Optimizing Oxitinib Mesylate Production: A Technical Breakthrough in High-Purity API Intermediate Manufacturing
Introduction to Advanced Oxitinib Mesylate Synthesis
The global demand for third-generation EGFR-TKI inhibitors, specifically Oxitinib Mesylate (AZD9291), has surged due to its critical efficacy in treating T790M mutation-positive non-small cell lung cancer. However, the commercial viability of this life-saving medication hinges on the robustness and purity of its synthetic pathway. Patent CN113651800B discloses a revolutionary preparation method that fundamentally restructures the manufacturing landscape for this complex API intermediate. By shifting away from traditional nitration and protection-deprotection sequences, this novel approach leverages anhydrous acid catalysis and green hydrogenation techniques to achieve unprecedented purity levels. For R&D directors and procurement strategists, this patent represents a pivotal shift towards a more cost-effective and environmentally sustainable production model, addressing long-standing issues regarding genotoxic impurities and low yields that have plagued earlier synthetic routes.
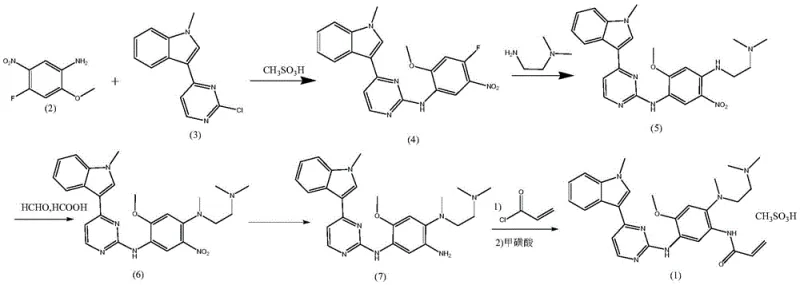
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
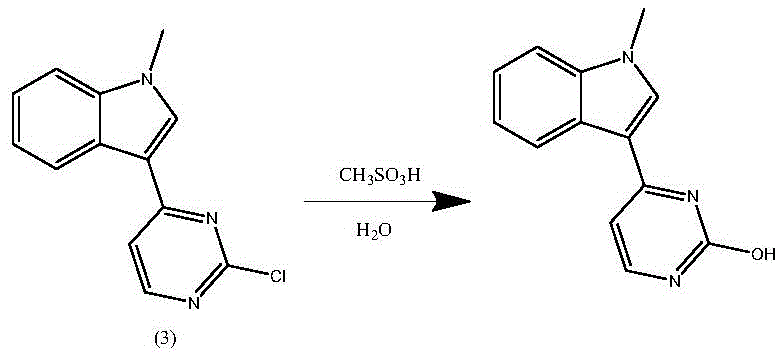
Historically, the synthesis of Oxitinib intermediates has been fraught with chemical inefficiencies and safety hazards. Conventional routes, such as those described in earlier patents like CN103702990B, often initiate with the nitration of 4-fluoro-2-methoxyaniline. This step is notoriously problematic because the amino group is susceptible to oxidation by sulfuric acid, leading to significantly depressed yields. Furthermore, the subsequent neutralization processes generate massive quantities of inorganic salts, creating a heavy burden on waste treatment facilities and complicating the isolation of the desired 4-fluoro-2-methoxy-5-nitroaniline. Another critical bottleneck in legacy methods is the reliance on p-toluenesulfonic acid (p-TsOH) as a catalyst. Since p-TsOH typically contains water, it promotes the hydrolysis of the sensitive chloro-pyrimidine ring, generating stubborn 2-hydroxyl impurities that are exceptionally difficult to remove. Additionally, the use of alcohol solvents in the presence of sulfonic acids can lead to the formation of alkyl methanesulfonates, which are potential genotoxic impurities posing severe regulatory risks for the final pharmaceutical product.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113651800B introduces a streamlined, four-step sequence that bypasses these historical pitfalls. The process begins with a direct condensation reaction between 4-fluoro-2-methoxy-5-nitroaniline and 3-(2-chloro-4-pyrimidinyl)-1-methyl-1H-indole, utilizing anhydrous methanesulfonic acid in a non-aqueous solvent like dioxane. This strategic choice of an anhydrous catalyst effectively suppresses the hydrolysis side reactions, driving the yield of the key intermediate up to over 92% with purity exceeding 99.5%. Furthermore, the route innovatively employs an Eschweiler-Clarke amine reduction alkylation to construct the complex side chain, rather than relying on expensive and impure pre-alkylated diamines. This not only simplifies the purification process by eliminating the need for column chromatography but also drastically reduces raw material costs. The final stages utilize water as a solvent for catalytic hydrogenation and isopropyl acetate for salting out, ensuring a green, scalable process that minimizes the risk of genotoxic ester formation.
Mechanistic Insights into Anhydrous Acid Catalysis and Impurity Control
The cornerstone of this improved synthesis lies in the rigorous control of water content during the critical condensation phase. In traditional acidic environments, the chlorine atom at the 2-position of the pyrimidine ring is highly susceptible to nucleophilic attack by water molecules. As illustrated in the reaction mechanism below, this hydrolysis leads to the formation of 3-(2-hydroxy-4-pyrimidinyl)-1-methyl-1H-indole, a structural analog that is chemically similar to the desired product and extremely difficult to separate via standard crystallization. By employing anhydrous liquid acids such as methanesulfonic acid or trifluoroacetic acid in dry organic solvents like 1,4-dioxane, the new method creates a moisture-free environment that kinetically favors the desired nucleophilic aromatic substitution of the fluorine atom on the aniline ring while preserving the chloro-pyrimidine integrity. This mechanistic precision is what allows the process to achieve such high selectivity and yield.
Beyond the initial condensation, the control of impurities extends to the alkylation and salting-out steps. The adoption of the Eschweiler-Clarke reaction allows for the in-situ generation of the tertiary amine functionality using formaldehyde and formic acid. This avoids the introduction of extraneous impurities often found in commercial grades of N,N,N'-trimethylethylenediamine, such as tetramethylethylenediamine. Moreover, the final salification step utilizes isopropyl acetate as the solvent. Unlike ethanol or other lower alcohols, isopropyl acetate is resistant to esterification with methanesulfonic acid under the reaction conditions. This chemical stability is crucial because it prevents the formation of isopropyl methanesulfonate, a known genotoxic impurity. By understanding and manipulating these specific reaction pathways, manufacturers can ensure a cleaner impurity profile that meets the stringent requirements of global regulatory bodies like the FDA and EMA.
How to Synthesize Oxitinib Mesylate Efficiently
The implementation of this novel synthetic route requires precise adherence to reaction conditions to maximize the benefits of anhydrous catalysis and green chemistry principles. The process is designed to be operationally simple, avoiding complex protection groups and hazardous reagents where possible. Below is a high-level overview of the operational workflow derived from the patent examples, demonstrating how the transition from nitro-reduction to amidation is managed efficiently.
- Condense 4-fluoro-2-methoxy-5-nitroaniline with 3-(2-chloro-4-pyrimidinyl)-1-methyl-1H-indole using anhydrous methanesulfonic acid in dioxane.
- Perform nucleophilic substitution with N,N-dimethylethylenediamine followed by Eschweiler-Clarke reduction alkylation using formic acid and formaldehyde.
- Execute catalytic hydrogenation in an aqueous acidic medium to reduce the nitro group to an amino group.
- React the resulting amine with acryloyl chloride and salt with methanesulfonic acid in isopropyl acetate to finalize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical improvements in patent CN113651800B translate directly into tangible commercial benefits. The elimination of column chromatography is perhaps the most significant cost driver; chromatography is notoriously difficult to scale, consumes vast amounts of silica and solvents, and creates bottlenecks in batch processing. By replacing this with crystallization and precipitation steps, the new method offers a pathway to substantial cost savings in Oxitinib Mesylate manufacturing. Furthermore, the shift to water-based hydrogenation and the use of recoverable solvents like isopropyl acetate aligns with modern environmental, social, and governance (ESG) goals, reducing the burden of hazardous waste disposal and lowering the overall carbon footprint of the production facility.
- Cost Reduction in Manufacturing: The replacement of expensive, high-purity N,N,N'-trimethylethylenediamine with cheaper N,N-dimethylethylenediamine and formaldehyde significantly lowers the bill of materials. Additionally, the avoidance of p-toluenesulfonic acid removes the need for extensive washing and neutralization steps required to remove residual sulfonates, thereby reducing utility consumption and labor hours associated with post-reaction workups.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as methanesulfonic acid, dioxane, and acryloyl chloride ensures a stable supply chain less vulnerable to the fluctuations of specialized reagent markets. The robustness of the anhydrous condensation step means that batch-to-batch variability is minimized, leading to more predictable production schedules and reliable delivery timelines for downstream API manufacturers.
- Scalability and Environmental Compliance: The use of water as a solvent for the hydrogenation step is a major advantage for scale-up. Water is non-flammable, inexpensive, and easy to handle in large reactors compared to volatile organic solvents. This green chemistry approach not only simplifies safety protocols in the plant but also facilitates easier regulatory approval by demonstrating a commitment to reducing volatile organic compound (VOC) emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the comparative data and beneficial effects reported in the patent documentation, providing clarity on why this method represents a superior choice for industrial production.
Q: How does the new method prevent genotoxic impurities compared to conventional routes?
A: By replacing p-toluenesulfonic acid with anhydrous methanesulfonic acid and utilizing isopropyl acetate for salting out, the process eliminates the formation of ethyl or isopropyl methanesulfonate esters, which are potential genotoxic impurities found in alcohol-based systems.
Q: Why is the Eschweiler-Clarke reaction preferred over direct alkylation?
A: Direct alkylation often requires expensive and impure N,N,N'-trimethylethylenediamine. The Eschweiler-Clarke approach uses cheaper dimethylethylenediamine and formaldehyde, avoiding difficult column chromatography purification steps and significantly lowering raw material costs.
Q: What role does the anhydrous catalyst play in yield improvement?
A: Using anhydrous liquid acids like methanesulfonic acid prevents the hydrolysis of the chloro-pyrimidine moiety into hydroxyl impurities, boosting the yield of the key intermediate from approximately 55% to over 92% while achieving purity levels exceeding 99.5%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxitinib Mesylate Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team has extensively analyzed the pathway described in CN113651800B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art reactors capable of handling anhydrous conditions and high-pressure hydrogenation safely, ensuring that the stringent purity specifications required for oncology intermediates are consistently met through our rigorous QC labs.
We invite global pharmaceutical partners to collaborate with us to leverage this optimized synthesis route for your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest standards of quality and efficiency.
