Advanced Silver-Promoted Synthesis of Tetrahydroindenone-isoselenazole Derivatives for Pharmaceutical Applications
Introduction to Novel Organoselenium Chemistry
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for safer, more efficient, and environmentally benign processes. A significant breakthrough in this domain is documented in Chinese Patent CN114773293A, which discloses a novel class of tetrahydroindenone-isoselenazole derivatives and their streamlined synthesis. These organoselenium compounds are of immense interest to the global medicinal chemistry community due to their potent biological activities, including cytoprotective, neuroprotective, and anti-inflammatory properties, reminiscent of the well-known drug Ebselen. The patent introduces a robust synthetic strategy that transforms simple o-cyano alkynones into complex heterocyclic scaffolds using elemental selenium as the selenium source.
This technological advancement addresses critical pain points in the production of high-purity pharmaceutical intermediates. By leveraging a silver-promoted cyclization mechanism, the process achieves high atom utilization and operational simplicity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this chemistry is vital. The core structure, as depicted in the general formula, allows for significant structural diversity, enabling the rapid generation of libraries for drug discovery programs targeting neurodegenerative diseases and rheumatoid arthritis.
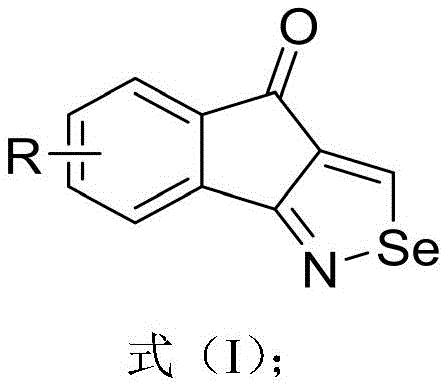
The versatility of this scaffold cannot be overstated. The ability to introduce various substituents such as halogens, alkyl, and alkoxy groups at specific positions on the benzene ring provides a powerful tool for structure-activity relationship (SAR) studies. Furthermore, the synthetic route described avoids the use of hazardous reagents often associated with selenium chemistry, marking a substantial step forward in green chemistry practices within the fine chemical industry. This report delves deep into the mechanistic insights, commercial viability, and scalability of this innovative process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoselenazole derivatives has been fraught with significant challenges that hinder their widespread adoption in commercial drug manufacturing. Traditional methodologies often rely on the use of toxic and unstable selenium reagents, such as selenium tetrachloride or organolithium selenium species, which pose severe safety risks and require specialized handling infrastructure. These reagents are not only hazardous to personnel but also generate substantial amounts of toxic waste, complicating environmental compliance and disposal protocols. Moreover, conventional routes typically involve multi-step sequences with low overall yields, leading to increased production costs and extended lead times.
Another major drawback of existing methods is the harsh reaction conditions frequently required, such as high temperatures or strong acidic/basic environments. These aggressive conditions can lead to the decomposition of sensitive functional groups, limiting the substrate scope and resulting in complex impurity profiles that are difficult to purify. For a procurement manager focused on cost reduction in API manufacturing, these inefficiencies translate directly into higher raw material costs and lower throughput. The reliance on difficult-to-obtain starting materials further exacerbates supply chain vulnerabilities, making it challenging to secure a consistent supply of high-quality intermediates for clinical and commercial needs.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN114773293A offers a paradigm shift through a direct, one-pot cyclization strategy. The core innovation lies in the use of inexpensive and stable elemental selenium powder as the sole selenium source, activated by a synergistic combination of a silver promoter and a cobalt additive. This approach eliminates the need for pre-functionalized selenium reagents, drastically simplifying the raw material supply chain. The reaction proceeds under remarkably mild conditions, typically at 40°C in dimethyl sulfoxide (DMSO), which minimizes energy consumption and thermal stress on the molecular framework.

The generality of this novel approach is demonstrated by its tolerance to a wide array of functional groups. Whether the substrate bears electron-withdrawing halogens like fluorine, chlorine, and bromine, or electron-donating groups like methyl and methoxy, the reaction consistently delivers the desired tetrahydroindenone-isoselenazole derivatives in good to excellent yields. This broad substrate scope is crucial for the commercial scale-up of complex pharmaceutical intermediates, as it allows for the flexible synthesis of diverse analogues without needing to re-optimize conditions for each new variant. The simplicity of the post-treatment process further enhances its appeal, enabling rapid isolation of the product with high purity, thereby reducing downstream processing costs.
Mechanistic Insights into Silver-Promoted Cyclization
The success of this synthesis hinges on the intricate interplay between the silver promoter, specifically silver acetate (AgOAc), and the cobalt additive, cobalt acetate (Co(OAc)2). From a mechanistic perspective, the silver salt acts as a soft Lewis acid that facilitates the activation of the alkyne moiety and the subsequent nucleophilic attack by the selenium species. Elemental selenium, initially inert, is likely activated in situ by the metal salts to form reactive selenolate or selenide intermediates. The silver ion coordinates with the nitrile group or the alkyne, lowering the energy barrier for the cyclization event. This coordination is critical for directing the regioselectivity of the ring closure, ensuring the formation of the isoselenazole ring fused to the indanone system rather than other potential isomers.
The role of the cobalt additive, while less explicitly detailed in terms of elementary steps, appears to be synergistic, potentially assisting in the oxidation state management of selenium or stabilizing key transition states during the ring formation. The choice of solvent plays a pivotal role as well; experimental data indicates that DMSO is superior to other polar aprotic solvents like DMF. For instance, switching from DMSO to DMF resulted in a dramatic drop in yield from 79% to 31% for the unsubstituted substrate. This suggests that DMSO may participate in stabilizing the charged intermediates or facilitating the solubility of elemental selenium, which is often a limiting factor in heterogeneous reactions involving solid selenium powder.
Regarding impurity control, the mild reaction temperature of 40°C is a decisive factor. High-temperature processes often promote side reactions such as polymerization of the alkyne or hydrolysis of the nitrile group. By maintaining a low thermal budget, this process effectively suppresses these degradation pathways, resulting in a cleaner crude reaction mixture. This inherent cleanliness reduces the burden on purification steps, such as column chromatography or recrystallization, which are often the most costly and time-consuming parts of intermediate production. For R&D teams, this means a more predictable impurity profile, simplifying the regulatory filing process for new drug applications.
How to Synthesize Tetrahydroindenone-isoselenazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and reaction environment to maximize efficiency. The patent outlines a precise molar ratio of 2:6:2:1 for the o-cyano alkynone, elemental selenium, silver promoter, and cobalt additive, respectively. While this ratio might seem to use an excess of selenium, it ensures complete conversion of the valuable organic substrate. The reaction is preferably conducted under a nitrogen atmosphere to prevent the oxidation of sensitive intermediates and to maintain the activity of the metal catalysts. Detailed standardized synthesis steps follow below, outlining the exact procedure for achieving optimal results.
- Prepare the reaction mixture by combining o-cyano alkynone substrates with elemental selenium in dimethyl sulfoxide (DMSO) solvent.
- Add the silver promoter (AgOAc) and cobalt additive (Co(OAc)2) to the mixture under a nitrogen atmosphere to initiate the cyclization.
- Maintain the reaction temperature at 40°C for approximately 1 to 3 hours, followed by standard workup to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from traditional multi-step syntheses to this streamlined one-pot process represents a significant opportunity for value creation. The primary driver of cost reduction in manufacturing here is the simplification of the bill of materials. By utilizing elemental selenium, a commodity chemical with stable pricing and abundant global supply, the process avoids the volatility associated with specialized organoselenium reagents. Furthermore, the elimination of multiple isolation and purification steps between synthetic stages drastically reduces solvent consumption, labor hours, and equipment occupancy time. This lean manufacturing approach translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing in the final API market.
Enhanced supply chain reliability is another critical benefit derived from this technology. The starting materials, o-cyano alkynones, are readily accessible and can be synthesized from common precursors, reducing the risk of supply disruptions. The robustness of the reaction conditions—operating at a mild 40°C rather than requiring cryogenic cooling or high-pressure reactors—means that the process can be easily transferred between different manufacturing sites without extensive re-validation. This flexibility is invaluable for maintaining business continuity and scaling production to meet fluctuating market demands. Additionally, the high yields reported across various substituted derivatives ensure that material throughput is maximized, minimizing waste and maximizing the return on investment for every kilogram of raw material purchased.
Scalability and environmental compliance are increasingly becoming deciding factors in vendor selection. This silver-promoted method aligns well with green chemistry principles by improving atom economy and reducing the generation of hazardous waste. The use of DMSO, while requiring proper recovery systems, is a well-understood solvent in the industry with established recycling protocols. The absence of toxic selenium gases or highly corrosive reagents simplifies the engineering controls required for safe operation, lowering the capital expenditure needed for facility upgrades. Consequently, this process supports the commercial scale-up of complex pharmaceutical intermediates while adhering to stringent environmental, health, and safety (EHS) standards, making it a sustainable choice for long-term production partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these tetrahydroindenone-isoselenazole derivatives. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this chemistry into your existing development pipelines or supply networks.
Q: What are the key advantages of this silver-promoted method over traditional isoselenazole synthesis?
A: Unlike traditional methods that often require toxic selenium sources and harsh multi-step conditions, this patent describes a one-pot reaction using inexpensive elemental selenium under mild temperatures (40°C), significantly improving safety and atom economy.
Q: Can this synthesis method accommodate various substituents on the benzene ring?
A: Yes, the method demonstrates excellent universality, successfully tolerating electron-withdrawing groups like halogens (F, Cl, Br) and electron-donating groups like methyl and methoxy, yielding products with isolated yields ranging from 45% to 88%.
Q: Is this process suitable for large-scale commercial manufacturing?
A: The process utilizes readily available raw materials and operates at low temperatures with simple post-treatment, making it highly amenable to commercial scale-up while maintaining stringent purity specifications required for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroindenone-isoselenazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the silver-promoted synthesis described in CN114773293A for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from bench-scale discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle organoselenium chemistry safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee the quality of every batch. We understand that consistency and reliability are paramount in the pharmaceutical supply chain, and we are committed to delivering intermediates that meet the highest global standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to deep process expertise and a robust supply network. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with confidence and efficiency.
