Scalable Production of Bromhexine Hydrochloride via Green Chemistry and Process Optimization
Scalable Production of Bromhexine Hydrochloride via Green Chemistry and Process Optimization
The pharmaceutical industry is constantly seeking robust, safe, and economically viable pathways for the production of essential respiratory therapeutics. A pivotal advancement in this domain is detailed in patent CN102617359B, which outlines a novel method for preparing Bromhexine Hydrochloride, a widely used mucolytic agent. This technology represents a significant departure from legacy manufacturing processes that rely on hazardous reagents and extreme conditions. By leveraging advanced green chemistry principles, this synthesis route ensures high product purity while drastically minimizing environmental impact. For global procurement leaders and R&D directors, understanding this technological shift is crucial for securing a reliable API intermediate supplier capable of meeting stringent quality and sustainability standards. The following analysis dissects the technical merits and commercial implications of this optimized synthetic pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Bromhexine Hydrochloride has been plagued by significant safety and efficiency challenges. Traditional Route 1 relies on nitrobenzyl bromide condensation followed by reduction with hydrazine hydrate over Raney Nickel; this introduces severe toxicity risks due to hydrazine and complicates waste treatment with heavy metal catalysts. Traditional Route 2 employs sodium azide, a highly explosive and toxic reagent that poses unacceptable safety liabilities for large-scale operations. Furthermore, Traditional Route 3 utilizes tosyl chloride catalysis at cryogenic temperatures of -70°C, demanding excessive energy consumption and specialized equipment that drives up production costs. These legacy methods often suffer from low yields, complex purification steps, and substantial environmental burdens, rendering them increasingly obsolete in a regulatory landscape focused on green manufacturing and operator safety.
The Novel Approach
The patented methodology introduces a streamlined three-step sequence that effectively circumvents the pitfalls of prior art. The process initiates with the selective reduction of 2-amino-3,5-dibromo benzaldehyde using sodium borohydride, a mild and safe reducing agent, to yield the corresponding benzyl alcohol intermediate. This step avoids the use of high-pressure hydrogenation or toxic hydrides.
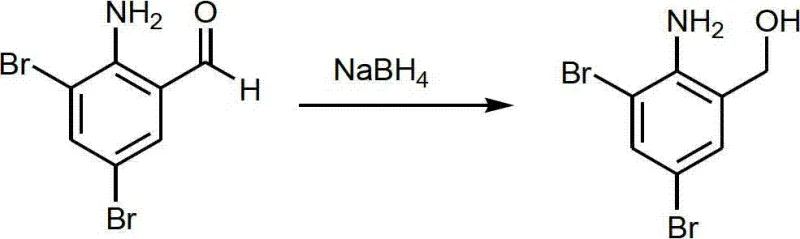
Subsequently, the alcohol undergoes chlorination using thionyl chloride, which uniquely serves a dual function as both the chlorinating reagent and the reaction solvent, thereby eliminating the need for additional volatile organic solvents.
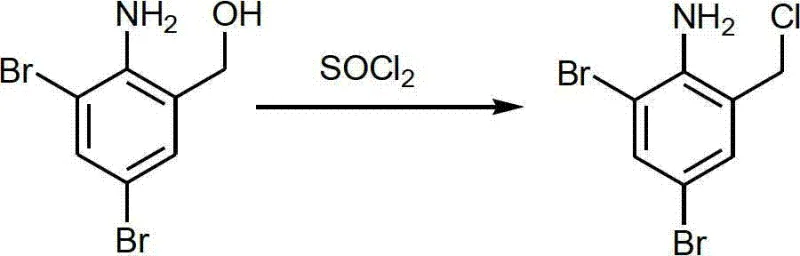
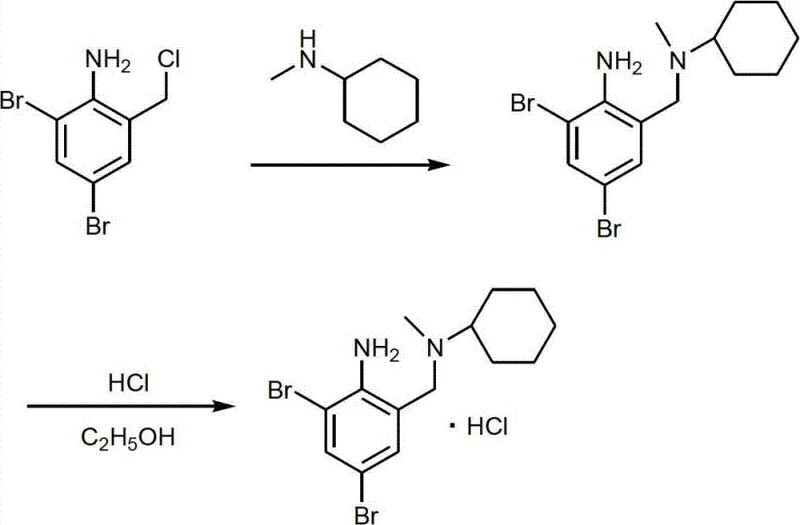
The final stage involves a nucleophilic substitution with N-methylcyclohexylamine, which also acts as an acid scavenger, followed by direct salification. This integrated approach not only enhances reaction kinetics but also simplifies downstream processing, offering a compelling solution for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into the Multi-Step Synthesis Strategy
A deep dive into the reaction mechanisms reveals why this pathway achieves superior impurity control and yield stability. In the initial reduction phase, sodium borohydride selectively targets the aldehyde carbonyl group without affecting the aromatic amine or bromine substituents, ensuring high chemoselectivity. The subsequent chlorination mechanism leverages the high reactivity of the benzylic hydroxyl group, allowing the reaction to proceed efficiently at ambient temperatures. By using excess thionyl chloride as the medium, the equilibrium is driven towards product formation, and the byproduct gases (HCl and SO2) are easily managed with basic scrubbing systems, preventing acid accumulation that could degrade the sensitive aniline moiety.
The critical amination step involves the displacement of the chloride by N-methylcyclohexylamine. Mechanistically, the amine serves a dual purpose: it acts as the nucleophile to form the C-N bond and simultaneously neutralizes the generated HCl, preventing the protonation of the product which would halt the reaction. This self-buffering capacity is a key innovation that stabilizes the reaction environment.
Structural integrity is rigorously maintained throughout, as confirmed by comprehensive spectroscopic analysis where carbon atoms are precisely assigned, such as the distinct shifts observed for the cyclohexyl ring carbons versus the aromatic core.
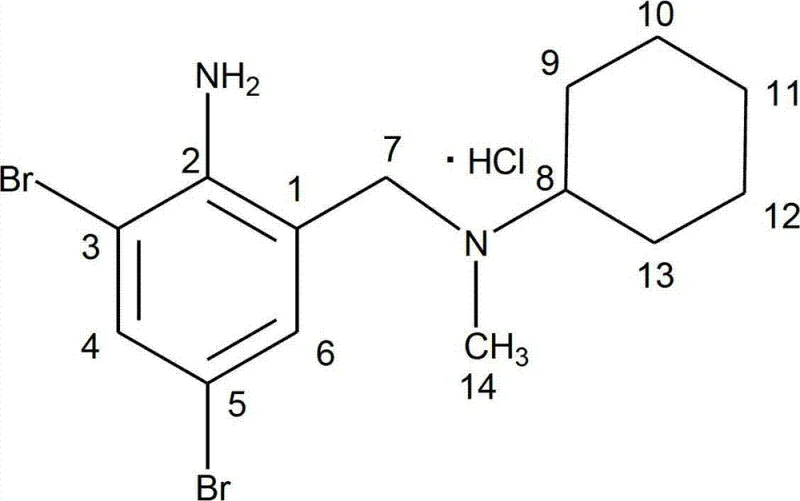
This level of mechanistic control ensures that side reactions, such as over-alkylation or polymerization, are minimized, resulting in a crude product that requires less aggressive purification, thereby preserving overall yield.
How to Synthesize Bromhexine Hydrochloride Efficiently
Implementing this synthesis requires precise control over stoichiometry and thermal conditions to maximize the benefits of the patented route. The process is designed to be operationally simple, avoiding the need for specialized cryogenic infrastructure or high-pressure vessels. Detailed standard operating procedures focus on the sequential addition of reagents to manage exotherms and ensure complete conversion at each stage. For technical teams looking to adopt this methodology, the following guide outlines the critical operational parameters derived from the patent examples.
- Reduce 2-amino-3,5-dibromo benzaldehyde using sodium borohydride in ethanol to form the benzyl alcohol intermediate.
- Convert the alcohol to the chloromethyl derivative using thionyl chloride acting as both reagent and solvent.
- Perform nucleophilic substitution with N-methylcyclohexylamine followed by salification with HCl and recrystallization in methanol.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this manufacturing route offers transformative advantages that directly impact the bottom line and operational resilience. The elimination of hazardous reagents like hydrazine and sodium azide removes the need for specialized storage, handling protocols, and expensive disposal services, leading to substantial cost savings in EHS (Environment, Health, and Safety) compliance. Furthermore, the ability to recover and reuse excess thionyl chloride and N-methylcyclohexylamine significantly reduces raw material consumption, enhancing the overall atom economy of the process.
- Cost Reduction in Manufacturing: The process operates under mild thermal conditions, typically between room temperature and 65°C, which eliminates the massive energy costs associated with maintaining cryogenic temperatures of -70°C required by older methods. Additionally, the dual role of reagents as solvents reduces the volume of waste solvent that needs to be distilled and treated, further driving down utility and waste management expenses.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials like 2-amino-3,5-dibromo benzaldehyde and avoiding reagents with restricted supply chains (such as sodium azide), manufacturers can ensure greater continuity of supply. The robustness of the reaction conditions means that production is less susceptible to interruptions caused by equipment failure or strict environmental shutdowns, providing a more dependable source for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The simplified work-up procedures, including direct salification and efficient recrystallization, facilitate seamless scale-up from pilot batches to multi-ton commercial production. The reduction in toxic waste streams aligns with increasingly stringent global environmental regulations, mitigating the risk of regulatory fines and ensuring long-term operational viability for the facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on feasibility and quality outcomes.
Q: How does this new method improve safety compared to traditional Bromhexine synthesis?
A: The patented process eliminates the use of highly toxic hydrazine hydrate and explosive sodium azide found in conventional routes, significantly reducing workplace hazards and environmental risks.
Q: What are the key advantages regarding solvent usage in this synthesis?
A: The process utilizes thionyl chloride and N-methylcyclohexylamine as both reactants and solvents, which simplifies the workflow, reduces solvent waste, and allows for efficient recovery and reuse of excess reagents.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the method operates under mild conditions (room temperature to 65°C) without requiring extreme cryogenic cooling (-70°C), making it energy-efficient and highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bromhexine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is vital for the future of the pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patented process are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Bromhexine Hydrochloride meets the highest international standards for potency and impurity profiles.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic advantages specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines.
