Scalable One-Pot Synthesis of Molnupiravir Intermediates for Global API Production
Introduction to Advanced Molnupiravir Intermediate Synthesis
The global demand for effective antiviral therapeutics has placed immense pressure on the pharmaceutical supply chain to deliver high-quality active pharmaceutical ingredients (APIs) with speed and reliability. Central to this effort is the production of Molnupiravir, a broad-spectrum oral antiviral drug that has shown significant potential in treating SARS-CoV-2 infections. However, the manufacturing of its key precursors has historically been plagued by complex multi-step syntheses and unstable intermediates that hinder scalability. A groundbreaking development detailed in patent CN113880902A introduces a novel preparation method for a Molnupiravir drug intermediate that fundamentally reshapes the production landscape. This technology utilizes a sophisticated one-pot methodology to achieve acetonylidene protection and esterification simultaneously, effectively bypassing the notorious instability issues of previous routes.
By integrating these critical transformation steps into a single reactor vessel, the process not only simplifies the operational workflow but also significantly enhances the utilization rate of raw materials and solvents. For R&D directors and process chemists, this represents a pivotal shift from fragile, multi-stage batch processes to a robust, streamlined protocol. The ability to cross the unstable state of the sulfate intermediate without isolation is a major technical victory, directly translating to improved process safety and consistency. As we delve deeper into the technical specifics, it becomes clear that this innovation addresses the core bottlenecks that have limited the commercial viability of earlier synthetic strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
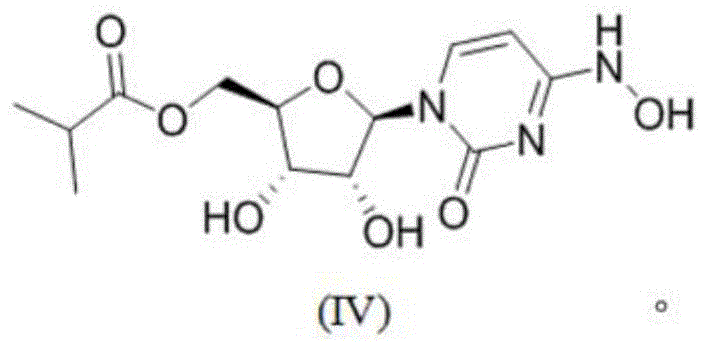
Prior to this innovation, the industry relied heavily on synthetic routes that were either economically prohibitive or technically fraught with risk. The original patent WO2019113462 described a pathway starting from uridine, which, while chemically feasible, suffered from severe economic drawbacks. This route required expensive starting materials and involved a cumbersome sequence of acetonide protection, esterification, substitution, hydroxylamine amination, and deprotection. The cumulative effect of these steps resulted in a dismal total yield of only 17 percent, accompanied by substantial generation of three wastes, making it unsuitable for cost-effective large-scale manufacturing.
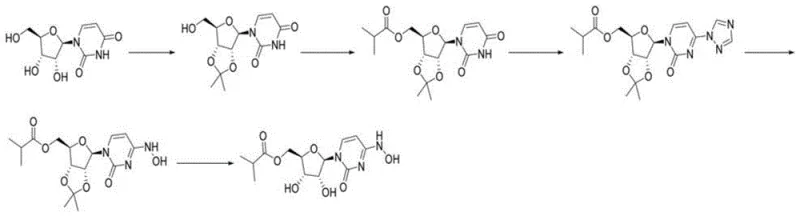
Subsequent attempts to improve this utilized cytidine as a cheaper raw material, which theoretically offered a shorter route with a higher total yield of around 44 percent. However, this second-generation approach introduced a critical failure point: the formation of a sulfate intermediate. Detailed stability studies revealed that this sulfate intermediate was extremely unstable, exhibiting self-deprotection at room temperature. Data indicated that in a wet state exposed to air, degradation reached 55 percent within just 2 hours, and even under a nitrogen atmosphere, degradation hit 70 percent after 15 hours. This inherent instability made the isolation and handling of the intermediate a logistical nightmare, posing significant risks for supply continuity and process control in a commercial setting.
The Novel Approach
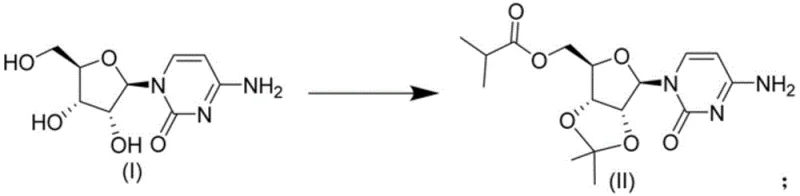
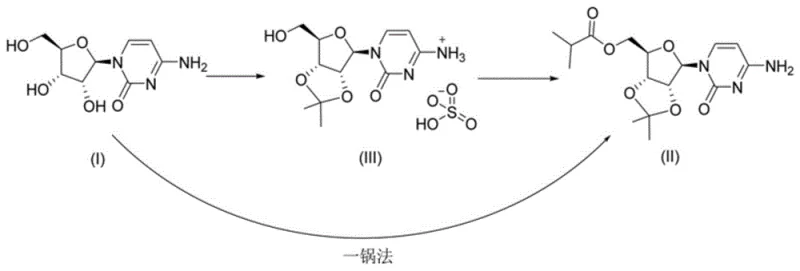
The methodology disclosed in CN113880902A offers a definitive solution to these challenges by employing a strategic one-pot synthesis. Instead of attempting to isolate the precarious sulfate species, the process treats cytidine, a reaction phase, and an organic solvent in a unified system. This allows for the realization of acetonylidene protection and esterification in a single operational step. By avoiding the isolation of the unstable intermediate, the process effectively “crosses” the unstable state, proceeding directly to the formation of the stable Molnupiravir intermediate (II). This approach not only mitigates the risk of degradation but also drastically reduces the number of unit operations required.
Furthermore, the post-reaction workup is remarkably simple, involving a water wash of the organic phase followed by concentration to yield a light yellow viscous substance. This simplicity stands in stark contrast to the complex purification sequences needed for the unstable sulfate salts of the past. For procurement and supply chain managers, this translates to a process that is not only faster but inherently more reliable, as it removes the variability associated with handling degradation-prone materials. The shift from a fragile multi-step isolation process to a continuous one-pot transformation marks a significant maturation in the manufacturing technology for this critical antiviral precursor.
Mechanistic Insights into One-Pot Acetonide Protection and Esterification
From a mechanistic perspective, the success of this one-pot strategy lies in the precise orchestration of reaction conditions that favor the desired transformations while suppressing side reactions. The process begins with the dissolution of cytidine in an organic solvent, followed by the addition of 2,2-dimethoxypropane. Under acidic catalysis, typically using sulfuric acid or methanesulfonic acid at temperatures between -10°C and 40°C, the diol functionality of the ribose ring is selectively protected as an acetonide. This step is crucial for defining the stereochemical integrity and solubility profile of the molecule for subsequent steps.
Without isolating the protected species, the reaction mixture is treated with a base, such as DBU or triethylamine, and a carboxylating agent like DMAP. The subsequent addition of isobutyric anhydride facilitates the esterification of the remaining hydroxyl group. The genius of this sequence is that the reaction environment is tuned to proceed directly to the esterified product (II) without allowing the hypothetical sulfate intermediate to accumulate to levels where decomposition becomes dominant. This kinetic control ensures that the unstable species, if formed transiently, is immediately consumed or bypassed, leading to the stable final intermediate.
Impurity control is inherently managed by this streamlined approach. In traditional multi-step syntheses, each isolation event introduces opportunities for impurity entrapment or thermal degradation. By maintaining the reaction in a homogeneous phase and minimizing exposure to potentially degrading conditions (such as the drying of unstable salts), the impurity profile is significantly cleaner. The patent data supports this, showing purities consistently above 95 percent in experimental examples. This high level of chemical fidelity is essential for downstream processing, as it reduces the burden on purification columns and crystallization steps later in the API synthesis, thereby preserving overall yield and quality.
How to Synthesize Molnupiravir Intermediate Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and temperature control to maximize the benefits of the one-pot design. The process is designed to be robust, accommodating a range of common organic solvents and catalysts, which provides flexibility for manufacturing sites with different inventory constraints. The following guide outlines the standardized approach derived from the patent examples, serving as a foundational protocol for process engineers looking to adopt this technology.
- Dissolve cytidine in an organic solvent such as dichloromethane, add 2,2-dimethoxypropane, and introduce an acid catalyst like sulfuric acid at controlled temperatures between 20°C and 25°C.
- After the protection phase, add a base such as DBU and a carboxylating agent like DMAP to the reaction mixture without isolating intermediates.
- Dropwise add isobutyric anhydride at 15°C to 20°C, maintain reaction for 1 to 3 hours, then wash the organic phase and concentrate to obtain the viscous intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and supply continuity, the implications of this technological shift are profound. The transition from a linear, multi-isolation process to a convergent one-pot synthesis addresses several critical pain points in pharmaceutical manufacturing. By collapsing multiple reaction steps into a single vessel, the process dramatically reduces the consumption of solvents and the energy required for heating, cooling, and drying between steps. This reduction in resource intensity directly correlates to a lower cost of goods sold (COGS), providing a competitive edge in the pricing of the final API.
- Cost Reduction in Manufacturing: The elimination of the unstable sulfate isolation step removes the need for specialized handling equipment and extensive safety protocols associated with degrading materials. Furthermore, the use of cytidine as a starting material is inherently more cost-effective than the uridine used in older routes. The high yields reported, reaching up to 100 percent in optimized examples, mean that raw material waste is minimized. This efficiency gain allows for substantial cost savings in [pharmaceutical intermediates] manufacturing, as fewer batches are required to meet production targets, and the throughput per reactor volume is significantly increased.
- Enhanced Supply Chain Reliability: Supply chain resilience is often compromised by processes that rely on unstable intermediates, which can lead to batch failures and unpredictable lead times. By stabilizing the synthesis through the one-pot method, the risk of batch rejection due to intermediate degradation is virtually eliminated. This reliability ensures a consistent flow of materials to downstream API manufacturers. The robustness of the process means that production schedules can be adhered to with greater confidence, reducing the lead time for high-purity [pharmaceutical intermediates] and preventing bottlenecks that could delay the availability of the final antiviral medication.
- Scalability and Environmental Compliance: The simplified workflow inherently generates less waste, aligning with modern green chemistry principles and stringent environmental regulations. Fewer workup steps mean less aqueous waste and solvent residue to treat, lowering the environmental footprint of the manufacturing site. Additionally, the process is highly scalable; the use of standard reagents and the absence of exotic or hazardous isolation steps facilitate the commercial scale-up of complex [pharmaceutical intermediates]. This scalability ensures that the supply can be rapidly expanded to meet surges in global demand without requiring disproportionate increases in infrastructure investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this one-pot synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption.
Q: Why is the one-pot method superior to the traditional sulfate route for Molnupiravir intermediates?
A: The traditional route involves an unstable sulfate intermediate that degrades rapidly (up to 55% in 2 hours), causing yield loss and safety risks. The one-pot method bypasses this isolation step entirely, ensuring higher stability and yield.
Q: What are the key cost drivers eliminated in this new synthetic pathway?
A: This pathway eliminates the need for expensive uridine starting materials and removes multiple isolation and purification steps associated with unstable intermediates, drastically reducing solvent usage and processing time.
Q: Can this process be scaled for commercial API manufacturing?
A: Yes, the process uses common solvents like dichloromethane and standard reagents, and the elimination of unstable intermediate handling makes it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Molnupiravir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires a partner with deep technical expertise and a commitment to quality. Our team has extensively analyzed the one-pot synthesis route described in CN113880902A and possesses the capability to implement this advanced methodology at scale. We understand the nuances of managing exothermic reactions and optimizing catalyst loading to ensure that the theoretical benefits of this process are fully realized in a commercial setting. Our facilities are equipped to handle diverse synthetic pathways, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency.
We invite you to collaborate with us to leverage this cutting-edge technology for your antiviral drug portfolio. Our rigorous QC labs and stringent purity specifications guarantee that every batch of Molnupiravir intermediate meets the highest international standards. To explore how we can optimize your supply chain, we encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us provide you with a Customized Cost-Saving Analysis that demonstrates the tangible economic benefits of switching to this superior synthetic route.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
