Scaling High-Purity Oxitinib Mesylate: Technical Upgrades and Commercial Manufacturing Capabilities
Introduction to Patent CN113651800A and Process Innovation
The pharmaceutical landscape for non-small cell lung cancer (NSCLC) treatment has been significantly advanced by third-generation EGFR-TKIs, specifically Oxitinib Mesylate. Patent CN113651800A, published in late 2021, discloses a robust and environmentally friendly preparation method that addresses critical bottlenecks in existing manufacturing protocols. This technical disclosure represents a paradigm shift from traditional multi-step syntheses plagued by low yields and toxic impurities to a streamlined, high-efficiency route. By leveraging anhydrous liquid acid catalysis and green hydrogenation techniques, the invention achieves a total yield of approximately 70 percent with exceptional purity profiles. For global procurement and R&D teams, understanding this methodology is essential for securing a reliable Oxitinib Mesylate supplier capable of meeting stringent regulatory standards while optimizing production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Oxitinib intermediates has relied on routes that introduce significant chemical and operational risks. As illustrated in prior art such as patent CN103702990B, traditional methods often involve the nitration of 4-fluoro-2-methoxyaniline using mixed acids. This step is notoriously problematic because the amino group is susceptible to oxidation by sulfuric acid, leading to depressed yields and complex post-treatment requirements. Furthermore, the formation of salts between the intermediate and sulfuric acid necessitates massive amounts of alkali for neutralization, generating inorganic waste far exceeding the weight of the product. Additionally, the widespread use of p-toluenesulfonic acid (p-TsOH) as a catalyst in alcoholic solvents creates a hazardous environment where genotoxic impurities, such as ethyl p-toluenesulfonate, are easily formed. These legacy processes not only burden the supply chain with difficult waste treatment but also compromise the safety profile of the final active pharmaceutical ingredient.
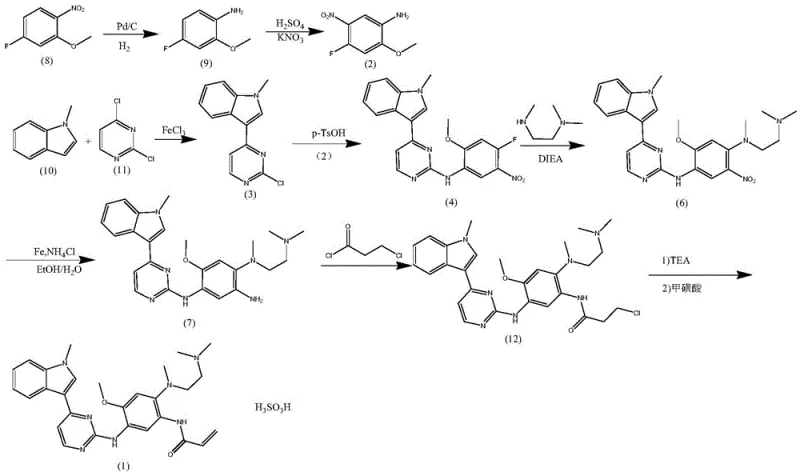
The Novel Approach
In stark contrast, the methodology outlined in CN113651800A introduces a sophisticated condensation strategy that bypasses these historical pitfalls. The novel route initiates with a direct condensation between 4-fluoro-2-methoxy-5-nitroaniline and 3-(2-chloro-4-pyrimidinyl)-1-methyl-1H-indole, catalyzed by anhydrous methanesulfonic acid rather than hydrated p-TsOH. This strategic substitution effectively suppresses the hydrolysis of the chloro-pyrimidine moiety, a common side reaction that generates stubborn 2-hydroxyl impurities. Following this, the process employs a nucleophilic substitution with N,N-dimethylethylenediamine, followed by an in-situ Eschweiler-Clarke amine reduction alkylation. This sequence eliminates the need for expensive and impure N,N,N'-trimethylethylenediamine, thereby simplifying the purification landscape and removing the necessity for industrial-scale column chromatography. The result is a cleaner, more direct pathway to the key intermediate, setting the stage for high-purity final product formation.
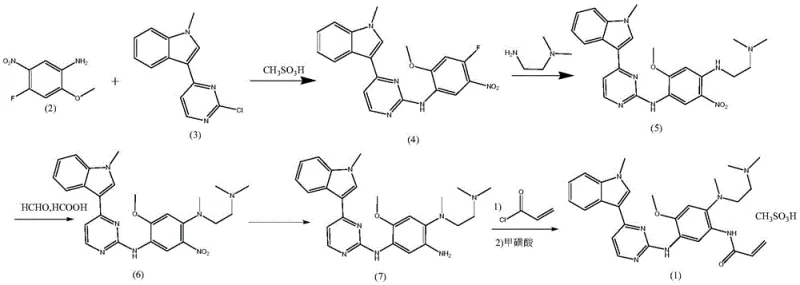
Mechanistic Insights into Anhydrous Acid Catalysis and Impurity Control
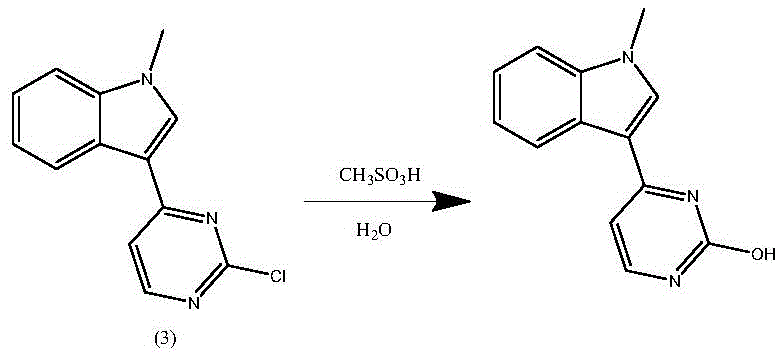
The core technical breakthrough of this patent lies in the meticulous control of reaction conditions to prevent hydrolytic degradation. In conventional acidic environments containing water, the chlorine atom at the 2-position of the pyrimidine ring is highly susceptible to nucleophilic attack by water molecules. This side reaction leads to the formation of 3-(2-hydroxy-4-pyrimidinyl)-1-methyl-1H-indole, an impurity that is chemically similar to the desired product and extremely difficult to separate. The patent explicitly demonstrates that by utilizing anhydrous liquid acids, such as methanesulfonic acid, in non-aqueous solvents like dioxane, the availability of free water is minimized. This effectively shuts down the hydrolysis pathway, ensuring that the condensation reaction proceeds with high selectivity towards the desired N-aryl pyrimidine-2-amine structure. The mechanistic precision here translates directly to a yield improvement from roughly 55 percent in older methods to over 92 percent in this new protocol.
Furthermore, the impurity control extends to the final salt formation step, addressing critical genotoxicity concerns. Traditional methods using ethanol or isobutanol for salt formation with methanesulfonic acid carry the risk of generating alkyl methanesulfonates, which are potent genotoxic impurities. The disclosed method creatively utilizes isopropyl acetate as the salifying solvent. Due to the steric hindrance and lower reactivity of isopropyl acetate compared to primary alcohols, the formation of isopropyl methanesulfonate is significantly inhibited. Coupled with a green catalytic hydrogenation step using water and acetic acid, the entire process minimizes the introduction of extraneous organic contaminants. This rigorous attention to mechanistic detail ensures that the final Oxitinib Mesylate meets the rigorous purity specifications required for oncology therapeutics, reducing the burden on downstream quality control laboratories.
How to Synthesize Oxitinib Mesylate Efficiently
The synthesis of Oxitinib Mesylate via this patented route involves four distinct chemical transformations that balance reactivity with selectivity. The process begins with the acid-catalyzed coupling of the aniline and indole-pyrimidine fragments, followed by the installation of the diamine side chain through substitution and methylation. The subsequent reduction of the nitro group and final acrylamide formation complete the molecular architecture. While the chemical principles are straightforward, the specific operational parameters—such as temperature controls between 70-90°C for condensation and pH adjustments during hydrogenation—are critical for success. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to guide process development teams in replicating this high-efficiency route.
- Condense 4-fluoro-2-methoxy-5-nitroaniline with 3-(2-chloro-4-pyrimidinyl)-1-methyl-1H-indole using anhydrous methanesulfonic acid in dioxane.
- Perform nucleophilic substitution with N,N-dimethylethylenediamine followed by Eschweiler-Clarke alkylation using formaldehyde and formic acid.
- Reduce the nitro group via catalytic hydrogenation in water/acetic acid, then react with acryloyl chloride and salt with methanesulfonic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN113651800A offers tangible strategic benefits beyond mere technical elegance. The elimination of column chromatography, a unit operation that is notoriously difficult to scale and expensive to maintain in a GMP environment, represents a major reduction in processing time and solvent consumption. By shifting to crystallization-based purifications and aqueous workups, the process becomes inherently more scalable, allowing for the commercial scale-up of complex pharmaceutical intermediates without proportional increases in capital expenditure. This operational simplicity directly correlates to enhanced supply chain reliability, as the process is less susceptible to the bottlenecks associated with specialized purification equipment and hazardous waste disposal.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the substitution of high-cost reagents with commodity chemicals. Replacing expensive N,N,N'-trimethylethylenediamine with the more accessible N,N-dimethylethylenediamine and formaldehyde significantly lowers the raw material bill of materials. Additionally, the dramatic improvement in reaction yield reduces the effective cost per kilogram of the active ingredient by minimizing material loss. The avoidance of p-toluenesulfonic acid also removes the cost associated with neutralizing large volumes of acidic waste and treating heavy metal or organic residues, leading to substantial cost savings in waste management and overall production overhead.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche reagents that have limited global suppliers. This novel method utilizes widely available starting materials such as 4-fluoro-2-methoxy-5-nitroaniline and standard solvents like dioxane and n-butanol. By diversifying the chemical inputs and avoiding proprietary or hard-to-source catalysts, manufacturers can mitigate the risk of supply disruptions. The robustness of the anhydrous acid catalysis also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the production schedule and ensuring consistent delivery timelines for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns with modern green chemistry principles. The use of water as a solvent for the catalytic hydrogenation step drastically reduces the volume of volatile organic compounds (VOCs) emitted during production. Furthermore, the high selectivity of the reaction minimizes the generation of hazardous byproducts, simplifying the effluent treatment process. This environmental compatibility facilitates easier regulatory approval and reduces the long-term liability associated with hazardous waste storage, making it an ideal candidate for sustainable commercial manufacturing facilities aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Oxitinib Mesylate using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this route is superior for industrial application. Understanding these nuances is vital for technical teams evaluating potential technology transfers or licensing opportunities for next-generation oncology drug production.
Q: Why is anhydrous methanesulfonic acid preferred over p-toluenesulfonic acid in this synthesis?
A: Anhydrous methanesulfonic acid prevents the hydrolysis of the chloro-pyrimidine intermediate into hydroxy-impurities, which are difficult to remove. It also eliminates the risk of generating genotoxic ethyl p-toluenesulfonate impurities associated with p-TsOH in alcohol solvents.
Q: How does the Eschweiler-Clarke reaction improve the process compared to direct alkylation?
A: Using N,N-dimethylethylenediamine followed by Eschweiler-Clarke alkylation avoids the use of expensive and impure N,N,N'-trimethylethylenediamine. This strategy significantly reduces byproduct formation and eliminates the need for column chromatography purification.
Q: What safety advantages does the isopropyl acetate salt formation offer?
A: Using isopropyl acetate for salt formation avoids the generation of genotoxic alkyl methanesulfonates (such as ethyl or isopropyl methanesulfonate) that can occur with other solvents, thereby enhancing the safety profile of the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxitinib Mesylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of the CN113651800A process are executed with precision at any volume. We maintain stringent purity specifications through our rigorous QC labs, employing advanced analytical techniques to monitor for genotoxic impurities and ensure that every batch of Oxitinib Mesylate meets the highest global pharmacopeial standards. Our commitment to quality assurance guarantees that your supply chain remains uninterrupted and compliant with international regulatory requirements.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your oncology portfolio. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and market goals. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can drive efficiency and value for your organization.
