Advanced Catalytic Synthesis of 3-Cyano Indoles for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient and safer pathways to construct critical heterocyclic scaffolds, particularly indole derivatives which serve as foundational structures for numerous bioactive molecules. A significant technological breakthrough in this domain is documented in Chinese Patent CN102718694B, which discloses a novel method for synthesizing 3-cyano substituted indole compounds. This patent represents a paradigm shift away from traditional, hazardous cyanation protocols by introducing a palladium and copper co-catalyzed system that utilizes tert-butyl isonitrile as a safe cyano source. For R&D directors and procurement managers alike, this innovation addresses two critical pain points: the severe toxicity associated with handling metal cyanides and the operational complexity of multi-step functionalization sequences. By enabling direct C-H bond activation at the 3-position of the indole ring, this technology streamlines the supply chain for high-value intermediates used in antidepressants and anticancer agents, offering a compelling value proposition for reliable pharmaceutical intermediate supplier partnerships focused on green chemistry and cost-effective manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a cyano group onto an aromatic or heteroaromatic ring has relied heavily on classical transformations that are increasingly viewed as unsustainable in modern industrial settings. The Sandmeyer reaction, while effective for aryl nitriles, suffers from poor atom economy, requires harsh acidic conditions, and involves multiple synthetic steps that erode overall yield and increase waste generation. Furthermore, alternative routes often necessitate the use of stoichiometric amounts of highly toxic reagents such as potassium cyanide or cuprous cyanide. As illustrated in prior art methods, the conversion of 3-formylindole to the corresponding nitrile typically employs potassium cyanide and ammonium dihydrogen phosphate, creating significant safety hazards and requiring specialized infrastructure for cyanide waste treatment.
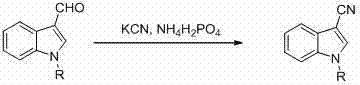
These legacy processes not only pose acute health risks to operators but also impose substantial regulatory burdens and disposal costs on manufacturing facilities. Additionally, many existing methods lack regioselectivity, often producing mixtures of isomers that require difficult and expensive purification steps, thereby complicating the commercial scale-up of complex pharmaceutical intermediates. The reliance on pre-functionalized starting materials, such as halogenated indoles, further adds to the raw material costs and extends the lead time for high-purity intermediates, making these conventional routes less attractive for large-scale production where margin compression is a constant challenge.
The Novel Approach
In stark contrast to these cumbersome traditional methods, the technology described in CN102718694B offers a streamlined, one-pot solution that leverages transition metal catalysis to achieve direct C-H cyanation. This novel approach utilizes tert-butyl isonitrile, a liquid reagent that is significantly safer and easier to handle than solid metal cyanides, effectively eliminating the immediate toxicity risks associated with cyanide ions. The reaction proceeds under moderate thermal conditions, typically between 100°C and 130°C, using common solvents like N,N-dimethylformamide, which simplifies the engineering requirements for the reactor setup. By bypassing the need for pre-halogenation or formylation, this method reduces the step count and improves the overall mass balance of the synthesis.

The versatility of this new protocol is evidenced by its compatibility with a wide range of N-substituted indoles, including those with phenyl, benzyl, allyl, and n-butyl groups, delivering the desired 3-cyano products with yields reaching up to 74%. This level of efficiency, combined with the operational simplicity of mixing reagents and heating, translates directly into reduced manufacturing costs and enhanced supply chain reliability. For procurement teams, this means access to a more robust supply of critical intermediates without the volatility associated with hazardous reagent sourcing, positioning this technology as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd/Cu Co-Catalyzed C-H Activation
The core of this technological advancement lies in the synergistic interaction between the palladium catalyst and the copper additive, which facilitates the activation of the relatively inert C-H bond at the 3-position of the indole ring. The proposed mechanism involves the initial coordination of the palladium species to the indole substrate, followed by a concerted metalation-deprotonation or electrophilic palladation step that generates a reactive organopalladium intermediate. The presence of copper trifluoroacetate is crucial, as it likely assists in the transmetallation process or acts as a Lewis acid to activate the tert-butyl isonitrile, enabling the transfer of the cyano group to the palladium center. This dual-catalyst system ensures high regioselectivity, preventing unwanted substitution at other positions on the indole scaffold and minimizing the formation of byproducts.
From an impurity control perspective, the use of tert-butyl isonitrile avoids the generation of inorganic cyanide salts that are notoriously difficult to remove from the final product matrix. The reaction byproducts are primarily organic and can be effectively separated during the standard aqueous workup and column chromatography purification steps described in the patent examples. The specific choice of ligands and additives, such as the acetate base, helps to maintain the catalytic cycle turnover number, ensuring that the precious metal catalyst is utilized efficiently. This mechanistic elegance results in a cleaner reaction profile, which is paramount for meeting the stringent purity specifications required for API intermediates, thereby reducing the burden on quality control laboratories and accelerating batch release times.
How to Synthesize 3-Cyano Substituted Indole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and safety. The process begins with the precise weighing of the indole substrate, the copper salt, and the tert-butyl isonitrile reagent, which are then dissolved in dry DMF under an inert atmosphere to prevent catalyst deactivation. The detailed standardized synthesis steps see the guide below, which outlines the optimal molar ratios and temperature profiles derived from the patent examples to ensure reproducible results.
- Dissolve the indole substrate, copper trifluoroacetate, and tert-butyl isonitrile in DMF solvent with a catalytic amount of palladium acetate.
- Heat the reaction mixture to between 100°C and 130°C and stir until the starting indole material is completely consumed as monitored by TLC.
- Quench the reaction with ammonia solution, extract the product with ethyl acetate, dry the organic phase, and purify the crude residue via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond simple chemical transformation. The elimination of highly toxic metal cyanides from the process flow drastically simplifies the regulatory compliance landscape, removing the need for specialized cyanide handling permits and expensive detoxification protocols. This shift not only mitigates the risk of operational shutdowns due to safety incidents but also lowers the total cost of ownership for the manufacturing facility by reducing insurance premiums and waste disposal fees. Furthermore, the use of readily available starting materials and common solvents ensures that the supply chain remains resilient against raw material shortages, enhancing the continuity of supply for downstream drug manufacturers.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot reaction significantly reduces processing time and labor costs compared to multi-step traditional routes. By avoiding the purchase and handling of expensive and hazardous cyanide salts, manufacturers can achieve substantial cost savings in raw material procurement and safety management. The high atom economy and improved yields mean less raw material is wasted, directly improving the gross margin of the final intermediate product and allowing for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: Relying on stable, non-hazardous reagents like tert-butyl isonitrile ensures a more predictable and secure supply chain, free from the strict transportation restrictions imposed on cyanide compounds. This reliability allows for larger batch sizes and longer campaign runs without the logistical bottlenecks associated with hazardous material delivery, ensuring that customers receive their orders on time and consistent quality is maintained across different production lots.
- Scalability and Environmental Compliance: The reaction conditions are mild and utilize standard equipment, making the scale-up from gram to ton quantities straightforward and low-risk. The reduced generation of toxic heavy metal waste and inorganic salts aligns with increasingly strict environmental regulations, facilitating easier permitting for new production lines and supporting corporate sustainability goals by minimizing the ecological footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 3-cyano indole synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for potential partners evaluating this process for their own supply chains.
Q: What is the primary safety advantage of this new synthesis method compared to traditional routes?
A: The primary safety advantage is the replacement of highly toxic metal cyanides (like KCN or CuCN) with tert-butyl isonitrile as the cyano source, significantly reducing hazardous waste handling risks.
Q: What yields can be expected from this catalytic process on an industrial scale?
A: According to the patent data, the process achieves yields of up to 74% for substrates like 1-phenyl-1H-indole, demonstrating robust efficiency suitable for commercial production.
Q: Does this method require complex protecting group strategies for the indole nitrogen?
A: No, the method is versatile and works effectively with various N-substituents including phenyl, benzyl, allyl, and n-butyl groups without the need for additional protection-deprotection steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyano Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalytic C-H activation technology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-cyano indole delivered meets the exacting standards required for subsequent drug synthesis.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this safer and more efficient synthetic route. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce your manufacturing costs with our expert expertise in advanced organic synthesis and process development.
