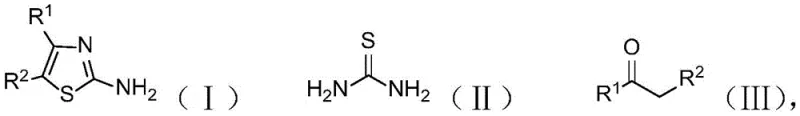
Scalable Iodine-Catalyzed Synthesis of 2-Aminothiazole Derivatives for Pharmaceutical Applications
Introduction to Advanced 2-Aminothiazole Manufacturing
The pharmaceutical industry continuously demands more efficient and sustainable pathways for constructing privileged heterocyclic scaffolds, among which the 2-aminothiazole motif stands out as a cornerstone structure found in numerous bioactive molecules. Patent CN110845437A discloses a groundbreaking methodology for the preparation of 2-aminothiazole compounds, utilizing a direct condensation reaction between thiourea and ketone compounds catalyzed by elemental iodine in dimethyl sulfoxide (DMSO). This innovation addresses critical bottlenecks in traditional synthesis by operating under mild conditions ranging from 50°C to 120°C, thereby offering a robust alternative for producing high-purity pharmaceutical intermediates. The significance of this technology lies in its ability to streamline the production of key drug precursors such as those found in anticancer agents like Dasatinib and cephalosporin antibiotics, ensuring a reliable supply chain for complex medicinal chemistry projects.
As a leading entity in fine chemical manufacturing, understanding the nuances of such patented processes is vital for maintaining competitive advantage in the global market. The described method not only simplifies the operational workflow but also aligns with modern green chemistry principles by minimizing the use of hazardous reagents. For R&D directors and procurement specialists, this represents a tangible opportunity to optimize the cost structure of API intermediate manufacturing while enhancing the overall purity profile of the final products. By leveraging this iodine-catalyzed approach, manufacturers can achieve substantial improvements in process efficiency without compromising on the stringent quality standards required by regulatory bodies worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aminothiazole derivatives has predominantly relied on the Hantzsch thiazole synthesis, which typically involves the reaction of alpha-haloketones with thiourea. While effective, this classical route necessitates the prior preparation or in-situ generation of alpha-haloketones using stoichiometric amounts of halogenating agents such as N-bromosuccinimide (NBS) or molecular bromine. These reagents are not only costly but also generate significant quantities of chemical waste, complicating downstream processing and environmental compliance. Furthermore, alternative methods employing solid-phase reagents like SiO2-Cl require rigorous preparation involving excess thionyl chloride, introducing additional safety hazards and operational complexity that hinder scalability.
Another prevalent strategy involves the use of transition metal catalysts, such as copper trifluoromethanesulfonate, to facilitate the cyclization of alpha-diazoketones and thiourea. Although these methods can offer high chemical selectivity, they suffer from severe drawbacks regarding commercial viability. The precursors like alpha-diazoketones are often unstable and expensive to prepare, while the use of heavy metal catalysts raises serious concerns about metal residues in the final pharmaceutical product. Removing trace metals to meet ppm-level specifications adds expensive purification steps, such as scavenging or recrystallization, which drastically erodes profit margins and extends lead times for critical drug intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the novel iodine-catalyzed protocol presented in the patent data offers a streamlined, one-pot solution that bypasses the need for pre-halogenated substrates or toxic transition metals. By employing elemental iodine as a catalyst in conjunction with DMSO as the solvent, the reaction proceeds efficiently at moderate temperatures, typically optimized at 80°C for 12 hours. This approach fundamentally alters the economic landscape of thiazole synthesis by utilizing cheap, commodity-grade starting materials like acetophenone and thiourea, which are readily available in bulk quantities from global chemical suppliers.
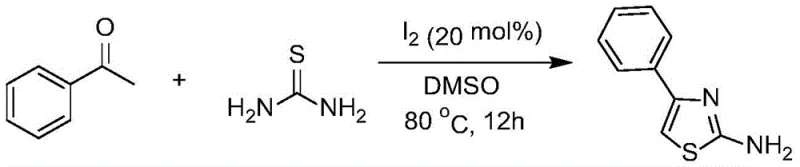
The practical superiority of this method is exemplified by its successful application across a broad spectrum of substrates, including various substituted acetophenones bearing electron-withdrawing or electron-donating groups. For instance, the conversion of acetophenone to 2-amino-4-phenylthiazole proceeds smoothly under these conditions, yielding the target compound as a white solid with a defined melting point, demonstrating the robustness of the chemistry. This versatility ensures that the process can be adapted for the commercial scale-up of complex pharmaceutical intermediates, providing a reliable pathway for synthesizing diverse thiazole derivatives required for next-generation therapeutics without the burden of handling hazardous halogenating agents.
Mechanistic Insights into Iodine-Catalyzed Cyclization
The core of this technological advancement lies in the unique role of elemental iodine acting as a Lewis acid catalyst to activate the carbonyl group of the ketone substrate towards nucleophilic attack by thiourea. In the DMSO solvent system, iodine facilitates the enolization or alpha-activation of the ketone, promoting the initial condensation step that leads to the formation of an intermediate species. This activation lowers the energy barrier for the subsequent cyclization and dehydration steps, allowing the reaction to proceed to completion under relatively mild thermal conditions compared to traditional thermal cyclizations which often require harsh acidic or basic environments.
From an impurity control perspective, the mechanism inherently minimizes the formation of side products associated with over-halogenation or metal-complexation. Since the iodine is used in catalytic amounts (typically 0.05 to 0.2 molar equivalents relative to thiourea), there is no excess halogen source to drive poly-halogenation of the aromatic ring or the alpha-position, which is a common issue in stoichiometric halogenation methods. Furthermore, the absence of transition metals eliminates the risk of metal-ligand complexes forming stable impurities that are difficult to remove. The reaction mixture can be simply quenched with saturated sodium bicarbonate and extracted, yielding a crude product that is amenable to standard purification techniques like silica gel chromatography, ensuring high purity specifications are met with minimal effort.
How to Synthesize 2-Aminothiazole Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and reproducibility. The process begins with the precise weighing of thiourea and the selected ketone compound, maintaining a molar ratio that favors the ketone slightly to drive the equilibrium forward, typically within a range of 1:1 to 1:3. The addition of the iodine catalyst must be controlled carefully, as its concentration directly influences the reaction rate and the potential for side reactions, with 20 mol% identified as the optimal loading for balancing speed and selectivity.
- Mix thiourea, ketone compound, and catalytic iodine (0.05-0.2 equiv) in DMSO solvent.
- Heat the reaction mixture to 50-120°C (optimally 80°C) and stir for 6-24 hours.
- Quench with saturated NaHCO3, extract with ethyl acetate, dry, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-catalyzed methodology translates into immediate and tangible strategic benefits that extend beyond simple reaction yields. The primary advantage stems from the drastic simplification of the raw material portfolio; by eliminating the need for specialized, high-cost halogenating reagents and sensitive transition metal catalysts, the purchasing department can source all necessary inputs from standard bulk chemical vendors. This consolidation of the supply base reduces vendor management overhead and mitigates the risk of supply disruptions caused by the scarcity of niche reagents, thereby enhancing the overall resilience of the manufacturing operation.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalytic system is profound, primarily driven by the elimination of expensive stoichiometric reagents and the associated waste disposal costs. Traditional methods often require equimolar or excess amounts of brominating agents which are not only pricey but also generate stoichiometric amounts of salt waste that must be treated. By using catalytic iodine, the material cost per kilogram of product is significantly lowered, and the simplified workup procedure reduces solvent consumption and labor hours, leading to substantial cost savings in the overall manufacturing budget without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acetophenone derivatives, thiourea, and elemental iodine ensures a stable and continuous supply of raw materials, as these are produced globally in massive volumes. Unlike specialized catalysts or unstable diazo compounds which may have long lead times or single-source dependencies, these foundational chemicals are readily accessible, allowing for just-in-time inventory strategies and reducing the capital tied up in raw material stockpiles. This reliability is crucial for maintaining consistent production schedules and meeting the tight delivery windows demanded by pharmaceutical clients.
- Scalability and Environmental Compliance: From an operational standpoint, the mild reaction conditions and simple aqueous workup make this process highly scalable from gram to multi-ton quantities without requiring exotic equipment or extreme safety measures. The absence of heavy metals simplifies the environmental compliance landscape, as there is no need for complex metal recovery systems or rigorous testing for residual copper or palladium in the final API. This ease of scale-up facilitates rapid technology transfer from R&D to commercial production, enabling faster time-to-market for new drug candidates while adhering to increasingly stringent environmental regulations regarding chemical waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-catalyzed synthesis platform. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and strategic value of adopting this method for large-scale intermediate production. Understanding these details is essential for technical teams evaluating the integration of this chemistry into existing manufacturing workflows.
Q: What are the advantages of using iodine over traditional halogenating reagents?
A: Using catalytic iodine eliminates the need for stoichiometric amounts of hazardous halogenating agents like NBS or bromine, significantly reducing waste generation and raw material costs while simplifying the purification process.
Q: Is this method suitable for large-scale production of API intermediates?
A: Yes, the method utilizes cheap and readily available raw materials and mild reaction conditions (80°C), avoiding expensive transition metal catalysts that pose residue risks, making it highly suitable for commercial scale-up.
Q: What is the typical substrate scope for this synthesis?
A: The protocol accommodates a wide range of ketones including acetophenones with various substituents (fluoro, chloro, bromo, methyl, methoxy) and aliphatic ketones, demonstrating robust versatility for diverse pharmaceutical scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminothiazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and robust. We are committed to delivering high-purity 2-aminothiazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical techniques to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced iodine-catalyzed technology for your upcoming projects. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce manufacturing costs while securing a reliable source of high-quality pharmaceutical intermediates for your global operations.
