Advanced Green Synthesis of Quinoxaline Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and environmentally benign pathways for constructing nitrogen-containing heterocycles. Patent CN101481357A introduces a transformative methodology for the preparation of quinoxaline derivatives, addressing critical pain points in traditional organic synthesis. This innovation leverages heterogeneous acid catalysis, specifically utilizing Amberlyst-15 or NKC-9 dry hydrogen catalytic resin (Styrene-DVB), to facilitate the condensation of o-diamino compounds with o-dicarbonyl compounds. Unlike legacy processes that rely heavily on volatile organic compounds, this protocol employs ordinary tap water as the reaction medium, operating effectively within a mild temperature range of 25°C to 100°C. For R&D directors and process chemists, this represents a significant leap forward in green chemistry, offering a route that not only simplifies downstream processing but also aligns with increasingly stringent global environmental regulations regarding solvent emissions and waste disposal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline scaffolds has been dominated by thermal condensation reactions conducted in solvents such as ethanol or acetic acid under reflux conditions. These traditional methodologies suffer from several inherent inefficiencies that impact both operational expenditure and environmental compliance. Typically, these reactions require prolonged heating periods ranging from 2 to 12 hours to reach completion, which translates to substantial energy consumption in a commercial setting. Furthermore, the reliance on organic solvents necessitates complex distillation and recovery systems to meet safety and environmental standards, adding layers of complexity to the manufacturing workflow. Perhaps most critically, the separation of the final product from the reaction mixture often involves cumbersome work-up procedures, and the lack of effective catalysis can lead to variable yields, sometimes falling as low as 34% to 85%, thereby reducing the overall atom economy of the process.
The Novel Approach
The technology disclosed in CN101481357A fundamentally reengineers this transformation by introducing a solid acid catalyst system that operates in an aqueous environment. By replacing toxic organic solvents with water, the process eliminates the fire hazards and health risks associated with volatile organic compounds (VOCs). The use of Amberlyst-15 or NKC-9 resins provides a highly active acidic surface that accelerates the condensation kinetics, reducing reaction times drastically to between 10 and 60 minutes. This shift not only enhances throughput but also simplifies the isolation of the target molecule; the solid catalyst can be removed via simple filtration, allowing for immediate recycling. This approach ensures high purity and yield while minimizing the generation of hazardous waste streams, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
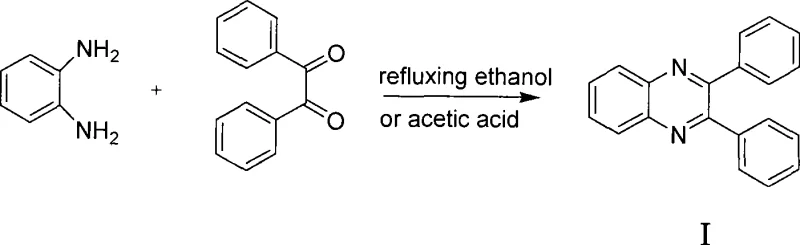
Mechanistic Insights into Heterogeneous Acid-Catalyzed Cyclization
The efficacy of this synthesis lies in the mechanistic role of the sulfonic acid groups present on the polymeric resin catalyst. In the aqueous medium, these strong acid sites protonate the carbonyl oxygen of the o-dicarbonyl substrate, significantly increasing its electrophilicity. This activation facilitates a nucleophilic attack by the amino groups of the o-diamine reactant, leading to the formation of a hemiaminal intermediate. Subsequent dehydration steps, driven by the acidic environment and thermal energy, promote the cyclization required to form the stable quinoxaline ring system. The heterogeneous nature of the catalyst ensures that the reaction occurs primarily at the solid-liquid interface, which helps in controlling side reactions such as polymerization or over-oxidation that might occur in homogeneous acidic solutions. This precise control over the reaction pathway is crucial for maintaining a clean impurity profile, a key metric for regulatory approval in drug substance production.
Furthermore, the substrate scope of this reaction is remarkably broad, accommodating various electronic and steric environments on both the diamine and dicarbonyl components. As illustrated in the structural variations below, electron-withdrawing groups like nitro substituents or electron-donating groups like methyl groups on the aromatic ring do not inhibit the reaction efficiency. This versatility is vital for medicinal chemists who need to generate diverse libraries of analogues for structure-activity relationship (SAR) studies. The ability to tolerate functional groups without the need for extensive protecting group strategies streamlines the synthetic route, reducing the number of unit operations and overall material costs. The robustness of the catalyst also means that it can withstand the reaction conditions without leaching significant amounts of metal or organic contaminants into the product stream.
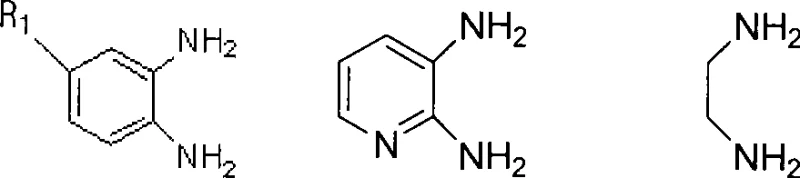
How to Synthesize Quinoxaline Derivatives Efficiently
Implementing this green synthesis protocol requires careful attention to the molar ratios of reactants and the loading of the solid catalyst to maximize turnover frequency. The standard procedure involves suspending the catalyst in water before adding the substrates to ensure uniform dispersion and optimal contact between the reactive species and the active sites. Monitoring the reaction progress via thin-layer chromatography (TLC) allows for precise determination of the endpoint, preventing unnecessary energy usage once conversion is complete. The following guide outlines the standardized operational parameters derived from the patent examples to ensure reproducible high-yield results suitable for scale-up.
- Mix o-diamino compound and o-dicarbonyl compound in water with Amberlyst-15 or NKC-9 catalyst.
- Stir the reaction mixture at temperatures between 25°C and 100°C for 10 to 60 minutes.
- Filter to recover the catalyst, extract the product with ethyl acetate, and purify via recrystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this water-based catalytic process offers tangible strategic benefits beyond mere technical feasibility. The elimination of expensive and hazardous organic solvents directly correlates to a reduction in raw material procurement costs and lowers the logistical burden associated with solvent storage and handling. Additionally, the ability to recycle the solid acid catalyst multiple times without regeneration losses significantly decreases the cost of goods sold (COGS) over the lifecycle of the product. This process stability ensures a reliable quinoxaline derivatives supplier can maintain consistent output levels without being disrupted by supply chain volatility associated with specialty solvents or precious metal catalysts.
- Cost Reduction in Manufacturing: The transition from homogeneous liquid acids or expensive transition metal catalysts to a cheap, polymeric solid acid resin results in substantial cost savings. Since the catalyst can be recovered by simple filtration and reused for multiple batches, the amortized cost per kilogram of product is drastically lowered. Furthermore, the use of tap water as a solvent removes the need for costly solvent recovery distillation columns and reduces the energy footprint of the facility, contributing to a leaner and more competitive manufacturing cost structure.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as o-phenylenediamines and benzils, are commodity chemicals with established global supply chains, reducing the risk of bottlenecks. The simplicity of the reaction setup, which does not require specialized high-pressure equipment or inert atmosphere conditions, allows for flexible production scheduling across multiple manufacturing sites. This flexibility ensures that high-purity quinoxaline derivatives can be delivered with shorter lead times, supporting just-in-time inventory models for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic runaway risks often associated with strong liquid acids. The aqueous waste stream is significantly easier to treat compared to mixed organic solvent waste, simplifying compliance with environmental protection regulations. This green profile enhances the corporate sustainability metrics of the manufacturer, making the supply chain more resilient against future regulatory tightening regarding industrial emissions and chemical safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders evaluating this synthetic route.
Q: Can the solid acid catalyst be recycled in this quinoxaline synthesis?
A: Yes, the patent specifies that catalysts like Amberlyst-15 or NKC-9 can be recovered by simple filtration and regenerated with sulfuric acid treatment, allowing for reuse at least three to five times without significant loss of activity.
Q: Does this method require toxic organic solvents?
A: No, a key advantage of this technology is the use of ordinary tap water as the sole polar solvent, eliminating the need for volatile and toxic organic solvents typically used in traditional reflux methods.
Q: What is the typical reaction time compared to conventional methods?
A: While conventional methods often require 2 to 12 hours of reflux, this catalytic process significantly reduces reaction time to between 10 and 60 minutes under mild temperature conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting sustainable and efficient synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team has extensively evaluated the water-based condensation protocol described in CN101481357A and validated its potential for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial supply is seamless. Our facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of quinoxaline intermediates meets the highest international standards for drug substance manufacturing.
We invite pharmaceutical companies and agrochemical manufacturers to collaborate with us to leverage this advanced green chemistry platform. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized supply chain can drive value and efficiency for your next-generation therapeutic programs.
