Advanced Synthesis of 3-Cyano Indole Intermediates for Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and safe methodologies for constructing complex heterocyclic scaffolds, particularly those serving as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN102718694B introduces a groundbreaking advancement in this domain by detailing a novel synthetic route for 3-cyano substituted indole compounds. These structures are pivotal precursors in the manufacture of renowned therapeutics such as Escitalopram (Lexapro) and Letrozole (Femara), where the cyano group acts as a versatile handle for further functionalization into amides, acids, or amines. The core innovation lies in the strategic replacement of hazardous metal cyanides with tert-butyl isonitrile, facilitated by a dual palladium and copper catalytic system. This shift not only addresses significant safety concerns regarding toxicity but also streamlines the operational workflow, offering a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a cyano group onto an aromatic or heteroaromatic ring has relied heavily on classical transformations that present substantial logistical and safety challenges for modern process chemistry. The Sandmeyer reaction, while effective for simple aryl systems, involves a multi-step sequence requiring the formation of unstable diazonium salts, which poses explosion risks and often results in modest overall yields due to decomposition pathways. 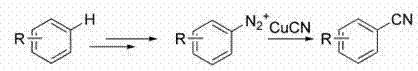 Furthermore, alternative strategies involving nucleophilic substitution of aryl halides typically necessitate harsh conditions and expensive ligands to activate the carbon-halogen bond, leading to increased production costs and waste generation. Another common route involves the conversion of 3-formylindoles using potassium cyanide; however, this approach mandates the prior synthesis of the aldehyde, adding unnecessary synthetic steps and exposing personnel to highly toxic inorganic cyanide salts. These legacy methods often suffer from poor atom economy and generate significant quantities of heavy metal waste, creating bottlenecks in both regulatory compliance and cost efficiency for large-scale operations.
Furthermore, alternative strategies involving nucleophilic substitution of aryl halides typically necessitate harsh conditions and expensive ligands to activate the carbon-halogen bond, leading to increased production costs and waste generation. Another common route involves the conversion of 3-formylindoles using potassium cyanide; however, this approach mandates the prior synthesis of the aldehyde, adding unnecessary synthetic steps and exposing personnel to highly toxic inorganic cyanide salts. These legacy methods often suffer from poor atom economy and generate significant quantities of heavy metal waste, creating bottlenecks in both regulatory compliance and cost efficiency for large-scale operations.
The Novel Approach
In stark contrast to these traditional limitations, the methodology disclosed in CN102718694B leverages direct C-H bond activation to install the cyano functionality in a single catalytic step. By utilizing tert-butyl isonitrile as a liquid, manageable cyanide equivalent, the process circumvents the need for handling lethal solid cyanides, thereby drastically reducing the safety infrastructure required for production. The reaction proceeds under relatively mild thermal conditions, typically between 100°C and 130°C, using standard polar aprotic solvents like DMF, which are readily available and easily recycled in industrial settings. This direct functionalization strategy eliminates the need for pre-functionalized substrates such as halides or aldehydes, effectively shortening the synthetic timeline and improving the overall material throughput. The simplicity of the workup procedure, involving basic aqueous quenching and organic extraction, further underscores the practical advantages of this novel approach for commercial manufacturing environments.
Mechanistic Insights into Pd/Cu-Catalyzed C-H Cyanation
The mechanistic elegance of this transformation relies on a synergistic catalytic cycle involving both palladium and copper species to activate the inert C-H bond at the 3-position of the indole ring. The process initiates with the coordination of the palladium catalyst to the electron-rich indole substrate, followed by a concerted metalation-deprotonation (CMD) or electrophilic palladation event that forms a stable organopalladium intermediate. 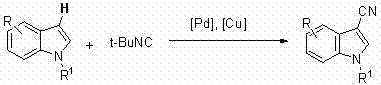 Subsequently, the tert-butyl isonitrile inserts into the palladium-carbon bond, a critical step that distinguishes this method from traditional cyanation protocols. The presence of the copper salt, specifically copper trifluoroacetate, plays a vital role in facilitating the transmetallation or assisting in the cleavage of the isonitrile C-N triple bond to deliver the cyano group efficiently. This dual-metal system ensures high turnover numbers and maintains catalytic activity throughout the reaction duration, preventing the accumulation of inactive palladium black which often plagues single-metal C-H activation processes. The result is a highly selective transformation that favors the thermodynamic product at the 3-position, minimizing regioisomeric impurities that are difficult to separate.
Subsequently, the tert-butyl isonitrile inserts into the palladium-carbon bond, a critical step that distinguishes this method from traditional cyanation protocols. The presence of the copper salt, specifically copper trifluoroacetate, plays a vital role in facilitating the transmetallation or assisting in the cleavage of the isonitrile C-N triple bond to deliver the cyano group efficiently. This dual-metal system ensures high turnover numbers and maintains catalytic activity throughout the reaction duration, preventing the accumulation of inactive palladium black which often plagues single-metal C-H activation processes. The result is a highly selective transformation that favors the thermodynamic product at the 3-position, minimizing regioisomeric impurities that are difficult to separate.
From an impurity control perspective, this mechanism offers distinct advantages over electrophilic substitution methods which often lead to poly-substitution or reaction at the nitrogen atom. The steric and electronic properties of the catalytic complex inherently direct the incoming cyano group to the C3 position, ensuring a clean reaction profile. Furthermore, the use of tert-butyl isonitrile avoids the generation of inorganic salt byproducts typical of metal cyanide reactions, resulting in a cleaner crude reaction mixture. This purity profile is crucial for pharmaceutical applications where strict limits on genotoxic impurities and heavy metal residues must be met. The ability to achieve yields up to 74% with minimal side products demonstrates the robustness of the catalytic system, providing R&D teams with a reliable platform for generating diverse libraries of indole derivatives for drug discovery programs.
How to Synthesize 3-Cyano Indole Efficiently
The practical implementation of this synthesis is designed to be straightforward, utilizing standard laboratory glassware and heating equipment without the need for specialized high-pressure reactors. The protocol involves dissolving the indole substrate, copper trifluoroacetate additive, and tert-butyl isonitrile in N,N-dimethylformamide, followed by the addition of a catalytic amount of palladium acetate. The detailed standardized synthesis steps are provided in the guide below.
- Dissolve indole substrate, copper trifluoroacetate, and tert-butyl isonitrile in DMF with palladium acetate catalyst.
- Heat the reaction mixture to 100-130°C and stir until the starting indole material is fully consumed.
- Quench with ammonia solution, extract with ethyl acetate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible operational improvements and risk mitigation strategies. The elimination of highly toxic metal cyanides from the supply chain removes a significant regulatory burden, simplifying the logistics of raw material sourcing and storage. Traditional cyanide sources often require specialized containment facilities and rigorous safety audits, which incur substantial overhead costs; by switching to tert-butyl isonitrile, facilities can operate with greater flexibility and reduced insurance liabilities. Moreover, the use of commodity chemicals like DMF and readily available indole starting materials ensures a stable supply base that is less susceptible to market volatility compared to specialized halogenated precursors. This stability is essential for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reduction in synthetic steps and the avoidance of expensive waste treatment protocols associated with cyanide disposal. By consolidating the synthesis into a single catalytic step, manufacturers save significantly on labor, energy, and solvent consumption compared to multi-step sequences involving protection and deprotection groups. The high atom economy of the C-H activation pathway means that a larger proportion of the starting mass is converted into the desired product, reducing the cost of goods sold (COGS). Additionally, the simplified purification process reduces the consumption of silica gel and eluents during chromatography, further driving down operational expenditures without compromising product quality.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for preventing production stoppages in API manufacturing. This method utilizes robust reaction conditions that are tolerant to minor variations in temperature and mixing, making it highly reproducible across different batch sizes and manufacturing sites. The availability of tert-butyl isonitrile from multiple chemical suppliers ensures that there is no single point of failure in the raw material supply chain. Furthermore, the moderate reaction temperatures allow for the use of standard stainless steel reactors, eliminating the need for exotic metallurgy required for corrosive acidic or basic conditions found in older methods, thus enhancing equipment longevity and uptime.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden challenges, but this methodology is inherently designed for scalability. The exothermic profile of the reaction is manageable, and the use of liquid reagents facilitates precise dosing and mixing in large vessels. From an environmental standpoint, the process aligns with green chemistry principles by avoiding the generation of stoichiometric amounts of heavy metal waste and toxic gas emissions. This compliance with increasingly stringent environmental regulations future-proofs the manufacturing process, ensuring long-term viability and reducing the risk of shutdowns due to non-compliance issues. The ability to produce high-purity intermediates with a lower environmental footprint is a key differentiator in the competitive fine chemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the feasibility and advantages of adopting this route for your specific project requirements.
Q: Why is tert-butyl isonitrile preferred over potassium cyanide for this synthesis?
A: Tert-butyl isonitrile serves as a non-toxic, liquid cyanide source that eliminates the severe safety hazards and regulatory burdens associated with handling solid metal cyanides like KCN or NaCN in large-scale manufacturing.
Q: What is the regioselectivity of this C-H activation method?
A: The patented Pd/Cu catalytic system exhibits high regioselectivity for the 3-position of the indole ring, minimizing the formation of unwanted 2-position isomers and simplifying downstream purification processes.
Q: Can this method be scaled for industrial production of API intermediates?
A: Yes, the process utilizes conventional solvents like DMF and moderate temperatures (100-130°C), making it highly suitable for commercial scale-up without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyano Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development pipelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify identity and assay. Our facility is equipped to handle the specific safety requirements of isonitrile chemistry, providing a secure and compliant environment for the production of these valuable building blocks.
We invite you to discuss how this innovative synthesis route can optimize your project economics and timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your supply chain goals. Let us be your partner in advancing the next generation of pharmaceutical therapies through superior chemical manufacturing.
