Revolutionizing Ensitrelvir Production: A Safe, High-Yield Acid-Catalyzed Route for Commercial Scale-up
The pharmaceutical industry is constantly seeking robust synthetic routes for critical antiviral agents, and recent advancements detailed in patent CN115141184A offer a transformative approach to the production of Ensitrelvir (S-217622), a potent SARS-CoV-2 3CL protease inhibitor. This patent discloses a novel preparation method that fundamentally shifts the reaction paradigm from hazardous base-catalyzed conditions to a safer, organic acid-catalyzed aromatic nucleophilic substitution. By replacing the traditionally used lithium bis(trimethylsilyl)amide (LiHMDS) with common organic acids, the process achieves a dramatic improvement in yield, escalating from a mere 25% in prior art to an impressive 70% in the new methodology. This technological leap not only addresses significant safety concerns associated with pyrophoric reagents but also aligns perfectly with the green chemistry principles demanded by modern regulatory frameworks. For stakeholders in the antiviral supply chain, this represents a pivotal opportunity to secure a more reliable API intermediate supplier capable of delivering high-purity materials with greater economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
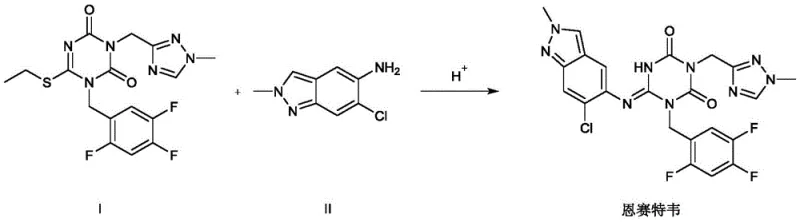
The Limitations of Conventional Methods
Historically, the synthesis of Ensitrelvir has relied heavily on the use of strong alkaline reagents, specifically LiHMDS, to facilitate the coupling of key precursors. As illustrated in the prior art reaction scheme, this conventional pathway necessitates extremely harsh operating conditions, including the dropwise addition of LiHMDS solution at cryogenic temperatures around 0°C. Such conditions introduce severe operational risks, primarily due to the intense exothermic nature of the reaction which is difficult to control on a large scale. Furthermore, the LiHMDS reagent is highly sensitive to moisture, mandating an absolutely anhydrous environment that significantly increases production costs through the need for specialized drying equipment and solvents. The inherent instability and danger of handling large quantities of pyrophoric bases make this route unsuitable for safe industrial expansion, while the low yield of 25% results in substantial material waste and inflated manufacturing costs.
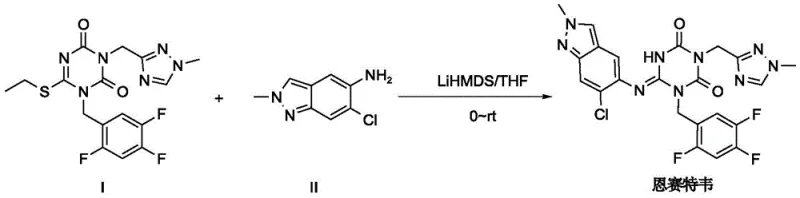
The Novel Approach
In stark contrast, the innovative method described in the patent utilizes organic acids such as formic acid, acetic acid, or oxalic acid to catalyze the aromatic nucleophilic substitution reaction. This shift allows the reaction to proceed under significantly milder conditions, with temperatures ranging from 0°C to 120°C, and preferably between 70°C and 100°C, eliminating the need for energy-intensive cryogenic cooling. The tolerance for moisture is vastly improved, removing the stringent requirement for absolute anhydrous environments and simplifying the overall process workflow. By avoiding the use of hazardous strong bases, the new route drastically reduces safety risks, making it far more amenable to commercial scale-up of complex antiviral intermediates. The result is a streamlined process that not only enhances operator safety but also delivers a threefold increase in yield, providing a compelling economic advantage for manufacturers.
Mechanistic Insights into Acid-Catalyzed Aromatic Nucleophilic Substitution
The core of this technological breakthrough lies in the mechanistic pathway where the organic acid facilitates the nucleophilic attack of the amino group from Compound II onto the N-heterocyclic ring of Compound I. Under acidic conditions, the protonation likely activates the leaving group or stabilizes the transition state, allowing the formation of an unstable intermediate that subsequently eliminates ethanethiol to yield the final Ensitrelvir product. This mechanism avoids the aggressive deprotonation steps required by LiHMDS, thereby preserving the integrity of sensitive functional groups within the molecular structure. The detailed reaction mechanism highlights how the acid catalyst lowers the activation energy barrier for the substitution, enabling the reaction to proceed efficiently at elevated temperatures without decomposing the reactants. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters for maximum throughput and purity.
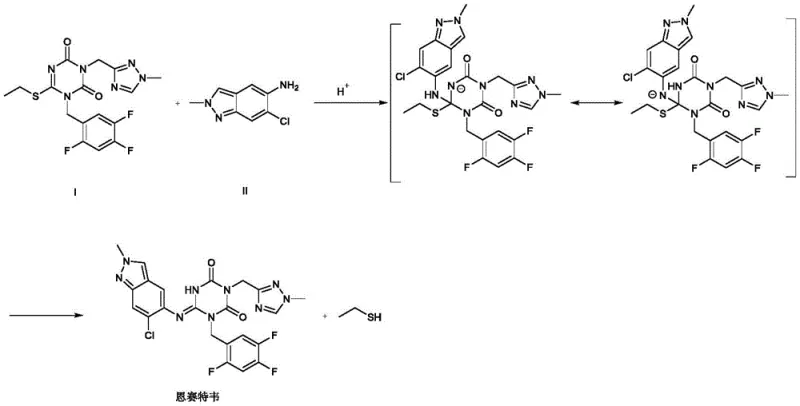
Furthermore, the patent elucidates critical impurity control mechanisms that are vital for ensuring high-purity Ensitrelvir suitable for pharmaceutical applications. It was discovered that maintaining the molar ratio of Compound I to organic acid between 1:1.0 and 1:10 is essential; exceeding this range leads to excessive hydrogen ion concentration, which can cause the 1,3,5-triazine ring to open, generating undesirable by-products. Similarly, temperature control is paramount, as reactions exceeding 120°C promote the detachment of the chlorine atom from the indole ring, leading to dechlorinated impurities. By strictly adhering to these optimized parameters, manufacturers can effectively suppress side reactions, ensuring that the final product meets stringent purity specifications required for clinical use. This level of control over the impurity profile is a key differentiator for any reliable API intermediate supplier targeting the global pharmaceutical market.
How to Synthesize Ensitrelvir Efficiently
Implementing this advanced synthesis route requires precise adherence to the optimized reaction conditions outlined in the patent to achieve the reported yields of up to 71.83%. The process involves combining Compound I and Compound II in a suitable solvent system, such as tetrahydrofuran (THF), N,N-dimethylformamide (DMF), or tert-butanol, followed by the addition of the selected organic acid catalyst. The reaction mixture is then heated to the optimal temperature range of 70°C to 100°C and maintained for a duration of 10 to 20 hours to ensure complete conversion. Following the reaction, standard workup procedures involving neutralization, extraction, and recrystallization are employed to isolate the product with purity levels exceeding 99%. The detailed standardized synthesis steps below provide a comprehensive guide for technical teams looking to replicate this high-efficiency process in a pilot or production setting.
- Combine Compound I, Compound II, and a suitable solvent (e.g., THF, DMF) in a reaction vessel.
- Add an organic acid catalyst (e.g., formic acid, acetic acid) maintaining a molar ratio of 1: 1.0 to 1:10 relative to Compound I.
- Heat the mixture to 70-100°C for 10-20 hours, then work up via extraction and recrystallization to obtain high-purity Ensitrelvir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this acid-catalyzed synthesis route offers profound strategic benefits that extend beyond simple yield improvements. The elimination of hazardous reagents like LiHMDS removes the need for specialized storage and handling protocols, thereby reducing insurance costs and regulatory compliance burdens associated with dangerous goods. Additionally, the ability to run the reaction at ambient or moderately elevated temperatures rather than cryogenic conditions significantly lowers energy consumption, contributing to substantial cost savings in utilities. The robustness of the process against moisture ingress further simplifies logistics, as solvents and reagents do not require rigorous drying, streamlining the supply chain and reducing lead time for high-purity pharmaceutical intermediates. These factors collectively enhance the overall resilience and cost-effectiveness of the manufacturing operation.
- Cost Reduction in Manufacturing: The transition from expensive, hazardous strong bases to inexpensive, commodity organic acids drastically reduces raw material costs. Moreover, the significant increase in yield from 25% to 70% means that less starting material is required to produce the same amount of final product, effectively lowering the cost per kilogram. The simplification of the process also reduces the need for complex reactor setups capable of handling extreme exotherms, leading to lower capital expenditure and maintenance costs for production facilities.
- Enhanced Supply Chain Reliability: By utilizing widely available organic acids and solvents that are less sensitive to environmental conditions, the supply chain becomes more robust against disruptions. The removal of strict anhydrous requirements allows for more flexible sourcing of raw materials, reducing the risk of production stoppages due to reagent quality issues. This stability ensures a consistent flow of materials, which is critical for meeting the demanding delivery schedules of global pharmaceutical clients.
- Scalability and Environmental Compliance: The safer reaction profile facilitates easier scale-up from laboratory to commercial production without the engineering challenges posed by pyrophoric reagents. The greener nature of the process, characterized by reduced waste generation and lower energy usage, aligns with increasingly strict environmental regulations, minimizing the risk of fines and enhancing the corporate sustainability profile. This makes the technology highly attractive for long-term investment and capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the new Ensitrelvir synthesis method, providing clarity on its implementation and benefits. These insights are derived directly from the experimental data and technical specifications found in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of adopting this technology within existing manufacturing frameworks.
Q: Why is the acid-catalyzed method superior to the traditional LiHMDS route?
A: The traditional route uses LiHMDS, a hazardous strong base requiring strict anhydrous conditions and cryogenic temperatures (-20°C to 0°C), resulting in only 25% yield. The new acid-catalyzed method operates at mild temperatures (70-100°C), eliminates moisture sensitivity, and boosts yield to approximately 70%.
Q: How does this process control impurities during synthesis?
A: Impurity formation is minimized by strictly controlling the molar ratio of Compound I to organic acid (1:1.0-10) to prevent triazine ring opening, and keeping reaction temperatures below 120°C to avoid dechlorination of the indole ring.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the removal of pyrophoric reagents like LiHMDS and the elimination of cryogenic cooling requirements significantly enhance operational safety and simplify equipment needs, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ensitrelvir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic technologies to meet the evolving demands of the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the acid-catalyzed Ensitrelvir route can be seamlessly transferred to large-scale manufacturing. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of API intermediate meets the highest international standards. Our capability to handle complex chemistries safely and efficiently positions us as a strategic partner for companies seeking to secure their supply chains for next-generation antiviral therapies.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this superior method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your drug development pipeline.
