilitates easier permitting for commercial scale-up of complex pharmaceutical intermediates. The robustness of the reaction conditions also allows for easy transfer from pilot plant to full-scale production reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation.
Q: How does this new route improve upon the conventional synthesis of Enzalutamide intermediates?
A: The conventional route often requires separating racemic mixtures of the final complex spiro-hydantoin using expensive chiral HPLC. This patent introduces a chiral tetrahydrofuran intermediate that allows for stereochemical control earlier in the synthesis, significantly simplifying purification.
Q: What catalysts are essential for the formation of the C-N bond in this process?
A: The process utilizes Copper(I) iodide (CuI) as the primary catalyst, often in conjunction with potassium carbonate and specific ligands like 2-acetylcyclohexanone, to facilitate the coupling between the bromo-benzamide and the amino-tetrahydrofuran moiety.
Q: Can this method be scaled for commercial production of ARN-509 precursors?
A: Yes, the methodology avoids sensitive reagents and relies on robust conditions such as refluxing in polar aprotic solvents, making it highly suitable for kilogram-to-ton scale manufacturing typical of API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Androgen Receptor Inhibitor Intermediate Supplier
The technological advancements described in CN110669027B highlight the critical need for partners who can navigate complex heterocyclic chemistry with precision. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactor systems capable of handling the specific temperature and pressure requirements of copper-catalyzed couplings and subsequent cyclizations. We maintain stringent purity specifications through our rigorous QC labs, ensuring that every batch of intermediate delivered meets the exacting standards required for oncology drug development.
We invite global pharmaceutical partners to collaborate with us to optimize their supply chains for next-generation prostate cancer therapies. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments for these novel tetrahydrofuran intermediates, and let us help you accelerate your path to market.
