Advanced One-Pot Synthesis of 1,4-Dichloroisoquinoline Derivatives for Commercial Scale-Up
Introduction to Next-Generation Isoquinoline Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and environmentally sustainable synthetic routes. A pivotal advancement in this domain is detailed in Chinese Patent CN101092395A, which discloses a novel "one-pot" methodology for the preparation of 1,4-dichloroisoquinoline derivatives. These heterocyclic compounds serve as critical scaffolds in the development of bioactive alkaloids with significant applications in medicine and agrochemicals. The traditional reliance on multi-step condensation and harsh dehydrogenation protocols has long posed challenges regarding atom economy and operational safety. This patent introduces a transformative approach utilizing alpha-azidomethyl aryl ketones and an in-situ generated Vilsmeier reagent, effectively streamlining the synthetic pathway. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic and commercial implications of this technology is essential for optimizing supply chains and reducing manufacturing costs in API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovation described in CN101092395A, the synthesis of isoquinoline skeletons predominantly relied on classical condensation reactions such as the Bischler-Napieralski, Pictet-Spengler, or Pomeranz-Fritsch reactions. Specifically, the preparation of 1,4-dichloroisoquinoline derivatives often involved a cumbersome two-step sequence starting from cinnamyl azides. As illustrated in the reaction scheme below, this conventional route necessitates a high-temperature cyclization followed by a chlorination step using phosphorus pentachloride (PCl5).
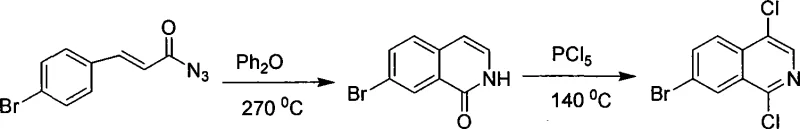
The reliance on phosphorus pentachloride presents severe drawbacks, including the generation of substantial phosphorus-containing waste, which complicates three-waste treatment and violates modern green chemistry principles. Furthermore, the reaction conditions are often severe, requiring elevated temperatures that can degrade sensitive functional groups and lead to lower overall yields. The multi-step nature of this process inherently increases production time, energy consumption, and the risk of impurity accumulation, making it less attractive for the commercial scale-up of complex pharmaceutical intermediates where purity profiles are strictly regulated.
The Novel Approach
In stark contrast, the novel methodology employs a direct cyclization strategy using alpha-azidomethyl aryl ketones as the starting material. By reacting these ketones with a Vilsmeier reagent generated in situ from bis(trichloromethyl) carbonate (BTC) and N,N-dimethylformamide (DMF), the target 1,4-dichloroisoquinoline derivatives are obtained in a single pot. This approach drastically simplifies the operational workflow, eliminating the isolation of intermediate isoquinolones and the need for separate chlorination steps. The reaction proceeds under significantly milder conditions, typically between 0°C and 100°C, with preferred ranges of 40°C to 80°C, ensuring better thermal control and safety.
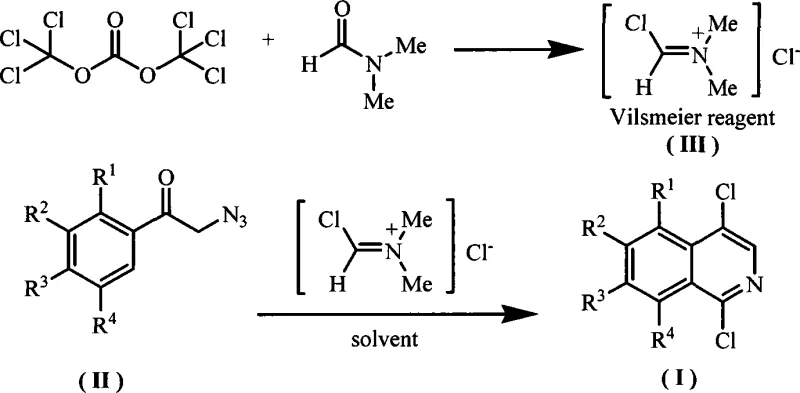
The versatility of this method is demonstrated by its compatibility with various substituents (R1-R4), including alkyl, alkoxy, and aryl groups, allowing for the synthesis of a diverse library of derivatives such as 1,4-dichloro-6-methoxyisoquinoline and 1,4-dichloro-benzo[h]isoquinoline. The streamlined nature of this one-pot synthesis not only enhances atom economy but also facilitates easier downstream processing, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing. By replacing hazardous reagents with more manageable alternatives, this technology aligns perfectly with the sustainability goals of modern chemical enterprises.
Mechanistic Insights into Vilsmeier-Mediated Cyclization
The core of this synthetic breakthrough lies in the effective generation and utilization of the Vilsmeier reagent. As depicted in the structural diagrams, the reaction initiates with the formation of the active chloroiminium species [Cl-CH=N+(Me)2]Cl- from the interaction of BTC and DMF. This electrophilic reagent then engages with the electron-rich alpha-azidomethyl aryl ketone substrate. The mechanism likely involves an initial activation of the carbonyl oxygen or direct electrophilic attack on the aromatic ring, followed by an intramolecular cyclization driven by the azide group acting as a nitrogen source. The loss of nitrogen gas serves as a thermodynamic driving force, pushing the equilibrium towards the formation of the isoquinoline ring system.
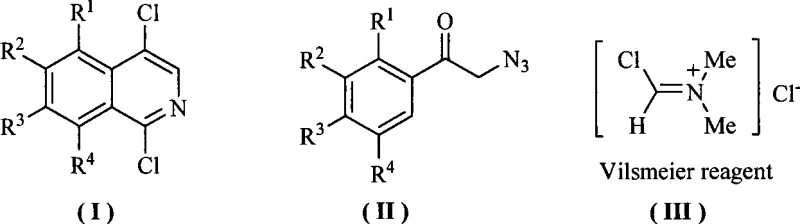
From an impurity control perspective, the mild reaction conditions play a pivotal role. Traditional high-temperature methods often promote polymerization or decomposition of the azide functionality, leading to complex impurity profiles that are difficult to purge. In this novel protocol, the controlled addition of the Vilsmeier reagent and the moderate temperature range (40°C–80°C) minimize side reactions. The use of solvents like 1,2-dichloroethane or acetonitrile further optimizes the solubility of intermediates, ensuring a homogeneous reaction environment. This results in crude products with higher purity, often exceeding 99.0% HPLC purity after simple recrystallization, thereby reducing the burden on purification units and enhancing the overall efficiency of high-purity pharmaceutical intermediate production.
How to Synthesize 1,4-Dichloroisoquinoline Derivatives Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction parameters to maximize yield and safety. The patent outlines a robust procedure where the molar ratio of alpha-azidomethyl aryl ketone to BTC to DMF is carefully balanced, typically ranging from 1:0.67–3.0:2.0–9.0. The process begins with the pre-formation of the Vilsmeier reagent at room temperature, followed by the addition of the ketone substrate. Reaction monitoring via TLC ensures complete conversion before quenching with saturated sodium bicarbonate. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Prepare the Vilsmeier reagent by dissolving bis(trichloromethyl) carbonate (BTC) and N,N-dimethylformamide (DMF) in an organic solvent such as 1,2-dichloroethane and stirring at room temperature for 0.5 to 2 hours.
- Add the alpha-azidomethyl aryl ketone substrate to the reaction mixture and maintain the temperature between 40°C and 80°C for 4 to 8 hours to facilitate cyclization.
- Quench the reaction with saturated sodium bicarbonate solution, separate the organic layer, dry over anhydrous sodium sulfate, and purify the crude product via recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this Vilsmeier-based protocol offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Bis(trichloromethyl) carbonate (BTC) and DMF are commodity chemicals with stable global supply lines, unlike specialized phosphorus reagents which may face regulatory scrutiny or availability fluctuations. This shift ensures greater supply continuity and reduces the risk of production stoppages due to raw material shortages, a critical factor for maintaining reliable delivery schedules to downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of the toxic phosphorus pentachloride step removes the need for expensive corrosion-resistant equipment and specialized waste disposal services associated with phosphorus byproducts. Furthermore, the one-pot nature of the reaction reduces solvent usage, energy consumption for heating and cooling cycles, and labor hours required for intermediate isolations. These cumulative efficiencies translate into substantial cost savings per kilogram of product, enhancing the margin potential for bulk buyers without compromising on quality standards.
- Enhanced Supply Chain Reliability: By adopting a synthesis route that operates under milder conditions (40°C–80°C versus high-temperature cyclization), the process becomes more robust and less prone to batch-to-batch variability caused by thermal excursions. The use of common organic solvents like 1,2-dichloroethane or acetonitrile simplifies solvent recovery and recycling protocols. This operational stability allows for more accurate production planning and shorter lead times for high-purity pharmaceutical intermediates, enabling partners to respond more agilely to market demands.
- Scalability and Environmental Compliance: The atom economy of this reaction is superior to traditional methods, generating significantly less hazardous waste. The absence of heavy metal catalysts or phosphorus sludge simplifies environmental compliance and reduces the carbon footprint of the manufacturing process. This aligns with increasingly stringent global environmental regulations, future-proofing the supply chain against potential regulatory bans on older, dirtier technologies. The process has been demonstrated to scale effectively, supporting the commercial scale-up of complex heterocyclic intermediates from pilot batches to multi-ton production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the process capabilities and limitations for potential adopters.
Q: What are the safety advantages of using BTC and DMF over phosphorus pentachloride?
A: The use of bis(trichloromethyl) carbonate (BTC) and DMF eliminates the need for highly toxic and corrosive phosphorus pentachloride (PCl5), significantly reducing hazardous waste generation and improving operator safety during the chlorination and cyclization steps.
Q: What yields can be expected from this one-pot synthesis method?
A: According to patent data, the reaction yields typically range from 30% to 80%, with specific optimized examples achieving yields as high as 83% for benzo[h]isoquinoline derivatives under mild thermal conditions.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the process utilizes readily available raw materials and avoids extreme high-temperature conditions, making it highly scalable for commercial production while maintaining high purity standards required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Dichloroisoquinoline Supplier
The technological potential of the Vilsmeier-mediated cyclization described in CN101092395A represents a significant leap forward for the production of isoquinoline-based scaffolds. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 1,4-dichloroisoquinoline derivatives meets the exacting standards required for pharmaceutical and agrochemical applications.
We invite global partners to leverage our technical expertise to optimize their supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and commercial reliability.
