Advanced Metal-Free Synthesis of Benzocoumarins for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable pathways to access high-value heterocyclic scaffolds. A pivotal advancement in this domain is detailed in Chinese Patent CN110862368B, which discloses a novel photocatalytic oxidation synthesis method for benzocoumarin compounds. This technology represents a paradigm shift from traditional thermal catalysis to a mild, visible-light-driven process that utilizes molecular oxygen as the terminal oxidant. By leveraging a dual catalytic system comprising 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) and tert-butyl nitrite (TBN), this method achieves efficient C-H functionalization and subsequent lactonization under ambient conditions. For R&D directors and procurement strategists, this patent offers a compelling solution to the persistent challenges of metal residue control and energy consumption in the manufacturing of complex pharmaceutical intermediates and OLED materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzocoumarins via oxidative lactonization of 2-arylbenzoic acids has relied heavily on transition metal catalysis. Prominent literature precedents describe systems utilizing Palladium(II) complexes, Copper(II) salts, or Silver(I) nitrates, often in conjunction with stoichiometric amounts of harsh chemical oxidants such as hypervalent iodine reagents or persulfates. While effective in small-scale laboratory settings, these conventional methodologies present severe drawbacks for industrial application. The primary concern is the inevitable introduction of toxic heavy metal residues into the final product, necessitating costly and time-consuming purification steps involving specialized scavenger resins to meet stringent regulatory limits for active pharmaceutical ingredients (APIs). Furthermore, the reliance on stoichiometric oxidants generates substantial quantities of hazardous waste, increasing the environmental footprint and disposal costs associated with the manufacturing process.
The Novel Approach
In stark contrast, the technology outlined in CN110862368B introduces a metal-free photocatalytic strategy that elegantly circumvents these limitations. By employing organic small-molecule catalysts (DDQ and TBN) activated by blue LED irradiation, the reaction proceeds efficiently at room temperature and normal pressure. This approach replaces expensive and toxic transition metals with readily available organic reagents and utilizes molecular oxygen—essentially free and abundant from the air—as the sole oxidant. The result is a cleaner reaction profile that inherently avoids heavy metal contamination, thereby simplifying the downstream workup and significantly enhancing the overall sustainability of the production line. This transition from thermal metal catalysis to photo-organocatalysis marks a significant upgrade in process safety and economic viability for large-scale operations.
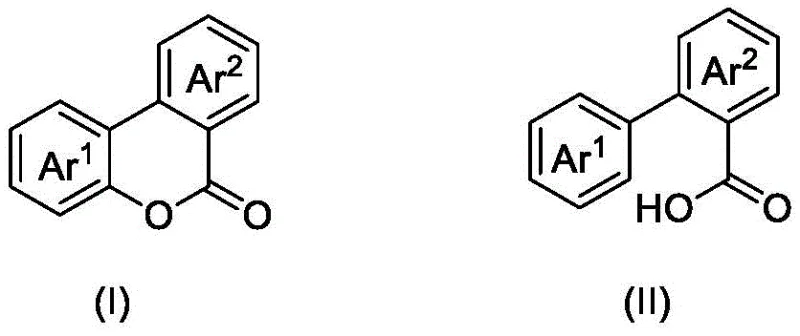
Mechanistic Insights into DDQ/TBN-Mediated Photocatalytic Lactonization
The core of this innovative synthesis lies in the synergistic interaction between the photocatalyst system and the substrate under visible light irradiation. Upon exposure to blue light, the tert-butyl nitrite (TBN) undergoes homolytic cleavage to generate reactive radical species, which facilitate the activation of the substrate. Concurrently, DDQ acts as an electron acceptor and hydrogen abstractor, promoting the oxidative cyclization process. This radical-mediated pathway allows for the direct formation of the lactone ring through C-O bond construction without the need for pre-functionalized starting materials or harsh acidic conditions. The mildness of this mechanism ensures that sensitive functional groups on the aromatic rings, such as halogens or alkoxy substituents, remain intact, providing excellent chemoselectivity that is often difficult to achieve with aggressive thermal oxidants.
From an impurity control perspective, the absence of transition metals is a decisive advantage for pharmaceutical manufacturing. In traditional Pd-catalyzed routes, trace metal impurities can catalyze unwanted side reactions during storage or formulation, potentially compromising drug stability. The organic nature of the DDQ/TBN system ensures that the resulting benzocoumarin products are free from elemental impurities like Palladium, Copper, or Nickel, which are strictly regulated under ICH Q3D guidelines. This intrinsic purity profile reduces the burden on quality control laboratories and minimizes the risk of batch rejection due to out-of-specification metal content, thereby streamlining the regulatory approval pathway for new drug candidates utilizing this scaffold.
How to Synthesize Benzocoumarins Efficiently
The operational simplicity of this photocatalytic method makes it highly attractive for process chemists aiming to translate laboratory discoveries into commercial reality. The procedure involves dissolving the 2-arylbenzoic acid substrate in a suitable organic solvent, such as 1,2-dichloroethane, and adding the catalytic amounts of DDQ and TBN. The reaction vessel is then purged with oxygen to establish an oxidative atmosphere and irradiated with a standard blue LED lamp. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining 2-arylbenzoic acid substrate, DDQ, and TBN in 1,2-dichloroethane solvent.
- Replace the atmosphere in the reaction vessel with oxygen gas to serve as the terminal oxidant.
- Irradiate the mixture with an 18W blue LED lamp at room temperature for 7 to 48 hours, followed by solvent evaporation and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic benefits beyond mere technical novelty. The elimination of precious metal catalysts directly impacts the bill of materials, removing the volatility associated with the pricing of Palladium and other rare earth elements. Moreover, the use of oxygen as a reagent drastically reduces the cost of goods sold (COGS) compared to purchasing expensive stoichiometric oxidants. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a leaner and more cost-effective manufacturing process that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts eliminates the need for expensive metal scavengers and complex filtration processes, leading to substantial cost savings in downstream processing. Additionally, the use of molecular oxygen as a terminal oxidant is significantly more economical than purchasing proprietary chemical oxidants, effectively lowering the raw material expenditure per kilogram of produced benzocoumarin intermediate.
- Enhanced Supply Chain Reliability: The starting materials, specifically various substituted 2-arylbenzoic acids, are commercially available and structurally diverse, ensuring a robust supply chain that is not dependent on single-source specialty catalysts. The stability of the organic catalyst system (DDQ/TBN) further guarantees consistent batch-to-batch reproducibility, minimizing the risk of production delays caused by catalyst degradation or supply shortages common with sensitive organometallic complexes.
- Scalability and Environmental Compliance: Operating at room temperature and normal pressure significantly reduces the safety risks associated with high-pressure reactors, facilitating easier scale-up from pilot plant to multi-ton commercial production. The generation of minimal hazardous waste and the avoidance of heavy metals simplify wastewater treatment and disposal, ensuring compliance with increasingly stringent environmental regulations and reducing the overall ecological footprint of the facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: Why is the metal-free photocatalytic method superior to traditional Pd-catalyzed lactonization?
A: Traditional methods often utilize expensive transition metals like Palladium or Copper, which pose significant challenges regarding residual metal limits in pharmaceutical products (ICH Q3D guidelines). The disclosed photocatalytic method uses organic catalysts (DDQ/TBN) and oxygen, completely eliminating the risk of heavy metal contamination and reducing downstream purification costs.
Q: What are the optimal reaction conditions for scaling up this benzocoumarin synthesis?
A: The patent specifies using 1,2-dichloroethane as the preferred solvent with an 18W blue LED light source at room temperature. The reaction tolerates normal pressure oxygen atmospheres, making it safer and more energy-efficient than high-pressure thermal oxidation processes typically required for similar transformations.
Q: Can this synthesis method accommodate diverse functional groups on the aromatic ring?
A: Yes, the method demonstrates excellent substrate scope. It successfully converts 2-arylbenzoic acids substituted with halogens (F, Cl, Br), trifluoromethyl groups, alkyl chains (methyl, tert-butyl), and alkoxy groups into the corresponding benzocoumarins with high yields, ensuring versatility for medicinal chemistry campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzocoumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free photocatalytic technologies in the synthesis of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110862368B are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzocoumarin delivered meets the highest standards required for global pharmaceutical applications.
We invite forward-thinking R&D and procurement teams to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can optimize your supply chain and accelerate your time to market.
