Advanced One-Pot Synthesis of 1,4-Dichloroisoquinoline Derivatives for Commercial Scale-Up
Advanced One-Pot Synthesis of 1,4-Dichloroisoquinoline Derivatives for Commercial Scale-Up
The landscape of heterocyclic chemistry is constantly evolving, driven by the urgent need for more sustainable and efficient manufacturing processes for bioactive scaffolds. A pivotal advancement in this domain is documented in patent CN100534986C, which discloses a novel one-pot methodology for synthesizing 1,4-dichloroisoquinoline derivatives. These compounds serve as critical building blocks in the development of alkaloids with significant physiological activities, finding extensive applications in both pharmaceutical and agrochemical sectors. The traditional approaches to constructing the isoquinoline skeleton often involve cumbersome multi-step sequences that suffer from low atom economy and harsh reaction conditions. In contrast, the technique outlined in this intellectual property utilizes alpha-azidomethyl aryl ketones as starting materials, reacting them with an in-situ generated Vilsmeier reagent. This strategic shift not only streamlines the synthetic route but also addresses critical pain points regarding safety and environmental impact, making it a highly attractive option for reliable pharmaceutical intermediates suppliers seeking to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovation described in the patent data, the synthesis of 1,4-dichloroisoquinoline derivatives was predominantly achieved through a pathway starting from cinnamoyl azide. This legacy route necessitates a two-step high-temperature cyclization process that poses significant engineering challenges. As illustrated in the reaction scheme below, the initial cyclization requires extreme thermal energy, often reaching temperatures as high as 270°C, which demands specialized equipment and rigorous safety protocols to prevent thermal runaway. Furthermore, the subsequent chlorination step relies heavily on phosphorus pentachloride (PCl5), a reagent known for its corrosive nature and the generation of substantial quantities of toxic waste. The combination of these factors results in a process with low total reaction yield and poor atom economy, creating a bottleneck for cost reduction in API manufacturing. The handling of such hazardous reagents also complicates the supply chain, requiring strict containment measures and increasing the overall operational expenditure for chemical producers.
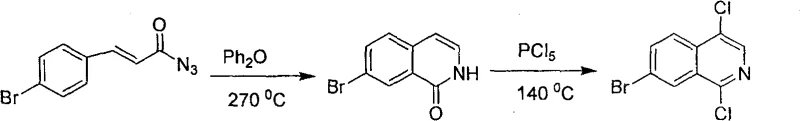
The Novel Approach
The breakthrough presented in the patent data offers a transformative alternative by employing a one-pot strategy that circumvents the drawbacks of the conventional method. By utilizing alpha-azidomethyl aryl ketones as the foundational substrate, the new process enables the direct construction of the 1,4-dichloroisoquinoline core under significantly milder conditions. The key to this efficiency lies in the use of a Vilsmeier reagent, which is prepared in-situ from bis(trichloromethyl) carbonate (BTC) and N,N-dimethylformamide (DMF). This approach eliminates the need for isolating unstable intermediates and avoids the use of solid phosphorus pentachloride, thereby simplifying the workup procedure. The reaction proceeds smoothly in common organic solvents such as 1,2-dichloroethane or acetonitrile, allowing for better heat dissipation and control. For procurement managers, this translates to a drastic simplification of the raw material list and a reduction in the complexity of waste treatment, ultimately leading to substantial cost savings and enhanced process robustness without compromising the structural integrity of the final high-purity OLED material or pharmaceutical intermediate.
Mechanistic Insights into Vilsmeier-Mediated Cyclization
To fully appreciate the technical superiority of this method, one must delve into the mechanistic details of the Vilsmeier-mediated cyclization. The reaction initiates with the formation of the active Vilsmeier complex, a chloroiminium salt generated from the interaction between BTC and DMF. This electrophilic species is crucial for activating the carbonyl group of the alpha-azidomethyl aryl ketone. Upon addition of the ketone substrate, the Vilsmeier reagent facilitates an intramolecular cyclization that constructs the isoquinoline ring system. The presence of electron-donating groups on the aromatic ring, such as methoxy or methyl substituents, can further enhance the nucleophilicity of the ring, promoting smoother cyclization. The reaction conditions are carefully optimized to operate within a temperature range of 0°C to 100°C, with a preferred window of 40°C to 80°C, ensuring that the delicate azide functionality is managed safely while driving the reaction to completion. This controlled environment minimizes the formation of polymeric by-products and other impurities that typically plague high-temperature syntheses.
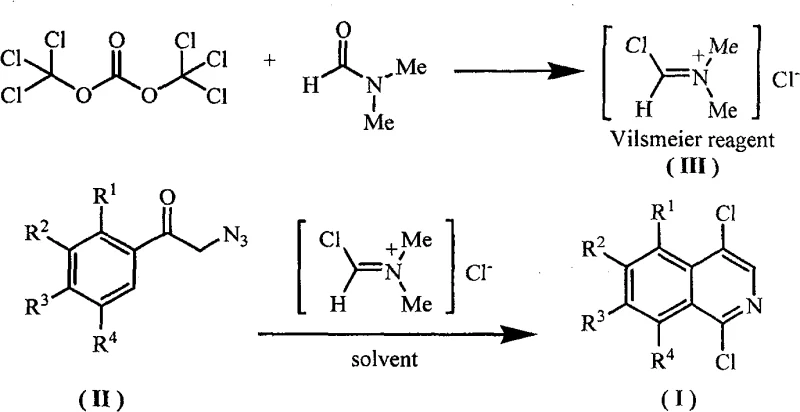
Impurity control is a paramount concern for R&D directors overseeing the scale-up of complex polymer additives or active ingredients. In this novel pathway, the mild reaction conditions play a pivotal role in maintaining a clean impurity profile. Unlike the harsh acidic environments of traditional methods, the Vilsmeier system offers a more selective activation that reduces the likelihood of over-chlorination or ring degradation. The stoichiometry is also finely tuned, with a molar ratio of alpha-azidomethyl aryl ketone to BTC to DMF typically ranging from 1:0.67-3.0:2.0-9.0. This flexibility allows chemists to adjust the reagent loadings to maximize conversion while minimizing excess reagent waste. Post-reaction processing is straightforward, involving a simple quench with saturated sodium bicarbonate solution followed by standard extraction and drying procedures. The resulting crude product can be purified via recrystallization or column chromatography to achieve HPLC purities exceeding 99.0%, meeting the stringent quality standards required for clinical-grade intermediates.
How to Synthesize 1,4-Dichloroisoquinoline Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to ensure reproducibility and safety. The protocol begins with the preparation of the Vilsmeier reagent, which serves as the driving force for the cyclization. Operators must dissolve bis(trichloromethyl) carbonate and N,N-dimethylformamide in a suitable organic solvent, such as 1,2-dichloroethane, and allow the mixture to stir at room temperature to ensure complete formation of the active complex. Once the reagent is ready, the alpha-azidomethyl aryl ketone is introduced, and the temperature is ramped to the optimal range. Monitoring the reaction progress via TLC is essential to determine the precise endpoint, preventing unnecessary exposure of the product to the reaction medium. The following section outlines the standardized steps derived from the patent examples to guide your technical team in executing this transformation effectively.
- Prepare the Vilsmeier reagent by reacting bis(trichloromethyl) carbonate (BTC) with N,N-dimethylformamide (DMF) in an organic solvent at room temperature.
- Add the alpha-azidomethyl aryl ketone substrate to the reaction mixture and maintain the temperature between 40°C and 80°C for 4 to 8 hours.
- Quench the reaction with saturated sodium bicarbonate, separate the organic layer, and purify the crude product via recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply chain resilience, the adoption of this one-pot synthesis method presents a compelling value proposition. The transition from a multi-step, high-hazard process to a streamlined one-pot operation fundamentally alters the cost structure of producing 1,4-dichloroisoquinoline derivatives. By eliminating the need for phosphorus pentachloride, manufacturers can avoid the significant costs associated with purchasing, storing, and disposing of this hazardous material. Furthermore, the reduction in synthesis steps directly correlates to lower labor costs and reduced equipment occupancy time, allowing for higher throughput in existing facilities. The use of commercially abundant reagents like BTC and DMF ensures that the supply chain remains robust, mitigating the risk of shortages that often affect specialty reagents. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the workflow and the elimination of expensive purification steps associated with heavy metal or phosphorus waste. By avoiding the use of PCl5, the process removes the necessity for specialized corrosion-resistant reactors and complex neutralization systems, leading to a significant decrease in capital expenditure and operational overhead. Additionally, the improved atom economy means that a higher proportion of the raw material mass is converted into the desired product, reducing the cost per kilogram of the final API intermediate. The ability to run the reaction in common solvents like dichloroethane or acetonitrile further lowers material costs compared to exotic solvent systems, making the overall process financially attractive for large-scale commercialization.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on readily available starting materials enhances the stability of the supply chain. Alpha-azidomethyl aryl ketones can be synthesized from common acetophenones, which are commodity chemicals with established global supply networks. This reduces dependency on single-source suppliers for niche reagents. Moreover, the mild reaction conditions reduce the risk of batch failures due to thermal excursions, ensuring consistent output quality. For supply chain heads, this predictability is invaluable, as it allows for more accurate forecasting and inventory management. The simplified post-treatment process also shortens the manufacturing cycle time, enabling faster turnaround from order placement to shipment, which is a critical competitive advantage in the fast-paced fine chemical industry.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently designed for scalability. The exothermic nature of the Vilsmeier formation is manageable under the specified conditions, and the absence of solid reagents like PCl5 simplifies dosing and mixing in large vessels. Environmental compliance is another major benefit, as the process generates significantly less hazardous waste compared to the conventional route. The reduction in phosphorus-containing waste streams simplifies wastewater treatment and lowers the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for making informed sourcing decisions. The following questions address common inquiries regarding the feasibility, safety, and quality aspects of the 1,4-dichloroisoquinoline production method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for technical evaluators. Whether you are assessing the purity profile for a new drug application or evaluating the safety protocols for a pilot plant, these insights provide a solid foundation for your due diligence process.
Q: What are the primary advantages of this one-pot method over traditional synthesis?
A: This method eliminates the need for hazardous phosphorus pentachloride and high-temperature cyclization steps, significantly reducing waste generation and improving operational safety while maintaining high product purity.
Q: What is the typical yield range for this synthesis?
A: According to the patent data, the reaction yields typically range from 30% to 80%, depending on the specific substituents on the aryl ring and the reaction conditions employed.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of mild reaction temperatures (0°C to 100°C) and commercially available reagents like BTC and DMF makes this protocol highly scalable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Dichloroisoquinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert chemists has extensively evaluated the synthetic route described in patent CN100534986C and confirmed its potential for robust commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. Our facilities are equipped with state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of 1,4-dichloroisoquinoline derivatives meets the highest industry standards. We are committed to delivering products that facilitate your research and manufacturing goals with reliability and precision.
We invite you to collaborate with us to optimize your supply chain for these valuable heterocyclic compounds. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. By leveraging our expertise in process optimization, we can help you identify opportunities to reduce lead time for high-purity pharmaceutical intermediates and lower your overall procurement costs. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Let us be your partner in navigating the complexities of chemical manufacturing and bringing your innovative therapies to market faster.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
