Industrial Scale Synthesis of Apalutamide Intermediates via Novel Copper-Catalyzed Route
The global demand for next-generation androgen receptor inhibitors continues to surge, driven by the critical need for effective treatments in non-metastatic castration-resistant prostate cancer (NM-CRPC). At the forefront of this therapeutic landscape is Apalutamide, a molecule whose complex spiro-hydantoin structure has historically posed significant challenges for process chemists aiming for commercial viability. A groundbreaking patent, CN113402466A, filed in September 2021, introduces a transformative synthetic strategy that fundamentally reimagines the construction of the Apalutamide core. This innovation pivots away from hazardous cyanide chemistry and expensive noble metal catalysis, offering a pathway that is not only chemically elegant but also commercially robust. By leveraging a novel intermediate designated as Formula 4, this methodology ensures high stability and exceptional repeatability, addressing the perennial pain points of yield loss and impurity accumulation that plague traditional API manufacturing.
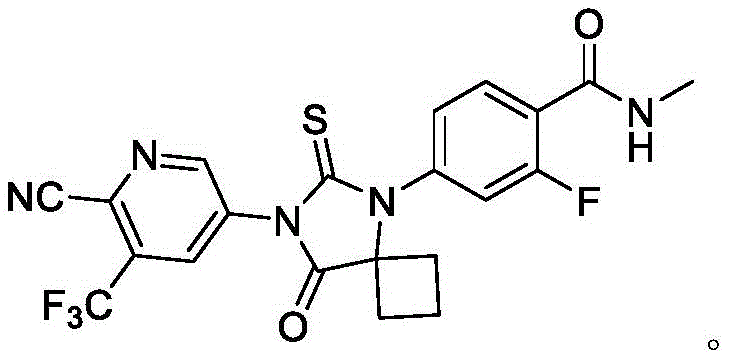
For procurement directors and supply chain managers, the implications of this patent are profound. It signals a shift towards a more sustainable and cost-efficient supply chain for this high-value oncology ingredient. The elimination of toxic reagents reduces the burden on environmental compliance teams, while the use of earth-abundant copper catalysts instead of palladium drastically lowers raw material costs. As we delve deeper into the technical specifics, it becomes clear that this route represents a significant leap forward in the industrial preparation of pharmaceutical intermediates, promising a reliable supply of high-purity Apalutamide for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Apalutamide has been fraught with operational hazards and economic inefficiencies that hinder large-scale production. Early literature, such as WO2007126765A2, described routes relying heavily on sodium cyanide (NaCN) for the introduction of the nitrile group and the construction of the cyclobutane ring. The use of NaCN presents severe safety risks, requiring specialized containment facilities and rigorous waste treatment protocols that inflate operational expenditures. Furthermore, these legacy processes often employed thiophosgene, a highly toxic and volatile reagent, under acidic conditions, creating a dangerous working environment. Another significant bottleneck in prior art, exemplified by PCT patent WO2016100645, was the reliance on palladium-catalyzed carbonyl insertion reactions. While chemically effective, palladium is a precious metal with volatile pricing and supply chain vulnerabilities. The requirement for noble metals not only increases the Bill of Materials (BOM) cost but also necessitates complex metal scavenging steps to meet stringent residual metal specifications for pharmaceutical products. Additionally, some existing methods utilize microwave irradiation for final cyclization steps, a technique that is notoriously difficult to scale from gram-scale laboratory synthesis to multi-ton industrial reactors.
The Novel Approach
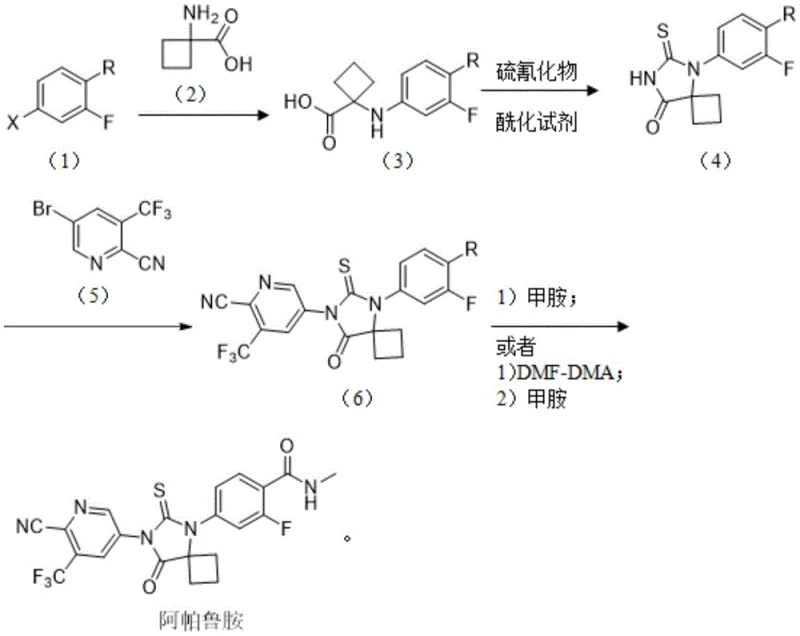
The methodology disclosed in CN113402466A offers a decisive break from these conventional constraints by introducing a streamlined, safety-first approach centered around a new intermediate, Formula 4. Instead of building the cyclobutane ring through dangerous cyanide condensation, this route utilizes 1-aminocyclobutanecarboxylic acid hydrochloride as a starting material. This reagent is a stable, crystalline solid that is far easier to handle, store, and dose than gaseous or liquid cyanide sources. The core transformation involves a copper-catalyzed coupling reaction between a halo-benzamide derivative and this amino acid, followed by a clever one-pot cyclization using thionyl chloride and a thiocyanide salt. This sequence constructs the critical spiro[3.4]octane-5,7-dione scaffold with high fidelity. By avoiding the need for microwave promotion and replacing palladium with inexpensive copper salts, the new process dramatically simplifies the engineering requirements for production. The result is a synthetic pathway that maintains high yields and purity while being inherently safer and more economically viable for kilogram-to-ton scale manufacturing.
Mechanistic Insights into Copper-Catalyzed Ullmann Coupling and Spiro-Cyclization
The heart of this innovative synthesis lies in the mechanistic elegance of the copper-catalyzed C-N bond formation and the subsequent intramolecular cyclization. The process begins with the coupling of a halogenated benzamide (Formula 1) and 1-aminocyclobutanecarboxylic acid (Formula 2). This transformation is mediated by a copper(I) catalyst, such as cuprous iodide, in the presence of a strong organic base like DBU (1,8-Diazabicyclo[5.4.0]undec-7-ene). Mechanistically, this proceeds via an oxidative addition-reductive elimination cycle typical of Ullmann-type condensations, where the copper center facilitates the nucleophilic attack of the amine nitrogen onto the aryl halide. The choice of solvent, typically polar aprotic solvents like DMSO or DMF, is crucial for stabilizing the charged intermediates and ensuring high conversion rates. This step effectively installs the cyclobutane-amino acid motif onto the aromatic core without the need for harsh conditions, preserving the integrity of sensitive functional groups.
Following the coupling, the route employs a sophisticated one-pot strategy to generate the spiro-hydantoin ring system found in Formula 4. The carboxylic acid moiety of the intermediate is first activated by thionyl chloride to form a reactive acyl chloride species in situ. Upon the addition of a thiocyanide salt (such as ammonium thiocyanate), the sulfur atom acts as a nucleophile, attacking the acyl chloride to form a transient acyl thiocyanate. This unstable intermediate rapidly undergoes intramolecular cyclization, where the neighboring amide nitrogen attacks the thiocarbonyl carbon, closing the five-membered hydantoin ring. This cascade reaction is highly efficient, driving the equilibrium towards the desired spiro-cycle while minimizing the formation of hydrolysis byproducts. The precise control of temperature during this phase—maintaining acylation at 15-35°C and cyclization at 40-60°C—is critical for suppressing side reactions and ensuring the high purity (>98%) observed in the examples. This mechanistic understanding allows process engineers to tightly control critical process parameters (CPPs) to guarantee consistent product quality.
How to Synthesize Apalutamide Intermediate Efficiently
The synthesis of the key spiro-hydantoin intermediate requires precise control over reaction stoichiometry and thermal profiles to maximize yield and minimize impurities. The patented procedure outlines a robust protocol that begins with the copper-catalyzed coupling in a polar solvent, followed by a seamless transition to the cyclization step without isolating the unstable acid chloride. This telescoped approach reduces unit operations and solvent consumption. For detailed operational parameters, including specific molar ratios, solvent volumes, and workup procedures, please refer to the standardized synthesis guide below.
- Perform a copper-catalyzed coupling reaction between a halo-benzamide derivative and 1-aminocyclobutanecarboxylic acid in the presence of a base like DBU to form the amino-acid linked intermediate.
- React the resulting intermediate with an acylating agent such as thionyl chloride to generate an acyl chloride species in situ.
- Add a thiocyanide salt to the reaction mixture to induce cyclization, forming the stable spiro[3.4]octane-5,7-dione core structure.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and supply continuity, the adoption of this novel synthetic route offers compelling strategic advantages. The shift from legacy cyanide-based chemistry to this amino-acid coupling method fundamentally alters the cost structure and risk profile of Apalutamide production. By eliminating the need for specialized cyanide handling infrastructure and expensive palladium catalysts, manufacturers can achieve substantial cost savings in both raw materials and waste management. Furthermore, the use of stable, crystalline starting materials enhances supply chain reliability, reducing the risk of production stoppages due to reagent instability or scarcity. The process's inherent safety and scalability make it an ideal candidate for long-term commercial supply agreements.
- Cost Reduction in Manufacturing: The replacement of noble metal palladium catalysts with earth-abundant copper salts results in a drastic reduction in catalyst costs, which is particularly significant given the volatility of precious metal markets. Additionally, the avoidance of toxic reagents like sodium cyanide and thiophosgene eliminates the need for costly detoxification processes and specialized containment equipment, leading to lower capital expenditure (CAPEX) and operational expenditure (OPEX). The high yields reported in the patent examples further contribute to cost efficiency by maximizing the output per batch and reducing the volume of raw materials required per kilogram of final API.
- Enhanced Supply Chain Reliability: The reliance on 1-aminocyclobutanecarboxylic acid hydrochloride as a key building block provides a significant logistical advantage. Unlike gaseous or highly toxic liquid reagents that require complex logistics and storage, this solid amino acid derivative is stable, easy to transport, and readily available from multiple chemical suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks. Moreover, the robustness of the copper-catalyzed reaction ensures consistent batch-to-batch performance, allowing for predictable production scheduling and reliable delivery timelines to downstream formulation partners.
- Scalability and Environmental Compliance: The process conditions described in the patent are exceptionally mild, with key reaction steps occurring at temperatures between 15°C and 100°C. This thermal profile is fully compatible with standard stainless steel reactors used in multipurpose pharmaceutical plants, removing the need for exotic metallurgy or specialized microwave equipment. From an environmental perspective, the reduction in toxic waste streams aligns with increasingly stringent global regulations on pharmaceutical manufacturing emissions. The simplified workup procedures, which often involve straightforward crystallization rather than complex chromatography, further reduce solvent usage and energy consumption, supporting sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Apalutamide synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of the technology's capabilities and limitations for potential licensees and manufacturing partners.
Q: Why is the new synthesis route for Apalutamide considered safer than previous methods?
A: The new route replaces highly toxic sodium cyanide (NaCN) and thiophosgene with safer reagents like 1-aminocyclobutanecarboxylic acid and ammonium thiocyanate, significantly reducing occupational hazards and waste treatment costs.
Q: What are the cost advantages of using copper catalysis over palladium in this synthesis?
A: Copper catalysts are substantially less expensive than noble metal palladium catalysts. Additionally, the copper-catalyzed Ullmann-type coupling operates under robust conditions that simplify downstream purification, leading to overall process cost reduction.
Q: How does this method improve the scalability of Apalutamide production?
A: The process utilizes mild reaction temperatures (15-60°C) and avoids harsh microwave conditions or cryogenic Grignard reactions, making it highly suitable for large-scale batch reactors and continuous manufacturing setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apalutamide Intermediate Supplier
The technical breakthroughs detailed in CN113402466A represent a significant opportunity for the pharmaceutical industry to secure a more stable and cost-effective supply of Apalutamide. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative chemistry to life. Our state-of-the-art facilities are equipped to handle copper-catalyzed reactions and sensitive cyclization steps with the utmost precision, ensuring that every batch meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to monitor impurity profiles, guaranteeing that our intermediates are perfectly suited for the demanding requirements of final API synthesis.
We invite global pharmaceutical partners to collaborate with us to leverage this superior synthetic route. By optimizing this process further, we can deliver significant value through improved margins and supply security. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your Apalutamide supply chain goals.
