Revolutionizing Terbinafine Production: A Scalable, One-Step Catalytic Coupling Strategy for High-Purity Intermediates
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antifungal agents, particularly Terbinafine Hydrochloride, where supply chain stability is paramount. Patent CN101624328B introduces a transformative methodology for synthesizing (E)-1-chloro-6,6-dimethyl-2-hepten-4-yne, a pivotal building block in this therapeutic class. Unlike legacy processes that rely on hazardous multi-step sequences, this invention leverages a direct catalytic condensation between trans-1,3-dichloropropene and tert-butylacetylene. This strategic shift not only simplifies the molecular construction but also drastically enhances the environmental profile of the production line. By bypassing the need for aggressive chlorinating reagents and sensitive organometallic preparations, manufacturers can achieve superior operational safety and consistency. The structural integrity of this key intermediate is fundamental to the efficacy of the final drug substance, making the precision of this synthetic route a critical quality attribute for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
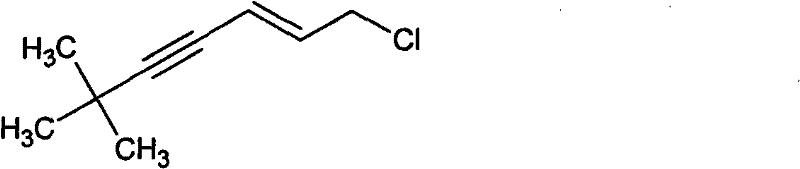
The Limitations of Conventional Methods
Historically, the industrial preparation of this chloro-alkyne intermediate has been plagued by significant inefficiencies and safety hazards inherent to organometallic chemistry. Traditional routes, as documented in prior art such as PCT Int. Appl. 2007052089, typically commence with the formation of a propargylic alcohol via Grignard or lithium acetylide addition to acrolein. This initial step demands rigorous anhydrous conditions and inert nitrogen atmospheres to prevent reagent decomposition, imposing severe constraints on reactor design and operational throughput. Furthermore, the subsequent conversion of the hydroxyl group to the requisite chloride necessitates the use of corrosive and toxic reagents like phosphorus oxychloride (POCl3) or thionyl chloride (SOCl2). These chlorination steps often proceed with poor atom economy and generate substantial acidic waste streams that require costly neutralization and disposal protocols. Additionally, the overall yield of these multi-step sequences rarely exceeds 70%, leading to significant material loss and inflated production costs that erode profit margins in competitive generic markets.
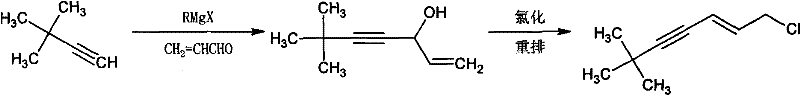
The Novel Approach
In stark contrast, the methodology disclosed in CN101624328B represents a paradigm shift towards atom-economical and operationally simple manufacturing. The core innovation lies in the direct coupling of trans-1,3-dichloropropene with tert-butylacetylene, effectively merging carbon skeleton construction and functional group installation into a single unit operation. This one-step condensation eliminates the isolation of unstable alcohol intermediates and completely avoids the use of hazardous chlorinating agents for the backbone modification. The reaction proceeds smoothly in tetrahydrofuran (THF) using a dual catalytic system comprising palladium complexes and cuprous iodide, moderated by an organic amine base. This approach not only reduces the number of processing stages but also mitigates the risks associated with handling pyrophoric reagents. The result is a streamlined workflow that delivers the target molecule with exceptional purity and yields consistently above 85%, offering a compelling alternative for facilities aiming to modernize their Terbinafine supply chains.
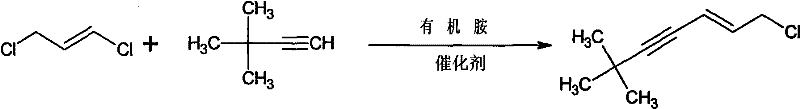
Mechanistic Insights into Pd/Cu Catalyzed Cross-Coupling
The success of this synthesis hinges on the synergistic interaction between the palladium catalyst and the copper co-catalyst, facilitating a cross-coupling mechanism reminiscent of Sonogashira chemistry but adapted for allylic substrates. The palladium species, typically a zero-valent complex like tetrakis(triphenylphosphine)palladium(0), undergoes oxidative addition with the trans-1,3-dichloropropene, selectively activating the allylic chloride bond while preserving the vinyl chloride moiety essential for the final structure. Simultaneously, the terminal alkyne, tert-butylacetylene, is activated by the cuprous iodide and the amine base to form a reactive copper-acetylide species. This nucleophilic organocopper intermediate then engages in a transmetallation event with the palladium-allyl complex. The subsequent reductive elimination step releases the coupled product, regenerating the active palladium catalyst for the next cycle. This catalytic cycle is highly efficient, operating at mild temperatures around 40°C, which minimizes thermal degradation of sensitive functional groups and prevents the formation of polymeric byproducts often seen in radical-based halogenations.
From a quality control perspective, the mechanistic pathway inherently favors the formation of the desired (E)-isomer due to the stereochemical retention of the starting trans-1,3-dichloropropene. The use of bulky phosphine ligands on the palladium center further enhances regioselectivity, ensuring that the coupling occurs at the correct position to yield the 2-hepten-4-yne framework rather than unwanted isomers. Impurity profiles are significantly cleaner compared to acid-catalyzed rearrangements, as the reaction does not generate the complex mixture of elimination and substitution byproducts typical of POCl3 treatments. The workup procedure, involving a simple wash with dilute ammonia solution, effectively removes copper salts and amine hydrochlorides, leaving a crude product that requires only vacuum distillation to achieve pharmaceutical-grade purity. This robustness in impurity control is vital for meeting the stringent specifications required for API intermediates destined for regulated markets.
How to Synthesize (E)-1-chloro-6,6-dimethyl-2-hepten-4-yne Efficiently
The implementation of this catalytic route requires precise control over stoichiometry and reaction parameters to maximize the benefits of the novel chemistry. Operators must ensure the correct molar ratios of the palladium catalyst, copper co-catalyst, and amine base are maintained to sustain the catalytic turnover without inducing side reactions. The following guide outlines the standardized operational protocol derived from the patent examples, designed to ensure reproducibility and safety during scale-up. Detailed standard operating procedures regarding specific equipment setup and safety precautions should be consulted prior to execution.
- Charge trans-1,3-dichloropropene, palladium catalyst (e.g., Pd(PPh3)4), cuprous iodide, and organic amine base into a THF solvent system under stirring.
- Maintain reaction temperature at 40°C while dropwise adding tert-butylacetylene, followed by continued agitation for 10 to 50 hours to ensure complete conversion.
- Remove solvent under reduced pressure, wash the residue with dilute ammonia solution to remove salts, and purify the crude product via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the adoption of this synthesis route offers tangible benefits that extend beyond mere chemical elegance. The reduction in synthetic steps directly correlates to a decrease in capital expenditure on processing equipment and a reduction in the overall manufacturing cycle time. By eliminating the need for specialized containment systems required for handling pyrophoric Grignard reagents or corrosive phosphorus halides, facilities can utilize standard glass-lined or stainless steel reactors, thereby lowering the barrier to entry for contract manufacturing organizations. The simplified workflow also reduces the manpower hours required per batch, allowing for more flexible production scheduling and faster response times to market demand fluctuations for Terbinafine. Furthermore, the reliance on commodity chemicals like trans-1,3-dichloropropene ensures a stable and diversified supply base, mitigating the risk of raw material shortages that often plague specialty reagent-dependent processes.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the drastic simplification of the process flow. By consolidating multiple reaction and isolation steps into a single pot operation, manufacturers eliminate the cumulative yield losses associated with each intermediate transfer and purification stage. The avoidance of expensive and hazardous chlorinating agents like POCl3 removes a significant cost center related to both reagent procurement and hazardous waste disposal. Additionally, the high yield performance, consistently demonstrated above 85% in patent examples compared to sub-70% for legacy routes, means that less raw material is required to produce the same amount of finished goods, directly improving the cost of goods sold (COGS). This efficiency gain allows for more competitive pricing strategies in the global generic pharmaceutical market without compromising margin integrity.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust, non-sensitive starting materials that do not require cryogenic storage or inert atmosphere transport. Unlike lithium or Grignard reagents which have limited shelf lives and strict handling requirements, the reagents for this process are stable and widely available from multiple chemical suppliers globally. This diversification of the supply base reduces dependency on single-source vendors and minimizes the risk of production stoppages due to reagent degradation or delivery delays. The mild reaction conditions also reduce the likelihood of unplanned shutdowns caused by thermal runaways or equipment corrosion, ensuring a predictable and reliable output of high-quality intermediate to support continuous API manufacturing campaigns.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial tonnage is facilitated by the absence of highly exothermic steps that are difficult to manage in large reactors. The moderate temperature profile (40°C) allows for efficient heat management using standard cooling systems, removing the need for specialized cryogenic infrastructure. From an environmental standpoint, the process generates significantly less hazardous waste, particularly avoiding the phosphate-rich effluents associated with traditional chlorination methods. This aligns with increasingly stringent global environmental regulations and corporate sustainability goals, reducing the liability and cost associated with wastewater treatment and regulatory compliance audits. The cleaner process profile also simplifies the permitting process for new manufacturing lines, accelerating time-to-market for new capacity expansions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent literature to provide accurate guidance for process development teams. Understanding these nuances is essential for evaluating the feasibility of technology transfer and integration into existing manufacturing portfolios.
Q: What are the primary advantages of this new synthesis route over traditional Grignard methods?
A: The novel Pd-catalyzed coupling eliminates the need for hazardous chlorinating agents like POCl3 or PCl5 and avoids the strict anhydrous conditions required for Grignard reagents, resulting in yields exceeding 85% compared to traditional yields below 70%.
Q: Is this process suitable for large-scale commercial manufacturing of Terbinafine intermediates?
A: Yes, the process operates at mild temperatures (40°C) using readily available raw materials like trans-1,3-dichloropropene, significantly reducing equipment requirements and safety risks associated with exothermic organometallic reactions.
Q: How is the stereochemical purity (E-isomer) controlled in this reaction?
A: The use of trans-1,3-dichloropropene as the starting material inherently directs the formation of the (E)-configuration in the final alkene moiety, ensuring high stereo-selectivity without complex isomer separation steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-1-chloro-6,6-dimethyl-2-hepten-4-yne Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical application. Our state-of-the-art facilities are equipped to handle the specific catalytic requirements of this synthesis, maintaining stringent purity specifications through our rigorous QC labs and advanced analytical instrumentation. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global healthcare industry, providing a secure foundation for your Terbinafine supply chain.
We invite you to engage with our technical procurement team to discuss how this innovative route can optimize your specific manufacturing requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits tailored to your production volume. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and our commitment to quality excellence.
