Advanced Aqueous Synthesis of 1,2,3,4-Tetrahydrocarbazole for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, environmentally benign pathways for synthesizing critical heterocyclic intermediates. Patent CN102249983A introduces a groundbreaking methodology for the preparation of 1,2,3,4-tetrahydrocarbazole compounds, addressing long-standing inefficiencies in traditional synthetic routes. This innovation utilizes phenylhydrazine hydrochloride or phenylhydrazine alongside cyclohexanone as primary raw materials, executing a one-step condensation and ring-closing reaction in an aqueous medium. Unlike legacy processes that rely heavily on volatile organic compounds, this technique employs water as the solvent and inorganic acids as catalysts, operating under moderate thermal conditions ranging from 40-90°C. The result is a robust process capable of delivering high-content 1,2,3,4-tetrahydrocarbazole with exceptional purity profiles. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for carbazole derivatives, which are pivotal in the manufacture of high-grade pigments, dyes, and active pharmaceutical ingredients.
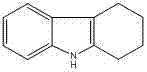
The structural integrity of the target molecule, as depicted in the patent documentation, underscores its value as a versatile building block in organic synthesis. The ability to produce this scaffold with yields exceeding 98% and HPLC purity greater than 99% positions this method as a superior alternative for reliable tetrahydrocarbazole supplier networks. By shifting the paradigm from organic solvents to aqueous systems, the technology not only enhances safety protocols but also aligns with global sustainability mandates, making it an attractive option for cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbazole and its tetrahydro-derivatives has been dominated by the Borsche method, which typically involves the condensation of phenylhydrazine with ketones under acidic conditions using glacial acetic acid as both solvent and catalyst. While this approach has served the industry for decades, it suffers from significant drawbacks that hinder modern large-scale production efficiency. The reliance on acetic acid creates a corrosive operational environment that demands specialized equipment resistant to acid degradation, thereby inflating capital expenditure. Moreover, the separation and purification processes associated with acetic acid are energy-intensive, requiring complex distillation and recovery units to manage solvent waste. Traditional literature methods often report yields fluctuating between 88% and 95%, with product purity compromised by side reactions and difficult-to-remove impurities. The environmental footprint of these conventional routes is substantial, generating acidic wastewater and volatile organic emissions that require costly treatment before discharge. For supply chain heads, these factors translate into longer lead times and higher variability in batch quality, posing risks to continuous manufacturing schedules.
The Novel Approach
In stark contrast, the methodology outlined in CN102249983A revolutionizes the production landscape by substituting acetic acid with water and utilizing mineral acids such as hydrochloric, sulfuric, or phosphoric acid as catalysts. This strategic shift eliminates the need for expensive solvent recovery systems and drastically simplifies the post-reaction workup. The process involves mixing phenylhydrazine hydrochloride with water and the chosen acid, followed by the controlled addition of cyclohexanone at temperatures between 40-90°C. The reaction proceeds smoothly to completion within a few hours, after which the product precipitates upon cooling and can be isolated via simple filtration. This aqueous-based protocol not only reduces raw material costs but also mitigates the severe operational hazards associated with handling large volumes of hot acetic acid. The resulting product demonstrates superior quality metrics, with purity levels consistently surpassing 99% as verified by HPLC analysis. For stakeholders focused on commercial scale-up of complex pharmaceutical intermediates, this approach offers a streamlined, eco-friendly pathway that enhances overall process economics without compromising on chemical performance.
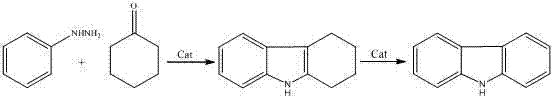
Mechanistic Insights into Acid-Catalyzed Aqueous Cyclization
The core of this technological advancement lies in the optimization of the Fischer indole synthesis mechanism within an aqueous environment. The reaction initiates with the protonation of the carbonyl oxygen of cyclohexanone by the mineral acid catalyst, increasing its electrophilicity and facilitating nucleophilic attack by the amino group of phenylhydrazine. This leads to the formation of a hydrazone intermediate, which subsequently undergoes a [3,3]-sigmatropic rearrangement, a critical step driven by the specific acid strength and thermal energy provided in the 40-90°C range. In traditional organic solvents, this rearrangement can be sluggish or prone to competing side reactions; however, the unique solvation properties of water in this system appear to stabilize the transition states effectively. The presence of water molecules likely assists in proton transfer steps, accelerating the cyclization and subsequent aromatization (or partial saturation in the case of tetrahydrocarbazole) to form the final fused ring system. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate or further optimize the process, as it highlights the delicate balance between acid concentration and temperature required to maximize conversion rates.
Impurity control is another critical aspect where this aqueous method excels, directly impacting the downstream processing requirements for high-purity API intermediates. In conventional acetic acid methods, acetylation side products or polymeric tars often form, necessitating rigorous recrystallization or chromatographic purification. The patented process minimizes these impurities through precise stoichiometric control, maintaining a molar ratio of phenylhydrazine to cyclohexanone between 1:1.05 and 1:1.5. Furthermore, the use of phenylhydrazine hydrochloride as a starting material, rather than free base phenylhydrazine, enhances stability and reduces the formation of oxidative byproducts. The final washing step to neutrality effectively removes residual acid catalyst and unreacted starting materials, yielding a crude product that is remarkably clean. This high level of intrinsic purity reduces the burden on quality control labs and ensures that the material meets stringent specifications for use in sensitive medicinal chemistry applications, thereby securing the supply chain against quality-related disruptions.
How to Synthesize 1,2,3,4-Tetrahydrocarbazole Efficiently
Implementing this synthesis route requires careful attention to the addition rate of cyclohexanone and the maintenance of the reaction temperature profile to ensure optimal kinetics. The patent details a procedure where the ketone is dripped into the acidic aqueous hydrazine solution over a period of 0.5 to 4 hours, preventing localized exotherms that could degrade the product. Following the addition, a保温 (holding) period of 0.5 to 3.0 hours allows the reaction to reach full conversion. The detailed standardized synthesis steps, including specific reagent grades and agitation speeds, are provided in the guide below to assist technical teams in replicating this high-yield process.
- Mix phenylhydrazine hydrochloride or phenylhydrazine with water and a mineral acid catalyst in a reactor.
- Slowly drip cyclohexanone into the mixture at a controlled temperature range of 40-90°C over 0.5-4 hours.
- Maintain the reaction temperature for an additional 0.5-3.0 hours, then cool, filter, wash to neutrality, and dry to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous synthesis technology translates into tangible strategic benefits that extend beyond mere chemical yield. The substitution of acetic acid with water represents a fundamental shift in cost structure, removing the volatility associated with organic solvent pricing and the logistical complexities of hazardous waste disposal. This change alone drives significant cost reduction in manufacturing operations, allowing for more competitive pricing models in the global market. Additionally, the simplicity of the workup—filtration and washing—reduces the demand for specialized labor and extensive processing time, further enhancing operational efficiency. These factors collectively contribute to a more resilient supply chain capable of responding rapidly to market demands for carbazole derivatives.
- Cost Reduction in Manufacturing: The elimination of glacial acetic acid as a solvent removes a major cost center from the production budget, as water is virtually free and requires no recovery infrastructure. Furthermore, the use of phenylhydrazine hydrochloride, which is often more stable and cheaper than the free base, contributes to lower raw material expenses. The high yield (>98%) ensures that raw material utilization is maximized, minimizing waste generation and the associated costs of disposal. By avoiding the need for complex distillation columns to recover acetic acid, capital investment in plant equipment is also significantly reduced, leading to a faster return on investment for facilities adopting this technology.
- Enhanced Supply Chain Reliability: Sourcing water and mineral acids is far less susceptible to geopolitical or logistical disruptions compared to specialty organic solvents. The robustness of the reaction conditions (40-90°C) means that the process can be run in standard glass-lined or stainless steel reactors without requiring exotic materials of construction, increasing the number of potential contract manufacturing partners. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates, as production slots can be secured more easily across a broader network of suppliers. The consistent quality of the output also reduces the risk of batch rejections, ensuring a steady flow of material to downstream customers.
- Scalability and Environmental Compliance: The aqueous nature of the reaction inherently aligns with green chemistry principles, making regulatory approval for scale-up smoother and faster. Wastewater treatment is simplified since the effluent primarily contains dilute mineral acids and salts, which are easier to neutralize than organic solvent mixtures. This environmental friendliness reduces the carbon footprint of the manufacturing process, a key metric for modern ESG-compliant supply chains. The process is readily scalable from kilogram to multi-ton quantities without significant re-engineering, supporting the commercial scale-up of complex pharmaceutical intermediates required for clinical and commercial drug production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. They are derived from the specific advantages and procedural details outlined in the patent documentation to provide clarity for potential partners.
Q: What are the primary advantages of using water as a solvent in tetrahydrocarbazole synthesis?
A: Using water instead of traditional organic solvents like acetic acid significantly reduces raw material costs and eliminates the need for complex solvent recovery systems. Furthermore, it drastically minimizes environmental pollution and simplifies the post-reaction workup, as the product can often be isolated via simple filtration after cooling.
Q: How does this novel method improve product purity compared to conventional Borsche methods?
A: The patented aqueous method achieves HPLC purity levels exceeding 99% by optimizing the acid catalyst concentration and temperature profile. This precise control suppresses side reactions and polymerization often seen in harsher organic solvent systems, resulting in a cleaner crude product that requires less intensive purification.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. The use of inexpensive mineral acids and water, combined with moderate reaction temperatures (40-90°C) and simple isolation steps (filtration and washing), makes it highly economical and safe for manufacturing at the 100 MT scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3,4-Tetrahydrocarbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous synthesis route described in CN102249983A for the production of high-value heterocyclic intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped to handle the specific thermal and corrosion requirements of acid-catalyzed reactions, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to leveraging this green chemistry innovation to deliver superior value to our global partners.
We invite pharmaceutical and agrochemical companies to collaborate with us to optimize their supply chains using this advanced technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this efficient synthesis method can enhance your project's economic viability and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
