Advanced Manufacturing of Bromhexine Hydrochloride: Technical Breakthroughs and Commercial Scalability
Advanced Manufacturing of Bromhexine Hydrochloride: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with operational efficiency, particularly for established respiratory therapeutics like bromhexine hydrochloride. Patent CN111470983A introduces a transformative methodology for synthesizing this critical active pharmaceutical ingredient (API) intermediate, addressing long-standing challenges in yield optimization and process safety. By implementing a telescoped three-step sequence that avoids the isolation of unstable intermediates, this technology offers a compelling value proposition for manufacturers aiming to enhance their production capabilities. The structural integrity of the final molecule, characterized by its dibromo-substituted benzylamine core, is preserved through mild reaction conditions that minimize degradation pathways. 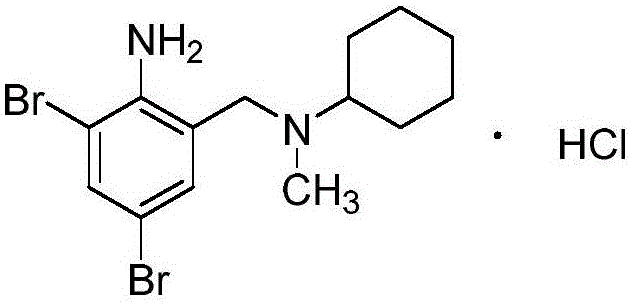 As a reliable pharmaceutical intermediates supplier, understanding these nuanced process improvements is essential for maintaining competitive advantage in the global market. The patent details a shift away from hazardous solvent systems and excessive reagent consumption, aligning modern manufacturing with stricter environmental and safety standards while simultaneously boosting economic output.
As a reliable pharmaceutical intermediates supplier, understanding these nuanced process improvements is essential for maintaining competitive advantage in the global market. The patent details a shift away from hazardous solvent systems and excessive reagent consumption, aligning modern manufacturing with stricter environmental and safety standards while simultaneously boosting economic output.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for bromhexine hydrochloride have historically relied on cumbersome protocols that introduce significant operational risks and inefficiencies into the supply chain. Prior art, such as the methods described in CN201210044011.0, often utilized thionyl chloride in vast excess, serving both as a reactant and a solvent, which necessitated complex removal steps and generated substantial chemical waste. Furthermore, the isolation of the key intermediate, 2,4-dibromo-6-chloromethylaniline, typically required crystallization using petroleum ether, a highly flammable and volatile solvent that poses severe safety hazards in large-scale industrial environments. These legacy processes not only increased the cost reduction in API manufacturing but also compromised the stability of the intermediate, leading to decomposition and lower overall yields. The requirement for rigorous drying steps at elevated temperatures, such as 80°C, further exacerbated the risk of oxidation, introducing impurities that were difficult to remove in downstream processing. Consequently, procurement managers faced higher raw material costs and supply chain heads dealt with extended lead times due to the multi-step isolation and purification requirements inherent in these older technologies.
The Novel Approach
The innovative strategy outlined in CN111470983A fundamentally reengineers the synthesis pathway by adopting a telescoped, one-pot philosophy that streamlines production and enhances safety profiles. A critical advancement is the precise stoichiometric control of thionyl chloride, reducing its usage from a solvent quantity to merely 1.8-2.2 molar equivalents strictly as a reactant. This drastic reduction eliminates the need for energy-intensive distillation to recover excess reagent and removes the necessity for hazardous petroleum ether crystallization steps entirely. By keeping the reaction mixture in solution and transferring the unstable chloromethyl intermediate directly to the next stage, the process mitigates decomposition risks and preserves material integrity. The substitution of safer solvents like tetrahydrofuran and dichloromethane, combined with mild temperature controls between 15-25°C, ensures a stable reaction environment that supports high-purity pharmaceutical intermediates. This approach not only simplifies the operational workflow but also significantly shortens the production cycle, allowing for faster turnaround times and improved responsiveness to market demand without compromising on quality or regulatory compliance standards.
Mechanistic Insights into Telescoped Reduction and Chlorination
The core of this synthetic breakthrough lies in the seamless integration of reduction, chlorination, and amination steps, each optimized to prevent the accumulation of deleterious impurities. The process initiates with the reduction of 2-amino-3,5-dibromobenzaldehyde using sodium borohydride in anhydrous ethanol or isopropanol, where careful pH adjustment to 6-7 facilitates the direct transfer of the resulting alcohol into the chlorination phase. 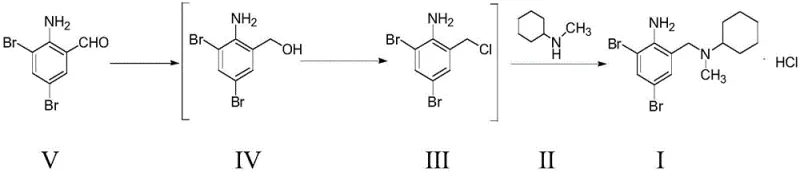 In the subsequent chlorination step, the alcohol reacts with thionyl chloride in tetrahydrofuran, generating the reactive chloromethyl species in situ. The mechanistic advantage here is the avoidance of isolating this thermally sensitive intermediate, which is prone to rapid degradation if exposed to air or heat during drying. Instead, the solution is concentrated and immediately redissolved in dichloromethane for the final amination with N-methylcyclohexylamine. This continuous flow of material minimizes exposure to potential degradation triggers and ensures that the nucleophilic substitution proceeds with high efficiency. The final salt formation using an ethanolic HCl solution at controlled acidic pH levels (4-5) ensures the precipitation of the hydrochloride salt in a highly crystalline form, effectively trapping impurities in the mother liquor and yielding a product with exceptional purity profiles suitable for stringent pharmacopeial standards.
In the subsequent chlorination step, the alcohol reacts with thionyl chloride in tetrahydrofuran, generating the reactive chloromethyl species in situ. The mechanistic advantage here is the avoidance of isolating this thermally sensitive intermediate, which is prone to rapid degradation if exposed to air or heat during drying. Instead, the solution is concentrated and immediately redissolved in dichloromethane for the final amination with N-methylcyclohexylamine. This continuous flow of material minimizes exposure to potential degradation triggers and ensures that the nucleophilic substitution proceeds with high efficiency. The final salt formation using an ethanolic HCl solution at controlled acidic pH levels (4-5) ensures the precipitation of the hydrochloride salt in a highly crystalline form, effectively trapping impurities in the mother liquor and yielding a product with exceptional purity profiles suitable for stringent pharmacopeial standards.
Impurity control is meticulously managed through the selection of specific solvent systems and reaction temperatures that suppress side reactions. For instance, maintaining the reaction temperature between 15-25°C is critical; temperatures exceeding 30°C promote the formation of unknown byproducts, while temperatures below 10°C slow the kinetics unnecessarily and complicate thermal management due to the exothermic nature of the reduction step. The use of sodium borohydride in controlled amounts (0.4-1.0 molar equivalents) prevents the presence of excess reducing agent that could interfere with subsequent chlorination. Furthermore, the decision to forego the drying of the intermediate alcohol at 80°C prevents oxidative degradation, a common source of colored impurities in aniline derivatives. By optimizing the molar ratios of N-methylcyclohexylamine to 3.5-5 equivalents, the process ensures complete conversion of the chloromethyl group while avoiding excessive amine residues that would complicate the final salt formation. This holistic approach to reaction engineering demonstrates a deep understanding of organic synthesis dynamics, ensuring that the commercial scale-up of complex pharmaceutical intermediates remains robust and reproducible.
How to Synthesize Bromhexine Hydrochloride Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized parameters regarding solvent choice, reagent stoichiometry, and thermal control to achieve the reported yields exceeding 62%. The process begins with the dissolution of the starting aldehyde in ethanol, followed by the controlled addition of the reducing agent under cooling to manage exotherms, after which the pH is adjusted to facilitate filtration and transfer. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational procedure. Following these guidelines ensures that the telescoped nature of the reaction is maintained, preventing the premature decomposition of the reactive chloromethyl intermediate and maximizing the throughput of the final API. Operators must pay close attention to the solvent swap from THF to dichloromethane, ensuring complete removal of volatiles to prevent interference with the amination kinetics. The final crystallization from ethanol/water mixtures serves as a powerful polishing step, leveraging solubility differences to exclude trace organic impurities and inorganic salts, resulting in a product that meets the rigorous specifications required for human therapeutic use.
- Reduce 2-amino-3,5-dibromobenzaldehyde with sodium borohydride in ethanol to form the alcohol intermediate.
- React the crude alcohol with 1.8-2.2 molar equivalents of thionyl chloride in THF to generate the chloromethyl derivative.
- Perform amination with N-methylcyclohexylamine followed by salt formation with HCl in ethanol to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond simple yield improvements. The elimination of petroleum ether, a solvent often subject to strict storage regulations and volatility concerns, drastically simplifies facility safety requirements and reduces insurance and compliance overheads. By reducing the consumption of thionyl chloride from a solvent volume to a stoichiometric reactant quantity, the process significantly lowers raw material procurement costs and minimizes the logistical burden of handling corrosive chemicals. The telescoped nature of the synthesis, which removes multiple isolation and drying steps, leads to substantial cost savings by reducing labor hours, energy consumption for heating and vacuum operations, and equipment occupancy time. These efficiencies allow manufacturers to offer more competitive pricing structures while maintaining healthy margins, a critical factor in the price-sensitive generic pharmaceutical market. Additionally, the robustness of the process at ambient temperatures (15-25°C) reduces the dependency on specialized cryogenic cooling infrastructure, further lowering capital expenditure requirements for production facilities.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive and hazardous solvent recovery systems associated with large volumes of thionyl chloride and petroleum ether. By operating with near-stoichiometric reagent quantities and avoiding intermediate isolation, the method reduces waste disposal costs and raw material usage significantly. The higher overall yield of over 62% means less starting material is required to produce the same amount of final product, directly improving the cost of goods sold (COGS). Furthermore, the simplified workflow reduces the man-hours required per batch, allowing existing personnel to manage higher production volumes without proportional increases in labor costs. This lean manufacturing approach ensures that cost reduction in API manufacturing is achieved through fundamental process intensification rather than superficial cuts.
- Enhanced Supply Chain Reliability: The use of common, commercially available solvents like ethanol, THF, and dichloromethane ensures that raw material sourcing remains stable and unaffected by niche supply disruptions. The robustness of the reaction conditions, which tolerate slight variations without significant yield loss, enhances batch-to-batch consistency, a key metric for supply chain reliability. By shortening the production cycle through the removal of purification steps, manufacturers can respond more rapidly to fluctuating market demand, reducing lead time for high-purity pharmaceutical intermediates. The improved stability of the process also minimizes the risk of batch failures, ensuring a continuous and predictable supply of critical respiratory medication ingredients to downstream formulators. This reliability is paramount for maintaining long-term contracts with major pharmaceutical partners who prioritize supply security.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been successfully demonstrated from pilot scales of kilograms to industrial scales of hundreds of kilograms with consistent results. The reduction in hazardous solvent usage aligns with increasingly stringent global environmental regulations, facilitating easier permitting and audit compliance for manufacturing sites. The absence of highly flammable petroleum ether removes a major safety bottleneck, allowing for larger batch sizes in standard reactors without requiring explosion-proof modifications. Waste streams are simpler and less toxic, reducing the environmental footprint and the cost associated with effluent treatment. This alignment with green chemistry principles not only future-proofs the manufacturing asset but also enhances the corporate sustainability profile, a growing priority for international stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and comparative data. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific advantages in yield, purity, and safety that distinguish this method from legacy processes. These insights provide a clear roadmap for technical teams assessing the transition to this more efficient production methodology, ensuring that all potential risks and benefits are fully understood before commitment.
Q: How does the new process improve safety compared to conventional methods?
A: The novel method eliminates the use of petroleum ether for crystallization, removing the risk associated with highly flammable solvents, and reduces thionyl chloride usage significantly.
Q: What is the expected yield and purity of this synthesis route?
A: The process achieves a total yield of over 62% with HPLC purity consistently stabilizing above 99.9%, surpassing traditional methods by more than 5%.
Q: Why is intermediate purification omitted in this protocol?
A: The intermediate 2,4-dibromo-6-chloromethylaniline is extremely unstable; skipping isolation prevents decomposition and simplifies the workflow for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bromhexine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we leverage deep technical expertise to translate innovative patent methodologies like CN111470983A into commercial reality for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this telescoped synthesis are fully realized in large-scale manufacturing. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced HPLC and NMR techniques to verify that every batch of bromhexine hydrochloride meets or exceeds the 99.9% purity benchmark. Our commitment to quality assurance means that we can consistently deliver high-purity pharmaceutical intermediates that support the efficacy and safety of your final drug products. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technically superior and compliant with international regulatory standards.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this more efficient manufacturing process. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to meet your volume and quality needs. Let us collaborate to enhance your supply chain resilience and drive down costs while maintaining the highest standards of pharmaceutical quality.
