Advanced Palladium-Organic Nitrite Co-Catalysis for Commercial Scale-Up of Complex Indole Derivatives
Advanced Palladium-Organic Nitrite Co-Catalysis for Commercial Scale-Up of Complex Indole Derivatives
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for synthesizing privileged scaffolds like indoles. A significant breakthrough in this domain is detailed in Chinese Patent CN107118145B, which discloses a novel method for the cyclization and synthesis of indoles under the co-catalysis of palladium and organic nitrite. This technology represents a paradigm shift from traditional oxidative cyclization methods that rely on harsh conditions or toxic stoichiometric oxidants. By utilizing a synergistic catalytic system where a palladium compound and an organic nitrite operate in concert under an oxygen atmosphere, this process achieves high yields under remarkably mild conditions. For R&D directors and process chemists, this patent offers a robust solution for accessing diverse indole libraries, while supply chain managers will appreciate the elimination of expensive and hazardous co-oxidants like benzoquinone. As a reliable pharmaceutical intermediate supplier, understanding and implementing such green chemistry innovations is crucial for maintaining competitiveness in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indoles has relied heavily on the classical Emil Fischer indole synthesis, a method that, while foundational, suffers from severe limitations in modern process chemistry. The Fischer synthesis typically requires strong acidic conditions and extreme heating, often reaching temperatures between 180°C and 250°C, which poses significant safety risks and energy costs. Furthermore, the substrate scope is often limited, and the harsh conditions can lead to the decomposition of sensitive functional groups, resulting in complex impurity profiles that are difficult to purge. In more recent developments, transition metal-catalyzed C-H bond oxidation of 2-aminostyrenes has emerged, but these methods frequently necessitate the addition of equivalent amounts of benzoquinone or divalent copper salts as co-oxidants. The reliance on stoichiometric benzoquinone not only increases the raw material cost significantly but also generates substantial quinone-related waste, complicating downstream processing and wastewater treatment. Additionally, many existing catalytic systems are restricted to specific substrate types, such as aryl-substituted styrylanilines, failing to provide a general solution for unsubstituted or differently substituted indoles.
The Novel Approach
The methodology described in patent CN107118145B overcomes these historical bottlenecks by introducing an elegant palladium-organic nitrite co-catalytic system. Instead of relying on toxic heavy metal co-oxidants or harsh thermal conditions, this novel approach utilizes molecular oxygen as the terminal oxidant, driven by the synergistic action of a palladium catalyst and an organic nitrite additive. The reaction proceeds efficiently at a mild temperature of 70°C in common solvents like tert-butanol, drastically reducing energy consumption and thermal stress on the molecules. A key innovation is the ability to synthesize indoles with or without substituents at the C2-position with excellent yields, a feat that many prior art methods struggle to achieve simultaneously. Moreover, the process allows for the strategic introduction of chlorine substituents on the aromatic ring of the precursor, enabling the direct synthesis of chloro-indoles which serve as versatile handles for further functionalization via cross-coupling reactions. This eliminates the need for cumbersome directing groups on the indole nitrogen, streamlining the synthetic route and enhancing overall atom economy.
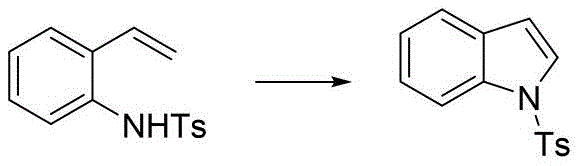
The versatility of this new catalytic system is exemplified by its compatibility with various nitrogen-protecting groups. As illustrated in the reaction schemes, substrates protected with tosyl, methyl carbamate, tert-butyl carbamate (Boc), and benzyl carbamate (Cbz) groups all undergo smooth cyclization to form the corresponding indole derivatives. This broad substrate tolerance is critical for process flexibility, allowing manufacturers to select the most cost-effective or easily removable protecting group for their specific API synthesis pathway. The ability to handle diverse electronic environments on the aniline ring without compromising yield demonstrates the robustness of the palladium-organic nitrite mechanism, making it a superior choice for the commercial scale-up of complex heterocycles compared to rigid, substrate-specific legacy methods.
Mechanistic Insights into Pd-Organic Nitrite Co-Catalyzed Cyclization
At the heart of this technological advancement lies a sophisticated catalytic cycle that leverages the redox properties of palladium and organic nitrites. The reaction initiates with the coordination of the palladium(II) species to the olefinic double bond of the 2-vinylaniline substrate, activating it for nucleophilic attack by the adjacent nitrogen atom. This intramolecular aminopalladation forms a key organopalladium intermediate. In traditional Wacker-type oxidations, the regeneration of the active Pd(II) catalyst from the reduced Pd(0) species often requires stoichiometric amounts of copper salts or benzoquinone. However, in this novel system, the organic nitrite plays a pivotal role as a redox mediator. It facilitates the re-oxidation of Pd(0) back to Pd(II) using molecular oxygen from the air or an oxygen balloon as the ultimate electron acceptor. This cooperative catalysis ensures that the palladium remains in its active oxidation state throughout the reaction cycle without the accumulation of reduced metal waste. The use of organic nitrites, such as tert-butyl nitrite or isoamyl nitrite, is particularly advantageous as they are soluble in organic media and decompose into volatile byproducts, simplifying the workup procedure.
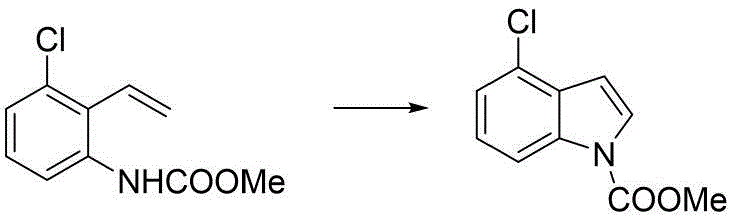
Furthermore, the mechanistic pathway offers distinct advantages regarding regioselectivity and impurity control. By pre-installing substituents like chlorine on the vinylaniline precursor, the electronic properties of the aromatic ring are tuned to favor specific cyclization outcomes. For instance, the synthesis of 4-chloroindole derivatives proceeds with high fidelity, avoiding the formation of regioisomers that often plague electrophilic substitution methods on pre-formed indoles. This precision is vital for R&D teams aiming to minimize impurity loads in early-stage drug development. The mild reaction conditions also suppress side reactions such as polymerization of the styrene moiety or over-oxidation of the indole core, which are common issues in high-temperature processes. The result is a cleaner reaction profile that translates directly to higher purity specifications and reduced burden on purification units, aligning perfectly with the stringent quality requirements of high-purity pharmaceutical intermediates.
How to Synthesize Indole Derivatives Efficiently
Implementing this palladium-catalyzed cyclization in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and safety. The general protocol involves charging a reaction vessel with the palladium catalyst, typically Pd(PhCN)2Cl2, and the organic nitrite co-catalyst in a molar ratio optimized for turnover. The substrate, a substituted 2-vinylaniline, is then introduced along with a solvent such as tert-butanol or ethanol. It is imperative to establish an oxygen-rich atmosphere, either by purging with oxygen gas or operating under an oxygen balloon, to drive the catalytic cycle. The mixture is heated to 70°C and stirred for a period ranging from 7 to 24 hours, depending on the electronic nature of the substrate. Following the reaction, the crude product is isolated and purified via flash column chromatography using a petroleum ether and diethyl ether gradient. For detailed operational specifics and safety guidelines, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining a palladium compound (7.5 mol%) and an organic nitrite (20 mol%) with the 2-vinylaniline substrate in a suitable solvent such as tert-butanol.
- Maintain the reaction under an oxygen atmosphere at a controlled temperature of 70°C for a duration ranging from 7 to 24 hours to ensure complete cyclization.
- Upon completion, purify the crude mixture using flash column chromatography with a petroleum ether and diethyl ether gradient to isolate the high-purity indole target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible economic and logistical benefits that extend beyond simple yield improvements. The most significant advantage lies in the drastic simplification of the oxidation system. By eliminating the need for stoichiometric benzoquinone or copper salts, the process removes two major cost drivers: the purchase of expensive oxidants and the disposal of hazardous heavy metal waste. This reduction in auxiliary chemicals leads to substantial cost savings in raw material procurement and waste management fees. Furthermore, the use of molecular oxygen as the oxidant is inherently economical, as air is freely available and requires minimal infrastructure compared to handling solid oxidants. The mild reaction temperature of 70°C also contributes to energy efficiency, reducing the steam or electricity load required for heating reactors, which accumulates to significant operational expenditure reductions over large production batches.
- Cost Reduction in Manufacturing: The elimination of stoichiometric co-oxidants like benzoquinone and copper salts removes a significant portion of raw material costs associated with traditional indole synthesis. Since these reagents are consumed in equivalent amounts to the substrate, their removal drastically lowers the bill of materials. Additionally, the simplified workup procedure reduces solvent consumption and labor hours required for purification, further driving down the cost of goods sold (COGS) for high-volume production.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as oxygen gas and common organic nitrites enhances supply chain resilience. Unlike specialized oxidants that may face availability fluctuations or long lead times, the reagents for this process are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The green chemistry profile of this method, characterized by the use of oxygen and the absence of heavy metal waste, aligns perfectly with increasingly stringent environmental regulations. Scaling this process from kilograms to tons does not introduce new environmental hazards, facilitating smoother regulatory approvals for manufacturing sites. The reduced E-factor (mass of waste per mass of product) makes the facility more sustainable and lowers the carbon footprint of the final pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-organic nitrite co-catalyzed indole synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: What are the primary advantages of using organic nitrite over traditional co-oxidants in indole synthesis?
A: Traditional methods often require stoichiometric amounts of benzoquinone or copper salts, which generate significant toxic waste and complicate purification. The use of organic nitrite as a co-catalyst allows for the use of molecular oxygen as the terminal oxidant, drastically reducing chemical waste and improving the environmental profile of the process.
Q: How does this method improve regioselectivity for functionalized indoles?
A: By pre-introducing substituents such as chlorine on the aromatic ring of the 2-vinylaniline precursor, this method enables the efficient synthesis of specific chloro-indoles. This avoids the need for difficult post-synthetic functionalization or the use of directing groups on the nitrogen atom, simplifying the overall synthetic route.
Q: Is this catalytic system suitable for large-scale manufacturing?
A: Yes, the reaction operates at mild temperatures (70°C) and uses oxygen gas, which is economically favorable for scale-up. The absence of expensive stoichiometric oxidants and the use of standard solvents like tert-butanol make the process highly adaptable for commercial production from kilogram to ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the palladium-organic nitrite co-catalysis method for producing high-value indole intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in the lab are faithfully translated to the manufacturing floor. Our state-of-the-art facilities are equipped to handle oxygen-sensitive reactions safely and efficiently, while our rigorous QC labs enforce stringent purity specifications to meet the exacting standards of the global pharmaceutical industry. We are committed to leveraging such advanced green chemistry technologies to deliver superior products that enhance your supply chain stability.
We invite you to collaborate with us to evaluate the feasibility of this route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis comparing this novel method against your current supply chain arrangements. Please contact our technical procurement team today to request specific COA data for our indole portfolio and discuss route feasibility assessments tailored to your API development timeline.
