Advanced Two-Step Synthesis of Molnupiravir Intermediates Using Lewis Acid Catalysis for Commercial Scale-Up
Introduction to Next-Generation Antiviral Synthesis
The global demand for effective antiviral therapeutics has necessitated the rapid development of robust and scalable synthetic routes for key active pharmaceutical ingredients (APIs). Patent CN114560894B discloses a groundbreaking preparation method for Molnupiravir (Compound I), a potent ribonucleoside analogue exhibiting broad-spectrum activity against RNA viruses including SARS-CoV-2. Unlike traditional multi-step syntheses that suffer from low overall yields and complex purification requirements, this invention leverages a sophisticated Lewis acid-catalyzed strategy to streamline production. The core innovation lies in the direct transformation of inexpensive uridine into the target molecule through a highly efficient two-step sequence, bypassing the need for cumbersome protecting group manipulations. This technological leap represents a significant advancement for any reliable API intermediate supplier seeking to optimize their manufacturing portfolio.
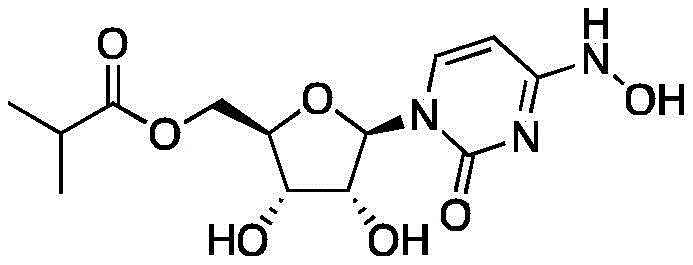
By utilizing readily available starting materials and minimizing unit operations, this process addresses critical bottlenecks in the supply chain for antiviral medications. The method not only enhances the economic viability of large-scale production but also aligns with green chemistry principles by reducing solvent usage and waste generation. For pharmaceutical manufacturers, adopting this route means securing a more resilient supply of high-quality intermediates capable of meeting stringent regulatory standards. The following analysis delves into the mechanistic advantages and commercial implications of this novel synthetic pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Molnupiravir has been plagued by inefficiencies inherent in protecting group chemistry and enzymatic limitations. As illustrated in prior art such as WO2019113462, conventional routes typically involve a laborious sequence where the cis-dihydroxyl groups of the ribose sugar must first be protected, often using acetone and sulfuric acid, before acylation can occur. This is followed by activation with phosphorus oxychloride and triazole, reaction with hydroxylamine, and finally, a deprotection step to reveal the final product. Such multi-step processes inevitably lead to cumulative yield losses, with total yields reported as low as 17%, creating substantial material waste and driving up costs.
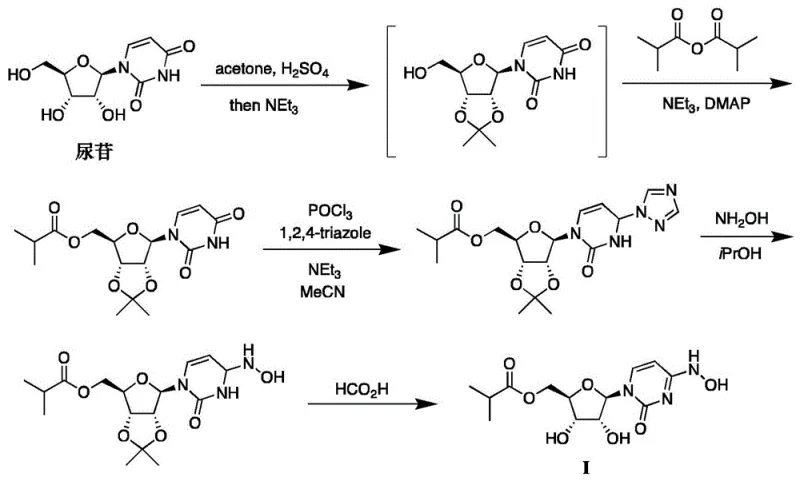
Furthermore, alternative enzymatic approaches, while offering shorter step counts, introduce their own set of supply chain vulnerabilities. The reliance on specialized lipases like Novozyme 435 imposes a heavy financial burden due to the high cost of biocatalysts and the specific conditions required for their activity. Additionally, enzymatic processes can be sensitive to substrate concentration and solvent compatibility, limiting their flexibility in large-scale industrial reactors. These factors combined make conventional methods less attractive for manufacturers aiming for cost reduction in pharmaceutical manufacturing and consistent high-volume output.
The Novel Approach
In stark contrast, the method disclosed in CN114560894B revolutionizes the synthesis by employing a direct, regioselective acylation strategy. The process begins with the reaction of uridine and isobutyric anhydride in the presence of a Lewis acid catalyst, such as copper trifluoromethane sulfonate. This single step achieves selective esterification at the 5'-position with exceptional efficiency, yielding the intermediate Compound IV in up to 92% yield without the need for prior protection of the 2' and 3' hydroxyl groups. This elimination of protection steps drastically shortens the synthetic timeline and reduces the consumption of auxiliary reagents.
The second stage involves a clever tandem activation and substitution reaction. The intermediate is treated with a silicon reagent system, typically hexamethyldisilazane combined with trimethylsilyl triflate, which activates the carbonyl group for nucleophilic attack by hydroxylamine. This one-pot transformation effectively installs the oxime functionality while simultaneously managing the reaction environment to prevent degradation. The result is a streamlined two-step process that delivers Molnupiravir with high purity and yield, offering a compelling alternative for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Lewis Acid-Catalyzed Regioselective Acylation
The success of this synthetic route hinges on the precise mechanistic role of the Lewis acid catalyst in directing the acylation reaction. In the absence of a catalyst, acylation of uridine with isobutyric anhydride would likely result in a mixture of products due to the similar reactivity of the 2', 3', and 5' hydroxyl groups. However, the introduction of metal triflates like Cu(OTf)2 or Zn(OTf)2 creates a coordination complex with the ribose ring oxygen and adjacent hydroxyls. This coordination effectively masks the secondary hydroxyls sterically and electronically, leaving the primary 5'-hydroxyl group as the most accessible and nucleophilic site for the anhydride attack. This phenomenon ensures that the reaction proceeds with high regioselectivity, minimizing the formation of difficult-to-separate isomeric byproducts.
Following the acylation, the conversion to the oxime requires careful activation of the pyrimidine base. The silicon reagent system serves a dual purpose: hexamethyldisilazane acts as a silylating agent that protects sensitive functionalities transiently, while the trimethylsilyl triflate acts as a potent Lewis acid to activate the C4-carbonyl of the nucleobase. This activation increases the electrophilicity of the carbon center, facilitating the nucleophilic addition of hydroxylamine. The subsequent elimination of the silyl group and tautomerization leads to the stable oxime configuration found in Molnupiravir. Understanding this mechanism is crucial for R&D teams aiming to replicate the process, as slight deviations in catalyst loading or reagent ratios could impact the impurity profile.
How to Synthesize Molnupiravir Efficiently
The implementation of this synthesis requires strict adherence to the optimized conditions described in the patent to ensure maximum yield and purity. The process is designed to be operationally simple, utilizing common solvents like butanone and ethyl acetate which are easily recovered and recycled. The initial acylation is performed at mild temperatures, reducing energy consumption, while the subsequent oximation step utilizes thermal activation to drive the reaction to completion. Detailed standardized operating procedures regarding reagent addition rates, temperature control profiles, and workup protocols are essential for transferring this technology from the laboratory to the pilot plant.
- Perform regioselective acylation of Uridine with isobutyric anhydride using a Lewis acid catalyst (e.g., Copper Triflate) to form the 5'-isobutyrate intermediate.
- React the intermediate with hydroxylamine in the presence of a silicon reagent (e.g., HMDS/TMSOTf) to effect simultaneous activation and oximation.
- Purify the final crude product via recrystallization to achieve high-purity Molnupiravir suitable for API manufacturing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Lewis acid-catalyzed route offers transformative benefits that extend beyond mere technical feasibility. The most immediate impact is seen in the drastic simplification of the raw material list. By eliminating the need for expensive protecting group reagents like acetone dimethyl acetal and specialized enzymes, the bill of materials becomes significantly leaner. This reduction in material complexity directly translates to lower procurement risks, as the supply chain relies on commodity chemicals that are widely available from multiple global vendors, ensuring continuity of supply even during market fluctuations.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven by the substantial reduction in unit operations. Conventional routes require multiple isolation and purification steps between protection, acylation, and deprotection, each incurring labor, solvent, and energy costs. By condensing the synthesis into two high-yielding steps, the new method minimizes processing time and equipment occupancy. Furthermore, the high selectivity of the Lewis acid catalyst reduces the burden on downstream purification, leading to significant savings in chromatography media and crystallization solvents, ultimately driving down the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Dependence on proprietary enzymes or scarce chiral catalysts poses a risk to supply chain stability. This chemical synthesis utilizes robust, non-biological catalysts that have long shelf lives and are not subject to the batch-to-batch variability often seen with biocatalysts. The use of standard chemical reagents allows for flexible sourcing strategies, enabling manufacturers to negotiate better pricing and secure long-term contracts. This reliability is critical for meeting the demanding delivery schedules of global pharmaceutical partners who require consistent quality and volume.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the shorter synthetic route generates significantly less waste. The elimination of acidic deprotection steps and the reduction in solvent volumes contribute to a lower E-factor (mass of waste per mass of product). This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The process is inherently scalable, as the exothermic nature of the acylation can be easily managed in large reactors, and the workup involves simple phase separations rather than complex distillations, making it ideal for multi-ton production campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into existing manufacturing facilities.
Q: How does the Lewis acid catalyst improve regioselectivity in Molnupiravir synthesis?
A: The Lewis acid catalyst, such as copper trifluoromethane sulfonate, coordinates specifically with the ribose hydroxyl groups, directing the acylation exclusively to the primary 5'-hydroxyl position. This eliminates the need for tedious protection and deprotection steps required in conventional methods, thereby simplifying the process and reducing impurities.
Q: What are the purity levels achievable with this new synthetic route?
A: Experimental data from the patent indicates that the final Molnupiravir product can achieve HPLC purity levels exceeding 99.7% after simple recrystallization. The high selectivity of the Lewis acid step minimizes side reactions, resulting in a cleaner crude profile that is easier to purify compared to enzymatic or multi-step chemical routes.
Q: Why is this method more cost-effective than enzymatic synthesis?
A: While enzymatic routes offer high yields, they rely on expensive biocatalysts like Novozyme 435 which significantly increase raw material costs. This chemical method utilizes inexpensive, commodity Lewis acid catalysts and standard chemical reagents, drastically lowering the comprehensive production cost while maintaining comparable efficiency and scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Molnupiravir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving antiviral medications. Our technical team has thoroughly analyzed the Lewis acid-catalyzed pathway disclosed in CN114560894B and possesses the expertise to execute this chemistry with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for API manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis can optimize your supply chain. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to validate the superior performance of our Molnupiravir intermediates. Let us collaborate to bring effective antiviral therapies to the market faster and more efficiently.
