Advanced Metal-Free Synthesis of Ebselen Compounds for Pharmaceutical Scale-Up
Advanced Metal-Free Synthesis of Ebselen Compounds for Pharmaceutical Scale-Up
The landscape of organoselenium chemistry is undergoing a significant transformation driven by the demand for safer, more efficient synthetic routes for biologically active molecules. A pivotal development in this field is detailed in patent CN114773294A, which discloses a novel preparation method for ebselen compounds, specifically benzo[d][1,2]selenazol-3(2H)-one derivatives. Ebselen is renowned as a potent glutathione peroxidase (GSH-Px) mimic with extensive therapeutic potential ranging from neuroprotection in Alzheimer's disease to anti-inflammatory applications. However, traditional synthetic methodologies have often been plagued by harsh reaction conditions, limited substrate scope, and the necessity for toxic transition metals. This new technology addresses these critical bottlenecks by introducing a mild, metal-free oxidative cyclization strategy that follows a visible-light mediated C-H selenylation, offering a robust pathway for the reliable pharmaceutical intermediates supplier seeking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
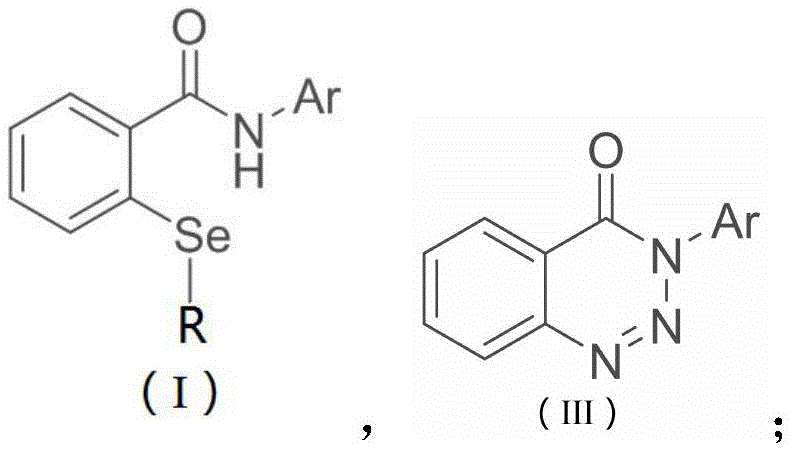
Historically, the construction of the seleno-nitrogen heterocyclic core found in ebselen has relied heavily on two primary strategies, both of which present substantial challenges for modern process chemistry. The first approach involves the diazotization of anthranilic acid followed by selenylation to form bis-seleno-phthalates, which then require further conversion with amines or acid chlorides; this multi-step sequence is not only lengthy but often suffers from poor atom economy and strict control requirements for diazonium species. The second, more common method utilizes an o-benzoylaniline lithiation strategy, where substrates are treated with n-butyllithium at cryogenic temperatures before reacting with selenium powder and closing the ring with copper dibromide. This lithiation route is particularly problematic for cost reduction in API manufacturing because it demands rigorously anhydrous and anaerobic conditions, utilizes hazardous pyrophoric reagents, and inevitably introduces copper residues that require expensive and time-consuming removal processes to meet stringent pharmaceutical purity standards.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN114773294A offers a streamlined, two-stage protocol that drastically simplifies the operational complexity while enhancing safety profiles. The innovation begins with a photoredox-catalyzed C-H selenylation that constructs the critical carbon-selenium bond under ambient conditions, utilizing visible light and stable selenosulfonate sources rather than elemental selenium or air-sensitive organolithiums. This is followed by a remarkably mild oxidative cyclization step that proceeds without any transition metal catalysts, relying instead on inexpensive N-halosuccinimides to drive the ring closure. By eliminating the need for cryogenic cooling, pyrophoric reagents, and heavy metal catalysts, this novel approach provides a foundation for industrial production that is inherently safer, more environmentally benign, and significantly easier to scale for the commercial scale-up of complex heterocycles.
Mechanistic Insights into Photoredox C-H Selenylation and Oxidative Cyclization
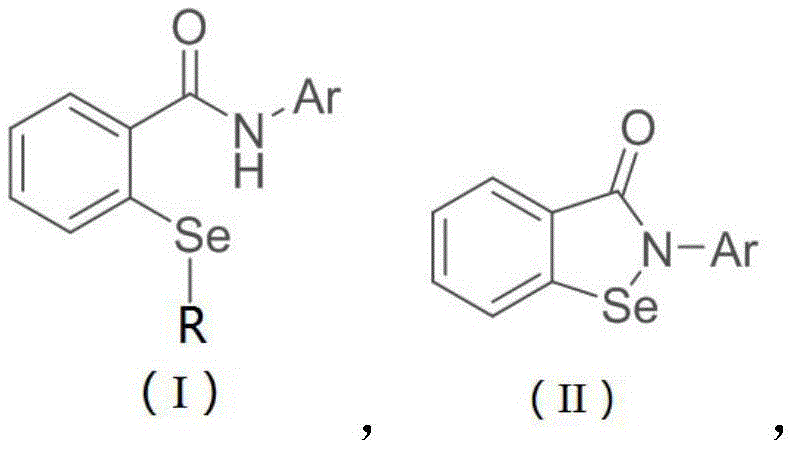
The first stage of this synthesis leverages the power of visible-light photoredox catalysis to achieve direct C-H functionalization, a transformation that was previously difficult to control with high selectivity. In this mechanism, a photosensitive catalyst, such as the metal iridium complex Ir(ppy)3 or organic alternatives like Eosin Y, absorbs blue LED irradiation to reach an excited state capable of single-electron transfer (SET). This excited species facilitates the generation of selenium-centered radicals from selenosulfonate precursors, which then selectively attack the electron-deficient aromatic system of the benzotriazinone substrate. The use of a base like DIPEA and a polar aprotic solvent such as DMSO is critical here, as it stabilizes the radical intermediates and promotes the efficient formation of the o-selenoether benzamide bond. This photochemical step operates at room temperature (20°C to 30°C), avoiding the thermal degradation pathways often seen in traditional thermal radical reactions, thereby ensuring high-purity ebselen derivatives with minimal byproduct formation.
Following the installation of the selenium moiety, the second stage involves an intramolecular oxidative cyclization that forms the final isoselenazolone ring. Unlike traditional copper-mediated cyclizations, this process utilizes N-chlorosuccinimide (NCS) or similar N-halosuccinimides as the stoichiometric oxidant. The reaction initiates at low temperatures (0°C) in dichloromethane to generate a reactive selenenyl halide intermediate, which is then heated to 50°C in DMSO to promote nucleophilic attack by the amide carbonyl oxygen or nitrogen, depending on the tautomeric state, ultimately closing the five-membered ring. This metal-free cyclization mechanism is crucial for reducing lead time for high-purity selenium compounds because it bypasses the need for downstream metal scavenging resins or complex aqueous workups designed to remove trace copper, resulting in a cleaner crude profile that simplifies final purification via standard column chromatography or crystallization.
How to Synthesize Ebselen Derivatives Efficiently
The practical implementation of this synthesis route is designed for reproducibility and ease of handling, making it accessible for both laboratory R&D and pilot plant operations. The process begins by charging a reaction vessel with the benzotriazinone precursor, a selenosulfonate source, and the photocatalyst under a protective nitrogen atmosphere, followed by irradiation with blue LEDs for approximately 10 to 12 hours to ensure complete conversion to the selenoether intermediate. Once the photochemical step is complete, the reaction mixture undergoes a straightforward extractive workup to isolate the intermediate, which is then subjected to the oxidative cyclization conditions using NCS in a two-solvent system of dichloromethane and DMSO. For detailed standardized operating procedures, safety data, and specific stoichiometric ratios optimized for your specific substrate, please refer to the technical guide below.
- Perform visible-light mediated C-H selenylation of benzotriazinone derivatives using a selenosulfonate source and an iridium or organic photocatalyst in DMSO.
- React the resulting o-selenoether benzamide intermediate with an oxidant such as N-chlorosuccinimide (NCS) in dichloromethane at 0°C.
- Heat the reaction mixture in DMSO at 50°C to facilitate intramolecular cyclization, yielding the target ebselen compound after purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic advantages that extend beyond mere technical feasibility. By shifting away from lithiation-based chemistries, manufacturers can eliminate the logistical burdens associated with sourcing and storing hazardous, short-shelf-life reagents like n-butyllithium, which often require specialized cold-chain logistics and pose significant safety risks during transport. Furthermore, the exclusion of transition metal catalysts in the critical ring-closing step removes a major cost center associated with precious metal recovery and regulatory compliance for residual metal limits in active pharmaceutical ingredients. This streamlined process flow enhances overall equipment effectiveness by reducing cycle times and minimizing the need for specialized reactor linings or cleaning protocols required for heavy metal containment, thereby driving substantial cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as copper dibromide and the avoidance of cryogenic cooling systems significantly lowers the operational expenditure per kilogram of product. Without the need for rigorous metal scavenging steps or the disposal of heavy metal waste streams, the downstream processing costs are drastically simplified, allowing for a more competitive pricing structure in the global market for selenium-based therapeutics.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available selenosulfonates and common organic oxidants like NCS ensures a robust supply chain that is less susceptible to the volatility often seen with specialty organometallic reagents. The mild reaction conditions (room temperature to 50°C) reduce the risk of batch failures due to thermal runaway or equipment malfunction, ensuring consistent delivery schedules and reliable inventory levels for long-term production contracts.
- Scalability and Environmental Compliance: The metal-free nature of the cyclization step aligns perfectly with green chemistry principles, reducing the environmental footprint of the manufacturing process by minimizing toxic waste generation. This inherent safety and environmental compatibility facilitate easier regulatory approval for commercial scale-up, allowing facilities to increase production volumes from pilot batches to multi-ton annual capacities without requiring extensive modifications to existing waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ebselen synthesis technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational parameters and scope of applicability for potential licensees or manufacturing partners looking to integrate this route into their existing pipelines. Understanding these nuances is essential for accurate process modeling and risk assessment prior to technology transfer.
Q: Does this synthesis method require transition metal catalysts for the cyclization step?
A: No, the cyclization step described in patent CN114773294A utilizes N-halosuccinimides (like NCS) as oxidants in DMSO, completely eliminating the need for copper or other transition metal catalysts typically required in traditional lithiation routes.
Q: What is the substrate scope for the aryl group in this ebselen synthesis?
A: The method demonstrates broad substrate tolerance, successfully accommodating substituted aryl groups including methyl, methoxy, halogen (fluoro, chloro), and trifluoromethyl substituents on the nitrogen atom.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's suitability for amplification reactions due to mild conditions (room temperature for step 1, 50°C for step 2), simple operation, and the avoidance of air-sensitive reagents like n-butyllithium.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ebselen Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic route for the next generation of antioxidant therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photoreactor arrays and rigorous QC labs capable of meeting the most stringent purity specifications, guaranteeing that every batch of ebselen intermediate delivered meets the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this innovative technology for your specific drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our reference standards and comprehensive route feasibility assessments for your target molecules.
