Advanced One-Step Synthesis of (E)-1-Chloro-6,6-Dimethyl-2-Hepten-4-Yne for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for high-volume active pharmaceutical ingredients (APIs), and the production of Terbinafine Hydrochloride, a leading broad-spectrum antifungal agent, relies heavily on the efficient supply of its key precursor, (E)-1-chloro-6,6-dimethyl-2-hepten-4-yne. Patent CN101624328A introduces a transformative methodology that addresses long-standing bottlenecks in the manufacturing of this critical intermediate. By shifting from traditional multi-step sequences to a direct, one-step condensation reaction, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains. The core innovation lies in the utilization of trans-1,3-dichloropropene and tert-butylacetylene as starting materials, coupled through a sophisticated palladium and copper catalytic system. This approach not only streamlines the operational workflow but also significantly enhances the overall atom economy and safety profile of the synthesis. As global demand for antifungal medications continues to rise, securing a reliable source of high-purity intermediates produced via such advanced methodologies becomes a strategic imperative for procurement and R&D teams alike.
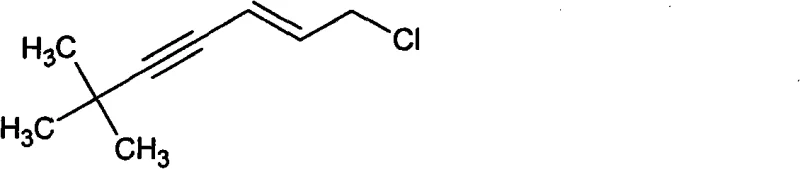
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of (E)-1-chloro-6,6-dimethyl-2-hepten-4-yne has been plagued by complex, multi-stage processes that impose significant operational burdens and safety risks. Traditional routes, as documented in earlier patents such as PCT Int. Appl. 2007052089, typically involve the reaction of tert-butylacetylene with acrolein via Grignard reagents or organolithium species to form an intermediate alcohol. This initial step requires stringent anhydrous conditions and inert atmosphere protection, often necessitating cryogenic temperatures to control exothermic reactions and prevent side products. Following the formation of the alcohol, a subsequent chlorination and rearrangement step using aggressive reagents like phosphorus oxychloride (POCl3), phosphorus pentachloride (PCl5), or thionyl chloride (SOCl2) is required to achieve the final chloro-alkyne structure. These legacy methods are characterized by low overall yields, frequently failing to exceed 70%, and generate substantial quantities of hazardous acidic waste that complicate environmental compliance. Furthermore, the handling of pyrophoric organometallic reagents and corrosive chlorinating agents increases the risk of workplace accidents and demands specialized, high-maintenance equipment.
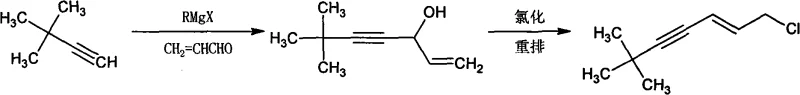
The Novel Approach
In stark contrast to the cumbersome legacy protocols, the novel methodology disclosed in CN101624328A achieves the target molecule through a direct, catalytic cross-coupling reaction that bypasses the need for isolated alcohol intermediates and harsh chlorinating agents. This streamlined process utilizes trans-1,3-dichloropropene and tert-butylacetylene as the sole carbon sources, reacting them in the presence of a palladium catalyst, cuprous iodide, and an organic amine base within a tetrahydrofuran (THF) solvent system. The reaction proceeds under remarkably mild thermal conditions, typically maintained around 40°C, which drastically reduces energy consumption compared to the heating or cooling cycles required by older methods. By eliminating the separate chlorination rearrangement step, the new route inherently avoids the generation of phosphorous-containing waste streams and simplifies the purification process to a straightforward extraction and vacuum distillation. This technological leap results in a dramatic improvement in process efficiency, with reported yields consistently reaching or exceeding 85%, thereby maximizing raw material utilization and minimizing production costs per kilogram of output.
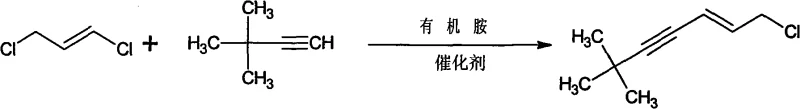
Mechanistic Insights into Pd/Cu-Catalyzed Cross-Coupling
The success of this innovative synthesis hinges on the precise orchestration of a palladium-copper co-catalytic cycle, mechanistically similar to the renowned Sonogashira coupling but adapted for the specific reactivity of dichloropropene. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the trans-1,3-dichloropropene, forming a reactive pi-allyl palladium complex. Simultaneously, the terminal alkyne, tert-butylacetylene, undergoes deprotonation by the organic amine base and coordination with the cuprous iodide co-catalyst to generate a copper-acetylide species. This nucleophilic copper intermediate then engages in a transmetallation step with the palladium complex, transferring the alkynyl group to the metal center. The final stage involves reductive elimination, which releases the desired (E)-1-chloro-6,6-dimethyl-2-hepten-4-yne product while regenerating the active palladium(0) catalyst to sustain the cycle. The stereoselectivity for the (E)-isomer is rigorously controlled by the geometry of the pi-allyl intermediate and the steric environment provided by the phosphine ligands, ensuring high isomeric purity essential for downstream pharmaceutical applications.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed rearrangements which often lead to polymerization or isomerization byproducts. The mild basic conditions employed in the new protocol prevent the acid-catalyzed degradation of the sensitive enyne system, which is prone to decomposition under the harsh acidic environments of POCl3 or SOCl2 treatments. Furthermore, the use of specific ligands such as triphenylphosphine helps stabilize the palladium center against aggregation into inactive palladium black, maintaining catalytic activity over extended reaction times of 10 to 50 hours. The workup procedure, involving a wash with dilute ammonia water, effectively removes residual copper salts and amine hydrochlorides, yielding a crude product that requires minimal further purification. This robustness in mechanism translates directly to a cleaner impurity profile, reducing the burden on quality control laboratories and ensuring that the final intermediate meets the stringent specifications required for API synthesis.
How to Synthesize (E)-1-Chloro-6,6-Dimethyl-2-Hepten-4-Yne Efficiently
The implementation of this synthesis route in a pilot or production plant follows a logical sequence designed to maximize safety and yield while minimizing operational complexity. The process begins with the preparation of the catalytic mixture in a standard stainless steel reactor equipped with efficient agitation and temperature control systems. Operators charge the solvent, base, and catalysts prior to the introduction of reactants to ensure immediate catalytic activity upon mixing. The dropwise addition of tert-butylacetylene is a critical control point, managed carefully to maintain the reaction temperature at the optimal 40°C setpoint, preventing thermal runaway while ensuring complete conversion. Following the reaction period, the isolation strategy leverages the volatility differences between the product and high-boiling impurities, utilizing vacuum distillation to obtain the final colorless to pale yellow liquid. For detailed standard operating procedures and specific parameter optimizations, please refer to the technical guide below.
- Charge trans-1,3-dichloropropene, organic amine base, and solvent (THF) into a reactor, then add Pd(0)/Pd(II) catalyst and CuI.
- Dropwise add tert-butylacetylene at 40°C under stirring and maintain reaction temperature for 10-50 hours.
- Remove solvent under reduced pressure, extract with petroleum ether, wash with dilute ammonia, and purify via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route represents a significant opportunity to enhance supply security and reduce total landed costs without compromising on quality. The shift from a multi-step batch process to a streamlined one-step condensation fundamentally alters the cost structure of manufacturing this intermediate. By eliminating the need for expensive and hazardous reagents like organolithiums and phosphorus chlorides, the direct material costs are substantially lowered. Moreover, the reduction in unit operations—from potentially four or five distinct chemical transformations down to a single reaction vessel—drastically cuts down on labor hours, utility consumption, and equipment occupancy time. This operational efficiency allows manufacturers to respond more agilely to market fluctuations in demand for Terbinafine, ensuring that supply contracts can be fulfilled with greater reliability and shorter lead times.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic pathway and the avoidance of costly reagents. Traditional methods require the purchase and safe handling of cryogenic-grade solvents and pyrophoric metals, which incur high logistics and storage costs; the new method utilizes stable, commodity-grade chemicals that are readily available in the global market. Additionally, the significant increase in yield from below 70% to over 85% means that less raw material is wasted per kilogram of finished product, directly improving the gross margin for producers. The elimination of the chlorination rearrangement step also removes the need for specialized corrosion-resistant equipment and extensive waste neutralization facilities, resulting in substantial capital expenditure (CAPEX) savings for new plant installations and lower maintenance costs for existing facilities.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of trans-1,3-dichloropropene and tert-butylacetylene, which are established bulk chemicals with robust global supply networks, unlike some specialized intermediates required in older routes. The mild reaction conditions reduce the risk of unplanned shutdowns due to equipment failure or safety incidents, which are more common in processes involving extreme temperatures or pressures. Furthermore, the simplified workup and purification steps reduce the turnaround time between batches, allowing for higher throughput and the ability to build inventory buffers more effectively. This reliability is crucial for pharmaceutical companies that require consistent, uninterrupted supply of key intermediates to maintain their own API production schedules and meet regulatory filing commitments.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial tonnage is facilitated by the inherent safety and simplicity of the reaction chemistry. The absence of highly exothermic steps and the use of non-corrosive reagents make the process easier to scale in standard glass-lined or stainless steel reactors without requiring exotic engineering solutions. From an environmental standpoint, the process aligns with modern green chemistry principles by reducing the E-factor (mass of waste per mass of product) through higher yields and fewer steps. The avoidance of phosphorous waste streams simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site, helping companies meet increasingly stringent regulatory standards and corporate sustainability goals without the need for expensive end-of-pipe treatment technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements. The answers reflect the consensus on the operational benefits and chemical realities of the Pd-catalyzed coupling route versus traditional methodologies.
Q: What are the primary advantages of the new Pd-catalyzed route over traditional Grignard methods?
A: The new route eliminates the need for hazardous Grignard reagents and cryogenic conditions, reducing the process from multiple steps to a single condensation step while increasing yields from below 70% to over 85%.
Q: How does this synthesis method impact environmental compliance?
A: By avoiding chlorinating agents like POCl3 or PCl5 used in traditional rearrangement steps, the new method significantly reduces hazardous waste generation and simplifies effluent treatment protocols.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the mild reaction conditions (40°C), use of stable raw materials, and simplified workup procedure make it highly scalable and robust for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-1-Chloro-6,6-Dimethyl-2-Hepten-4-Yne Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficient production of (E)-1-chloro-6,6-dimethyl-2-hepten-4-yne is pivotal to the global supply of antifungal therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging advanced catalytic technologies similar to those described in CN101624328A, we optimize our manufacturing processes to offer competitive pricing while maintaining the highest standards of quality and safety.
We invite you to collaborate with us to explore how our manufacturing capabilities can support your supply chain objectives. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating the tangible economic benefits of partnering with us. Please contact us today to request specific COA data for our current batches and to discuss route feasibility assessments for your upcoming projects. Let us be your strategic partner in delivering high-quality pharmaceutical intermediates to the market efficiently and reliably.
