Advanced Cyanide-Free Synthesis of Apalutamide Intermediates for Commercial Scale-Up
Advanced Cyanide-Free Synthesis of Apalutamide Intermediates for Commercial Scale-Up
The pharmaceutical landscape for prostate cancer treatment has been significantly advanced by the development of second-generation non-steroidal androgen inhibitors, specifically Apalutamide. However, the manufacturing of this critical Active Pharmaceutical Ingredient (API) has historically faced substantial hurdles regarding safety and scalability. Patent CN115536634A introduces a transformative synthetic methodology that addresses these challenges by fundamentally redesigning the construction of the spirocyclic core. This innovation replaces hazardous cyanide-based chemistry with a greener, more robust pathway utilizing 1-aminocyclobutanecarboxylic acid. For R&D directors and supply chain leaders, this shift represents not merely a procedural update but a strategic opportunity to secure a more reliable pharmaceutical intermediates supplier network while drastically mitigating regulatory and environmental risks associated with toxic reagent handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in WO2007126765, have long relied on high-risk reagents that complicate industrial adoption. As illustrated in the reaction scheme below, traditional routes typically involve the condensation of benzamide derivatives with cyclobutanone and sodium cyanide under acidic conditions. Furthermore, the final cyclization step often necessitates microwave irradiation, a technology that presents significant engineering challenges when transitioning from laboratory benchtop to multi-ton reactor vessels. The reliance on sodium cyanide introduces severe safety protocols, expensive waste treatment requirements, and potential supply chain disruptions due to the tightly regulated nature of cyanide salts. These factors collectively inflate the cost of goods sold (COGS) and limit the agility of manufacturers to respond to market demand fluctuations.
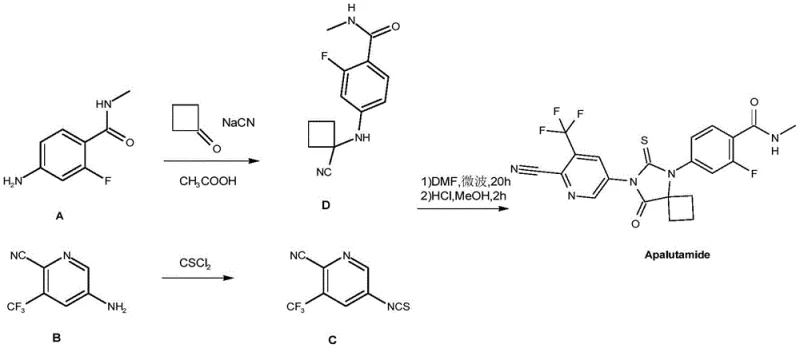
The Novel Approach
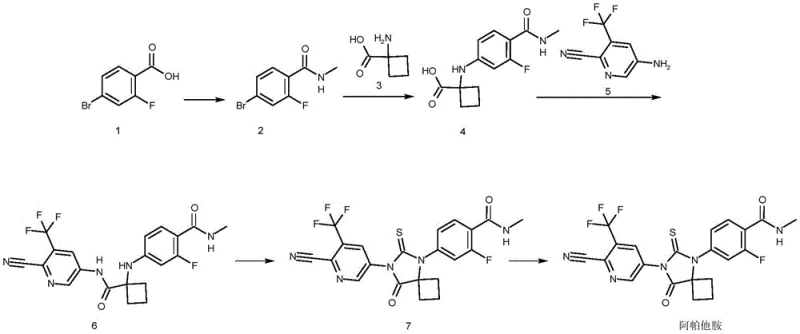
In stark contrast, the novel approach detailed in CN115536634A constructs the molecular architecture through a stepwise assembly that prioritizes reagent stability and process safety. The new route initiates with the conversion of 2-fluoro-4-halobenzoic acid into a stable amide intermediate, which then undergoes a copper-catalyzed substitution with 1-aminocyclobutanecarboxylic acid. This specific building block is notably superior to cyclobutanone; it is a solid with stable properties, easier to refine, and generates fewer side-product impurities during subsequent transformations. By eliminating the need for microwave promotion and toxic cyanides, this methodology facilitates cost reduction in API manufacturing through simplified equipment requirements and reduced safety overhead, making it an ideal candidate for continuous or batch processing in standard chemical plants.
Mechanistic Insights into Copper-Catalyzed Substitution and Cyclization
The core of this synthetic breakthrough lies in the efficient formation of the C-N bond between the aromatic ring and the cyclobutane moiety. In the conversion of Compound 2 to Compound 4, the process utilizes a copper salt catalyst, preferably cuprous chloride, in the presence of an inorganic base such as potassium carbonate. Mechanistically, this likely proceeds via a nucleophilic aromatic substitution or a copper-mediated Ullmann-type coupling, where the amine nitrogen of the cyclobutane derivative attacks the electron-deficient aromatic ring activated by the fluorine and amide groups. The choice of polar aprotic solvents like DMAc or DMF is critical here, as they stabilize the transition state and solubilize the inorganic base, ensuring high conversion rates at temperatures between 95°C and 105°C. This robust catalytic system avoids the use of expensive palladium catalysts, further contributing to the economic viability of the process for large-scale production.
Following the assembly of the linear precursor (Compound 6), the formation of the spiro-hydantoin ring (Compound 7) is achieved using a thiocarbonyl source. Unlike the hazardous thiophosgene gas used in older methods, this patent advocates for the use of phenyl thiocarbonate or thiocarbonyl diimidazole. These solid or liquid reagents react with the urea linkage in Compound 6 under mild thermal conditions (55-60°C) in the presence of an activator like DMAP and a tertiary amine base. This cyclization step is highly selective, minimizing the formation of polymeric byproducts. Crucially, the subsequent purification utilizes a binary solvent system of ethyl acetate and n-heptane. This specific ratio creates a solubility differential that precipitates the desired product while keeping structurally related impurities, such as the open-chain urea or over-reacted species, in solution, thereby achieving a purity profile exceeding 99.5% essential for regulatory compliance.
How to Synthesize Apalutamide Efficiently
The synthesis of Apalutamide via this novel pathway involves a logical sequence of acylation, substitution, condensation, and cyclization reactions that are amenable to standard chemical processing equipment. The process begins with the activation of the benzoic acid derivative, followed by the critical copper-catalyzed coupling that installs the cyclobutane ring. Subsequent amide coupling with the pyridine fragment and final ring closure yield the crude API, which is then polished through a specialized crystallization protocol. The detailed standardized synthesis steps, including specific reagent equivalents, temperature profiles, and workup procedures, are outlined in the guide below.
- Preparation of Amide Intermediate (Compound 2) via acylation and condensation with methylamine.
- Copper-Catalyzed Substitution reacting Compound 2 with 1-aminocyclobutanecarboxylic acid to form Compound 4.
- Condensation and Cyclization using CDI and phenyl thiocarbonate to form Compound 7, followed by crystallization to Apalutamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits beyond mere technical feasibility. The primary advantage stems from the elimination of Schedule 1 toxic substances like sodium cyanide, which simplifies logistics, reduces insurance premiums, and minimizes the risk of production stoppages due to regulatory audits. Furthermore, the use of 1-aminocyclobutanecarboxylic acid as a starting material ensures a more stable supply chain, as this reagent is less prone to degradation and easier to source in bulk quantities compared to volatile ketones or hazardous gases. This stability translates directly into enhanced supply chain reliability, allowing for consistent production schedules and reduced lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of toxic cyanide reagents eliminates the need for specialized containment systems and expensive hazardous waste disposal protocols, leading to substantial cost savings. Additionally, the replacement of microwave heating with conventional thermal heating allows for the use of standard glass-lined or stainless steel reactors, avoiding the capital expenditure associated with specialized microwave infrastructure. The high efficiency of the ethyl acetate/n-heptane purification system also reduces solvent consumption and loss, further optimizing the overall production economics.
- Enhanced Supply Chain Reliability: By utilizing stable, non-hazardous raw materials, the manufacturing process becomes less susceptible to supply disruptions caused by transportation restrictions on dangerous goods. The robustness of the copper-catalyzed step ensures high yields and consistent quality, reducing the need for re-processing or batch rejection. This reliability is critical for maintaining uninterrupted supply to downstream API manufacturers and ensuring that patient access to life-saving medications is not compromised by production delays.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, significantly reducing the environmental footprint of the manufacturing operation. The avoidance of heavy metal catalysts (other than low-level copper which is easily removed) and toxic gases aligns with increasingly stringent global environmental regulations. This compliance facilitates easier permitting for new production facilities and supports the corporate sustainability goals of pharmaceutical partners, making the commercial scale-up of complex pharmaceutical intermediates both technically and environmentally sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on purity profiles, safety metrics, and scalability potential. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is this new synthetic route considered safer than conventional methods?
A: This route eliminates the use of highly toxic sodium cyanide and potassium cyanide, as well as thiophosgene gas. Instead, it utilizes stable solid reagents like 1-aminocyclobutanecarboxylic acid and phenyl thiocarbonate, significantly reducing environmental and operator hazards.
Q: How does the process ensure high purity of the final intermediate?
A: The process employs a specific purification system using a mixture of ethyl acetate and n-heptane. This solvent combination effectively removes key impurities (such as HCS702 and HCS713), achieving a product content exceeding 99.5% without requiring complex chromatography.
Q: Is this method suitable for large-scale industrial production?
A: Yes, unlike prior art that relies on difficult-to-scale microwave heating, this method uses standard thermal heating (55-60°C) and common organic solvents. The use of stable raw materials and simplified workup procedures makes it highly adaptable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apalutamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is paramount for the future of pharmaceutical manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this cyanide-free process are fully realized in a commercial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped to detect and quantify trace impurities, guaranteeing that every batch of Apalutamide intermediate meets the highest global standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive value and security for your organization.
