Advanced Convergent Synthesis of SGLT2 Inhibitor Intermediates for Commercial Scale-Up
Introduction to Next-Generation SGLT2 Inhibitor Synthesis
The global pharmaceutical landscape is witnessing an unprecedented surge in the demand for Sodium-Glucose Cotransporter 2 (SGLT2) inhibitors, a critical class of therapeutics for managing type 2 diabetes. As patent CN114478454A elucidates, the current market leaders such as Canagliflozin, Dapagliflozin, and Empagliflozin share a distinct structural architecture comprising a glycosidic sugar ring linked to a biphenyl or heteroaryl system. 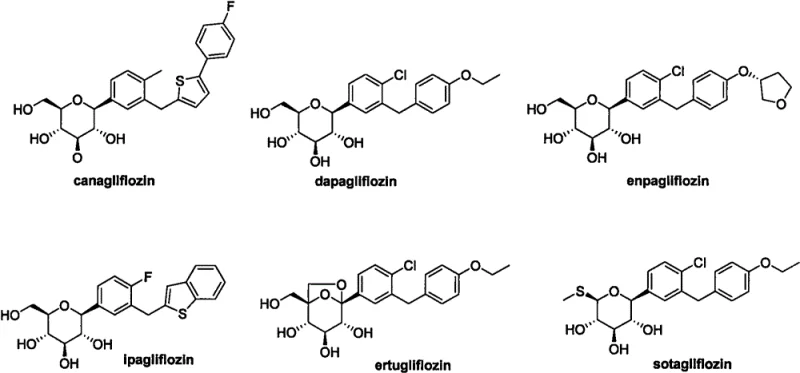 From a process chemistry perspective, optimizing the synthesis of these complex molecules is paramount for ensuring long-term supply security and cost-effectiveness. The patent introduces a novel key intermediate and a preparation method that fundamentally rethinks the disconnection strategy, moving away from linear sequences toward a more convergent and economically viable approach. This technological breakthrough addresses the critical pain points of high material costs and low overall yields that have historically plagued the industrial production of these life-saving medications.
From a process chemistry perspective, optimizing the synthesis of these complex molecules is paramount for ensuring long-term supply security and cost-effectiveness. The patent introduces a novel key intermediate and a preparation method that fundamentally rethinks the disconnection strategy, moving away from linear sequences toward a more convergent and economically viable approach. This technological breakthrough addresses the critical pain points of high material costs and low overall yields that have historically plagued the industrial production of these life-saving medications.
Structurally, these inhibitors can be deconstructed into three primary fragments: the leftmost sugar ring, a central aromatic heterocyclic or aromatic portion, and the rightmost aromatic ring. 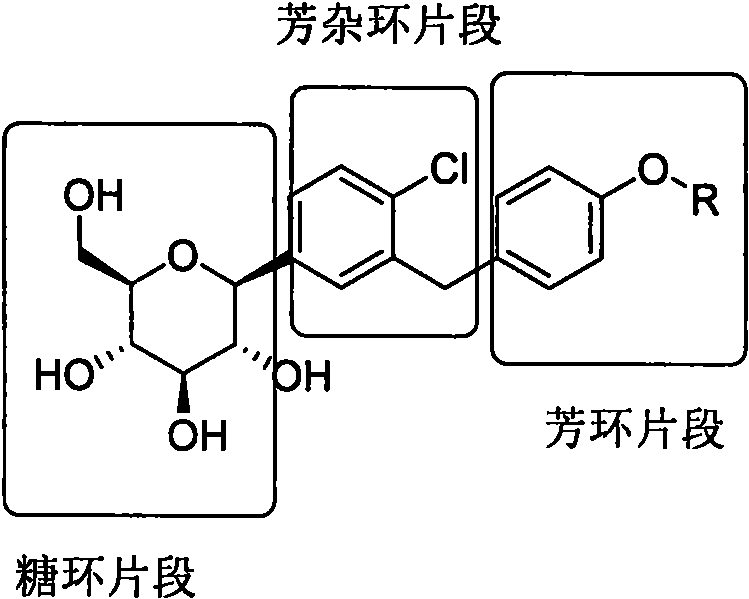 Traditional synthetic methodologies often prioritize the construction of the aromatic core first, followed by the challenging docking of the sugar moiety. However, the disclosed invention proposes a paradigm shift by prioritizing the assembly of the sugar and central aromatic ring system first. This strategic rearrangement allows for the introduction of the most expensive and structurally diverse right-hand aromatic fragment at the latest possible stage, thereby maximizing material efficiency and minimizing the financial impact of any potential yield losses in earlier processing steps.
Traditional synthetic methodologies often prioritize the construction of the aromatic core first, followed by the challenging docking of the sugar moiety. However, the disclosed invention proposes a paradigm shift by prioritizing the assembly of the sugar and central aromatic ring system first. This strategic rearrangement allows for the introduction of the most expensive and structurally diverse right-hand aromatic fragment at the latest possible stage, thereby maximizing material efficiency and minimizing the financial impact of any potential yield losses in earlier processing steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the dominant synthetic routes for SGLT2 inhibitors have relied on constructing the complex aromatic or heteroaromatic ring system prior to glycosylation. 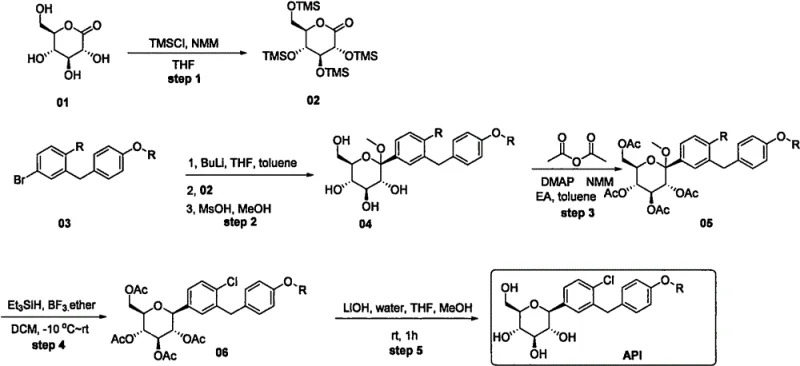 This linear approach presents significant economic and operational drawbacks for large-scale manufacturing. The primary limitation is the premature introduction of high-cost heterocyclic fragments. If any step following the attachment of this expensive moiety suffers from poor yield or requires extensive purification, the financial loss is magnified because the value of the intermediate has already been inflated by the costly starting material. Furthermore, these early-stage complex fragments often require harsh reaction conditions that can be incompatible with the sensitive protecting groups needed for the sugar moiety, leading to complicated protection-deprotection sequences that increase both the step count and the environmental footprint of the process.
This linear approach presents significant economic and operational drawbacks for large-scale manufacturing. The primary limitation is the premature introduction of high-cost heterocyclic fragments. If any step following the attachment of this expensive moiety suffers from poor yield or requires extensive purification, the financial loss is magnified because the value of the intermediate has already been inflated by the costly starting material. Furthermore, these early-stage complex fragments often require harsh reaction conditions that can be incompatible with the sensitive protecting groups needed for the sugar moiety, leading to complicated protection-deprotection sequences that increase both the step count and the environmental footprint of the process.
The Novel Approach
In stark contrast, the novel methodology described in the patent employs a convergent strategy that decouples the synthesis of the expensive right-hand aromatic ring from the core scaffold construction. 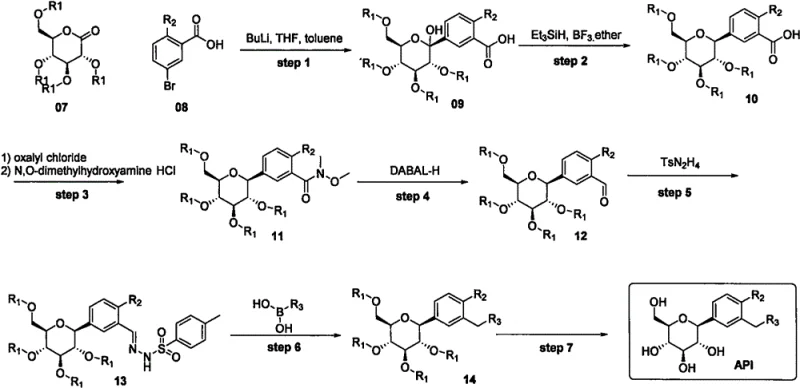 By first establishing the robust connection between the sugar ring and the central chlorophenyl ring, the process creates a versatile intermediate that can be stored or transported before the final diversification step. The key innovation lies in the use of a Barluenga-type coupling reaction to install the final aromatic fragment. This allows manufacturers to utilize a single, common intermediate for the production of multiple different SGLT2 inhibitors simply by swapping the boronic acid coupling partner. This modularity not only streamlines inventory management but also drastically reduces the cost of goods sold (COGS) by deferring the expenditure on expensive reagents until the very end of the synthesis, ensuring that high-value materials are not wasted on failed upstream reactions.
By first establishing the robust connection between the sugar ring and the central chlorophenyl ring, the process creates a versatile intermediate that can be stored or transported before the final diversification step. The key innovation lies in the use of a Barluenga-type coupling reaction to install the final aromatic fragment. This allows manufacturers to utilize a single, common intermediate for the production of multiple different SGLT2 inhibitors simply by swapping the boronic acid coupling partner. This modularity not only streamlines inventory management but also drastically reduces the cost of goods sold (COGS) by deferring the expenditure on expensive reagents until the very end of the synthesis, ensuring that high-value materials are not wasted on failed upstream reactions.
Mechanistic Insights into Barluenga Cross-Coupling
The core of this technological advancement is the application of the Barluenga coupling reaction, a powerful tool for forming carbon-carbon bonds between tosylhydrazones and boronic acids. Unlike traditional palladium-catalyzed cross-couplings that might require sensitive organohalides or strict anhydrous conditions, this mechanism proceeds through a diazo intermediate generated in situ from the tosylhydrazone. In the context of SGLT2 synthesis, the aldehyde functionality on the central aromatic ring is converted into a tosylhydrazone, which then reacts with the desired aryl boronic acid under basic conditions. This mechanism is particularly advantageous for pharmaceutical manufacturing because it tolerates a wide range of functional groups present on the sugar moiety, such as hydroxyls and ethers, without requiring excessive protection. The reaction typically proceeds with high stereoselectivity, preserving the critical beta-configuration of the glycosidic bond which is essential for biological activity.
From an impurity control perspective, this mechanistic pathway offers superior cleanliness compared to alternative alkylation or coupling methods. The byproducts of the reaction, primarily nitrogen gas and sulfinate salts, are easily removed during the aqueous workup, leaving behind a high-purity organic phase. This reduces the burden on downstream purification processes such as chromatography or recrystallization, which are often the bottlenecks in API production. The ability to generate the final biaryl linkage under relatively mild thermal conditions further minimizes the risk of epimerization at the anomeric center, a common degradation pathway in carbohydrate chemistry that can lead to difficult-to-separate diastereomeric impurities. Consequently, this route ensures a cleaner impurity profile, facilitating regulatory approval and reducing the risk of batch rejection.
How to Synthesize SGLT2 Inhibitor Intermediates Efficiently
The synthesis of the key intermediate involves a sequence of highly controlled transformations starting from protected glucono-lactone derivatives. The process begins with the lithiation of a brominated aromatic precursor, followed by nucleophilic attack on the lactone to establish the C-C bond between the sugar and the aromatic ring. Subsequent reduction and functional group manipulations yield the crucial aldehyde intermediate. This aldehyde is then transformed into the reactive tosylhydrazone species, setting the stage for the final coupling event. The detailed standardized operating procedures, including specific stoichiometric ratios, temperature gradients, and solvent systems required to execute this synthesis with GMP compliance, are outlined in the comprehensive guide below.
- Preparation of the protected glucono-lactone derivative and subsequent lithiation to attach the central aromatic ring containing a chloro substituent and an aldehyde or protected alcohol group.
- Conversion of the aldehyde functionality into a tosylhydrazone intermediate using p-toluenesulfonyl hydrazide under heating conditions in dioxane.
- Execution of the Barluenga coupling reaction by reacting the tosylhydrazone with the corresponding aryl boronic acid in the presence of a base like potassium carbonate to yield the final coupled product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates directly into enhanced operational resilience and cost predictability. The shift towards a convergent synthesis model mitigates the risk associated with volatile pricing of specialized heterocyclic building blocks. By postponing the incorporation of these high-value fragments, the manufacturer effectively insulates the bulk of the production process from supply shocks affecting niche raw materials. This strategic sourcing approach ensures that the majority of the synthetic workload relies on commodity chemicals and bulk solvents, which are subject to more stable market dynamics and are readily available from multiple qualified vendors globally.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the principle of late-stage diversification. In traditional linear syntheses, a yield loss in the final steps results in the wastage of all accumulated value, including the expensive aromatic fragments. By contrast, this convergent route ensures that the costly coupling partners are only introduced when the core scaffold is fully validated and ready. This significantly reduces the cost of wasted materials and lowers the overall capital tied up in work-in-progress inventory. Furthermore, the elimination of complex protection-deprotection sequences reduces solvent consumption and waste disposal costs, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The modular nature of this synthesis enhances supply chain continuity. Since the key intermediate is common to multiple SGLT2 inhibitors, manufacturers can maintain a strategic stockpile of this semi-finished good. When demand fluctuates for specific drugs like Dapagliflozin versus Empagliflozin, production can be rapidly pivoted by simply altering the final coupling step without needing to restart the entire synthesis from the sugar starting material. This agility allows for faster response times to market demands and reduces the lead time for delivering high-purity pharmaceutical intermediates to downstream API producers, ensuring uninterrupted drug availability for patients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing robust reaction conditions that translate well from laboratory to pilot and commercial scales. The avoidance of highly pyrophoric reagents in the final coupling step simplifies safety protocols and reduces the need for specialized containment equipment. Additionally, the generation of benign byproducts aligns with modern green chemistry principles, facilitating easier waste treatment and regulatory compliance. This environmental stewardship not only reduces the ecological footprint but also minimizes the risk of regulatory delays related to effluent discharge, ensuring a smoother path to commercial authorization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the feasibility and advantages of adopting this route for industrial production.
Q: What is the primary cost advantage of this new synthetic route compared to conventional methods?
A: The primary advantage lies in the delayed introduction of the expensive right-hand aromatic fragment. Conventional routes often attach complex heterocycles early, risking high material loss if upstream steps fail. This convergent approach builds the core sugar-aromatic scaffold first, minimizing financial risk.
Q: How does the Barluenga coupling improve process scalability?
A: The Barluenga coupling utilizes stable tosylhydrazone intermediates and commercially available boronic acids. This avoids the need for sensitive organometallic reagents in the final coupling step, making the process more robust and easier to control during large-scale manufacturing.
Q: Can this intermediate platform be used for multiple SGLT2 inhibitors?
A: Yes, the patent highlights that the key intermediate (Formula I) serves as a common precursor. By simply varying the boronic acid component in the final coupling step, manufacturers can access a series of different SGLT2 inhibitors like Dapagliflozin or Empagliflozin from a single shared intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SGLT2 Inhibitor Intermediate Supplier
As the global demand for diabetes therapeutics continues to escalate, securing a reliable supply of high-quality intermediates is critical for pharmaceutical success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced process technologies like the one described in CN114478454A to deliver superior value. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of multinational corporations. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of SGLT2 intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and discover how our innovative synthesis platforms can drive efficiency and profitability in your API manufacturing operations.
