Scalable Manufacturing of ((2R,7aS)-2-fluorohexahydro-1H-pyrrolazin-7a-yl)methanol for Oncology APIs
Introduction to Advanced Oncology Intermediate Synthesis
The rapid advancement of targeted cancer therapies has placed immense pressure on the supply chain for complex small molecule intermediates, particularly those serving as key building blocks for KRAS G12D inhibitors. Patent CN115894503A discloses a groundbreaking preparation method for an azacyclopentane derivative, specifically ((2R,7aS)-2-fluorohexahydro-1H-pyrrolazin-7a-yl)methanol, which serves as a critical chiral scaffold in the design of next-generation oncology drugs like MRTX1133. This compound features a fused bicyclic system with precise stereochemistry that is notoriously difficult to establish efficiently using traditional methods. The structural complexity of this molecule requires a synthesis strategy that balances high stereoselectivity with operational simplicity to meet the rigorous demands of pharmaceutical manufacturing.
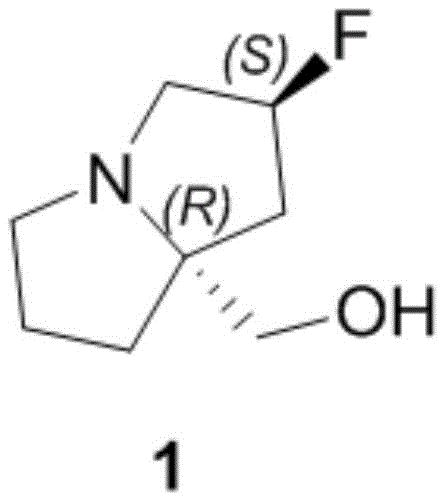
For R&D directors and process chemists, the availability of a robust, scalable route to this intermediate is paramount. The disclosed technology addresses the historical bottlenecks associated with constructing the fluorinated pyrrolizine core. By leveraging abundant natural amino acids as starting materials, this innovation shifts the paradigm from reliance on hazardous, low-yielding transformations to a more predictable, linear synthesis. This report analyzes the technical merits of this novel pathway, highlighting its potential to become the industry standard for producing high-purity pharmaceutical intermediates required for clinical and commercial supply of KRAS inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing similar fluorinated bicyclic scaffolds often rely on aggressive and operationally challenging reagents that pose significant risks at scale. For instance, earlier patents such as WO2022031678A1 describe routes involving ozonolysis followed by reductive workups with dimethyl sulfide, alongside the use of DAST (Diethylaminosulfur trifluoride) and lithium aluminum hydride in multiple steps. These processes are fraught with safety hazards; ozone generation requires specialized equipment and strict safety protocols to prevent explosive incidents, while DAST is thermally unstable and can decompose violently upon heating. Furthermore, the use of stoichiometric amounts of heavy metal hydrides generates substantial waste streams, complicating environmental compliance and increasing disposal costs.
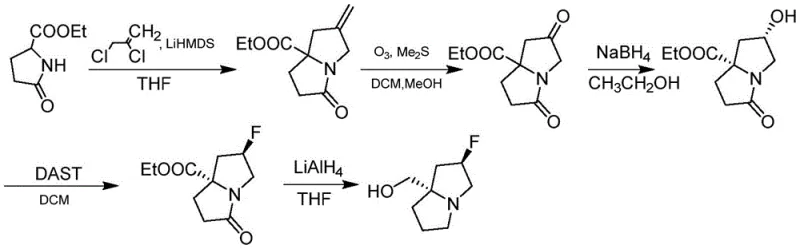
From a supply chain perspective, these conventional routes are fragile. The reliance on multiple protection-deprotection sequences and harsh oxidation states often leads to cumulative yield losses, driving up the cost of goods sold (COGS). The impurity profiles generated by ozonolysis can be difficult to purge, requiring extensive chromatographic purification which is not feasible for multi-ton production. Consequently, procurement managers face volatility in pricing and lead times when sourcing intermediates produced via these legacy methods, as manufacturers struggle to maintain consistent quality and throughput without incurring prohibitive safety overheads.
The Novel Approach
In stark contrast, the methodology outlined in CN115894503A presents a streamlined, five-to-six-step sequence originating from L-proline hydrochloride, a cheap and readily available chiral pool material. This approach elegantly bypasses the need for ozonolysis entirely, replacing it with a mild alkylation and a highly selective intramolecular cyclization. The strategic placement of the fluorination and reduction steps towards the end of the synthesis ensures that high-value intermediates are not subjected to unnecessary processing, thereby maximizing overall material efficiency. The route capitalizes on the inherent chirality of proline to set the initial stereocenters, reducing the need for expensive chiral resolution or asymmetric catalysis later in the sequence.
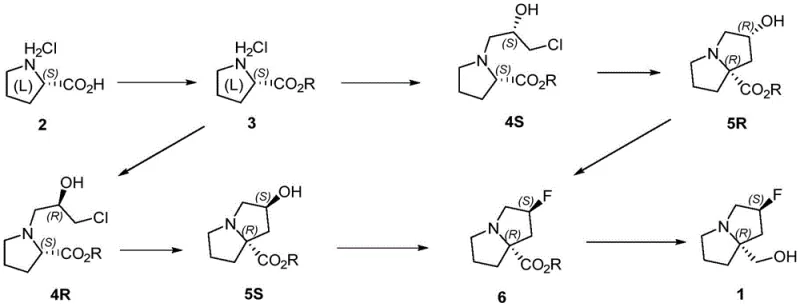
This novel pathway offers a distinct competitive advantage by simplifying the operational landscape. The reactions utilize common organic solvents like methanol, THF, and dichloromethane, and the reagents, such as epichlorohydrin and LiHMDS, are commercially available in bulk quantities. By avoiding the extreme hazards associated with ozone and unstable fluorinating agents in the early stages, the process becomes inherently safer and more amenable to continuous manufacturing or large-batch processing. This shift not only enhances the reliability of the supply chain but also aligns with modern green chemistry principles by reducing the E-factor and minimizing the use of toxic reagents.
Mechanistic Insights into LiHMDS-Mediated Cyclization and Fluorination
The cornerstone of this synthetic strategy is the stereoselective construction of the bicyclic pyrrolizine core, achieved through a base-mediated intramolecular cyclization. In this critical step, the chlorohydrin intermediate (Formula 4S or 4R) is treated with a strong non-nucleophilic base, such as lithium hexamethyldisilazide (LiHMDS) or lithium diisopropylamide (LDA), at cryogenic temperatures ranging from -78°C to -50°C. The low temperature is essential to kinetically control the deprotonation of the alpha-position relative to the ester, generating a specific enolate geometry that favors intramolecular SN2 displacement of the chloride. This mechanism ensures the inversion of configuration required to establish the trans-fused ring system with the correct (2R,7aS) or (2S,7aR) stereochemistry, preventing the formation of cis-fused diastereomers which would be difficult to separate.
Following the ring closure, the introduction of the fluorine atom is executed with high precision. The patent describes two viable options for this transformation: direct fluorination using DAST or a two-step activation-fluorination sequence using trifluoromethanesulfonic anhydride followed by an HF-pyridine complex. The latter option is particularly noteworthy for process safety, as it avoids handling neat DAST on a large scale. The hydroxyl group at the C2 position is activated as a leaving group (triflate or similar) and subsequently displaced by fluoride ion. This nucleophilic substitution proceeds with inversion of configuration, preserving the stereochemical integrity established during the cyclization. The careful selection of fluorinating reagents allows process chemists to balance reactivity with safety, ensuring that the final API intermediate meets stringent purity specifications regarding residual metals and halogenated impurities.
How to Synthesize ((2R,7aS)-2-fluorohexahydro-1H-pyrrolazin-7a-yl)methanol Efficiently
The execution of this synthesis requires strict adherence to temperature controls and anhydrous conditions during the cyclization and fluorination phases to ensure optimal yield and stereochemical purity. The initial esterification and alkylation steps are robust and can be performed with standard laboratory equipment, but the subsequent lithiation step demands high-quality dry solvents and inert atmosphere techniques. Detailed standard operating procedures for each transformation, including specific quenching protocols and workup strategies to remove lithium salts and amine byproducts, are critical for successful technology transfer. For a comprehensive guide on the specific molar ratios, addition rates, and purification methods validated in the patent examples, please refer to the standardized protocol below.
- Esterify L-proline hydrochloride with thionyl chloride in methanol to obtain the methyl ester intermediate.
- React the ester with (S)-epichlorohydrin using a base like DBU or triethylamine to form the chlorohydrin side chain.
- Perform intramolecular cyclization using LiHMDS at -78°C to construct the pyrrolizine core with high stereoselectivity.
- Conduct fluorination using DAST or trifluoromethanesulfonic anhydride-HF pyridine complex to introduce the fluorine atom.
- Reduce the ester group using LiAlH4 or borane to yield the final hydroxymethyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this proline-based route represents a significant opportunity to optimize cost structures and mitigate supply risks. The primary driver of cost reduction lies in the raw material selection; L-proline hydrochloride is a commodity chemical produced in massive volumes for the food and feed industries, ensuring a stable and low-cost supply baseline compared to specialized chiral building blocks. Furthermore, by deferring the expensive fluorination and reduction steps to the end of the synthesis, the process minimizes the value-at-risk. If a batch fails in the early stages, the financial loss is negligible compared to failing a batch after multiple high-cost transformations, a common issue in linear syntheses of complex heterocycles.
- Cost Reduction in Manufacturing: The elimination of ozonolysis equipment and the associated safety infrastructure drastically lowers capital expenditure (CAPEX) for manufacturing facilities. Additionally, the avoidance of stoichiometric heavy metal reagents in favor of catalytic or recyclable base-mediated steps reduces waste disposal costs. The simplified workup procedures, often involving straightforward extractions and crystallizations rather than preparative HPLC, further drive down operational expenses, resulting in a more competitive price point for the final intermediate.
- Enhanced Supply Chain Reliability: Reliance on globally available commodities like proline and epichlorohydrin decouples the supply chain from the bottlenecks of niche reagent suppliers. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, leading to higher first-pass yields and more predictable delivery schedules. This stability is crucial for maintaining continuity in the production of life-saving oncology medications, where supply interruptions can have severe clinical consequences.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that are compatible with large-scale reactor systems. The reduction in hazardous waste generation, particularly the removal of sulfur-containing byproducts from DMS quenching and ozone off-gassing, simplifies environmental permitting and compliance. This 'green' profile not only aligns with corporate sustainability goals but also future-proofs the manufacturing site against tightening environmental regulations, ensuring long-term viability of the production asset.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology compares to existing market solutions. Understanding these nuances is essential for making informed decisions about process adoption and vendor qualification.
Q: What are the key advantages of the proline-based route over ozonolysis methods?
A: The proline-based route eliminates the need for hazardous ozonolysis and dimethyl sulfide quenching, significantly improving process safety and reducing equipment costs associated with ozone generation and scrubbing systems.
Q: How is stereochemical control achieved in the cyclization step?
A: Stereocontrol is maintained through the use of chiral starting materials (L-proline) and specific reaction conditions during the LiHMDS-mediated cyclization at low temperatures (-78°C), ensuring the formation of the desired (2R,7aS) configuration.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the route utilizes cheap, commodity raw materials like L-proline hydrochloride and avoids exotic reagents in the early stages, making it highly scalable and cost-effective for multi-kilogram to ton-level manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ((2R,7aS)-2-fluorohexahydro-1H-pyrrolazin-7a-yl)methanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation cancer therapies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to global supply. Our state-of-the-art facilities are equipped to handle the specific safety requirements of fluorination chemistry and cryogenic reactions, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for API synthesis.
We invite you to collaborate with us to leverage this advanced proline-based technology for your KRAS inhibitor programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can accelerate your timeline and reduce your overall cost of goods.
