Advanced One-Step Synthesis of N-Acylindoles for Scalable Pharmaceutical Manufacturing
Advanced One-Step Synthesis of N-Acylindoles for Scalable Pharmaceutical Manufacturing
The structural motif of indole is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to potent anti-tumor compounds. As illustrated in the diverse pharmacological profiles shown below, the ability to efficiently construct functionalized indole derivatives is a critical capability for any advanced chemical manufacturer. 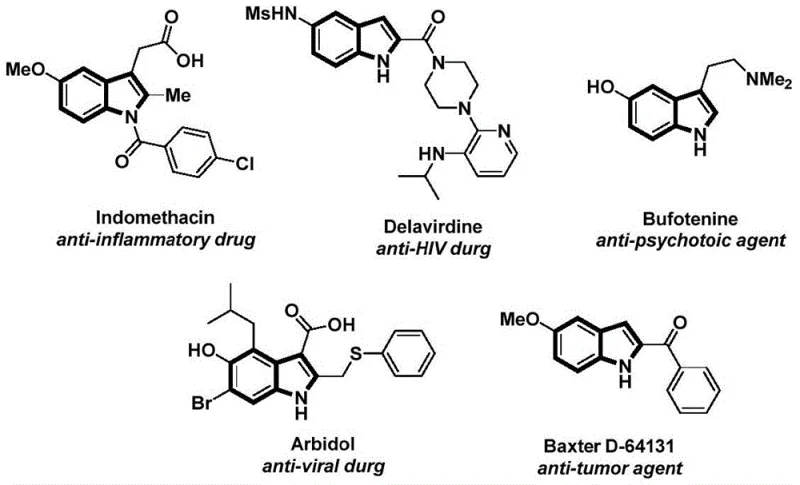 Recent advancements in organic synthesis have sought to streamline the construction of these complex heterocycles. Specifically, Patent CN112898192A discloses a groundbreaking preparation method for N-acylindole compounds that utilizes a palladium-catalyzed carbonylative cyclization strategy. This technology represents a significant leap forward in process chemistry, offering a robust, one-pot solution that bypasses the limitations of traditional multi-step sequences. By leveraging readily available starting materials and mild reaction conditions, this patented approach provides a reliable pathway for the commercial scale-up of complex pharmaceutical intermediates, addressing key pain points in both R&D feasibility and supply chain stability.
Recent advancements in organic synthesis have sought to streamline the construction of these complex heterocycles. Specifically, Patent CN112898192A discloses a groundbreaking preparation method for N-acylindole compounds that utilizes a palladium-catalyzed carbonylative cyclization strategy. This technology represents a significant leap forward in process chemistry, offering a robust, one-pot solution that bypasses the limitations of traditional multi-step sequences. By leveraging readily available starting materials and mild reaction conditions, this patented approach provides a reliable pathway for the commercial scale-up of complex pharmaceutical intermediates, addressing key pain points in both R&D feasibility and supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acylindole derivatives has often relied on cumbersome multi-step protocols that involve the separate formation of the indole ring followed by N-acylation, or vice versa. These conventional routes frequently suffer from poor atom economy, requiring harsh reagents and generating substantial chemical waste. Furthermore, traditional carbonylation methods often necessitate the use of high-pressure carbon monoxide gas, which poses severe safety hazards and requires specialized, expensive equipment that is not readily available in standard pilot plants. The handling of toxic CO gas also introduces significant regulatory and logistical burdens, complicating the supply chain and increasing the overall cost of goods sold. Additionally, many existing methods exhibit limited substrate scope, failing to tolerate sensitive functional groups such as halogens or electron-withdrawing groups, which are often essential for the biological activity of the final drug molecule. These inefficiencies create bottlenecks in the development timeline, delaying the transition from laboratory discovery to commercial production.
The Novel Approach
In stark contrast, the methodology described in Patent CN112898192A introduces a highly efficient, tandem reaction sequence that constructs the N-acylindole core in a single operational step. This novel approach utilizes 2-alkynyl aniline and aryl iodide as building blocks, reacting them in the presence of a palladium catalyst and a solid carbon monoxide surrogate known as TFBen (phenol 1,3,5-tricarboxylate). 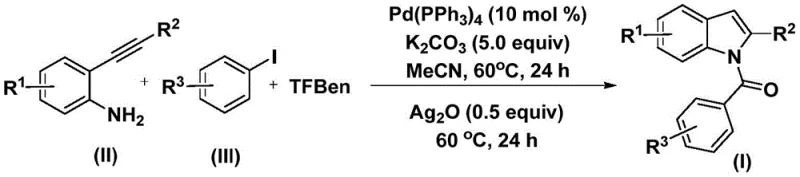 By replacing gaseous CO with a stable solid surrogate, the process dramatically enhances operational safety and simplifies the reactor setup, making it ideal for large-scale manufacturing. The reaction proceeds under mild thermal conditions (60°C) in acetonitrile, demonstrating exceptional compatibility with a wide range of functional groups. This streamlined protocol not only reduces the number of unit operations but also minimizes solvent consumption and waste generation, aligning perfectly with green chemistry principles. The result is a versatile platform technology capable of producing high-purity N-acylindoles with impressive yields, as evidenced by the successful synthesis of various derivatives shown in the patent examples.
By replacing gaseous CO with a stable solid surrogate, the process dramatically enhances operational safety and simplifies the reactor setup, making it ideal for large-scale manufacturing. The reaction proceeds under mild thermal conditions (60°C) in acetonitrile, demonstrating exceptional compatibility with a wide range of functional groups. This streamlined protocol not only reduces the number of unit operations but also minimizes solvent consumption and waste generation, aligning perfectly with green chemistry principles. The result is a versatile platform technology capable of producing high-purity N-acylindoles with impressive yields, as evidenced by the successful synthesis of various derivatives shown in the patent examples.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The success of this transformation lies in the intricate interplay between the palladium catalyst and the unique reactivity of the substrates. The mechanism initiates with the oxidative addition of the aryl iodide to the active Pd(0) species, generating an aryl-palladium(II) intermediate. Subsequently, carbon monoxide, which is slowly released in situ from the decomposition of TFBen, inserts into the palladium-carbon bond to form an acyl-palladium complex. This acyl species then undergoes nucleophilic attack or insertion with the 2-alkynyl aniline, followed by reductive elimination to yield an intermediate amide. The brilliance of this system is further highlighted in the second stage, where the addition of silver oxide promotes the intramolecular cyclization of the amide intermediate. The silver oxide likely acts as a mild oxidant or base to facilitate the C-H activation or deprotonation required to close the five-membered pyrrole ring,最终 yielding the stable N-acylindole structure. This dual-stage mechanism ensures high conversion rates and minimizes the formation of byproducts, providing a clean reaction profile that is highly desirable for industrial applications.
From an impurity control perspective, the use of TFBen as a controlled CO source is particularly advantageous. Unlike bubbling CO gas, which can lead to uneven concentrations and potential over-carbonylation or polymerization side reactions, the slow release of CO from TFBen maintains a steady, low concentration of the reactive gas in the solution. This kinetic control suppresses competing pathways and ensures that the carbonylation step occurs selectively at the intended site. Furthermore, the choice of acetonitrile as the solvent optimizes the solubility of both the organic substrates and the inorganic bases, facilitating homogeneous catalysis. The broad substrate tolerance observed, accommodating substituents like fluorine, chlorine, methoxy, and methyl groups at various positions on the aromatic rings, indicates that the catalytic cycle is robust against steric and electronic variations. This mechanistic resilience translates directly to process reliability, allowing manufacturers to produce a diverse library of analogues without needing to re-optimize reaction conditions for each new derivative.
How to Synthesize N-Acylindole Compounds Efficiently
The practical implementation of this synthesis is straightforward and relies on standard laboratory equipment, making it highly accessible for process development teams. The protocol involves a simple charge of reagents followed by a controlled heating sequence. To ensure optimal results, it is critical to maintain the specified molar ratios, particularly the 0.1:5:5:0.5 ratio of palladium catalyst to base to CO source to silver oxide. The detailed standardized synthetic steps, including precise workup and purification procedures to achieve pharmaceutical-grade purity, are outlined in the guide below.
- Combine palladium catalyst, potassium carbonate, TFBen, 2-alkynyl aniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the initial carbonylation and coupling.
- Add silver oxide and continue heating at 60°C for another 24 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible strategic benefits that extend beyond mere chemical efficiency. The shift from hazardous gas reagents to solid surrogates fundamentally alters the risk profile of the manufacturing process, reducing insurance costs and regulatory compliance burdens. Moreover, the reliance on commodity chemicals like aryl iodides and 2-alkynyl anilines ensures a stable and competitive raw material supply base, mitigating the risk of shortages that often plague specialty reagent markets.
- Cost Reduction in Manufacturing: The elimination of high-pressure CO gas infrastructure represents a significant capital expenditure saving, as facilities do not require specialized autoclaves or gas handling systems. Additionally, the one-pot nature of the reaction reduces labor costs and utility consumption by combining what would traditionally be two or three separate steps into a single vessel operation. The high atom economy and reduced solvent usage further contribute to a lower cost of goods, allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials and avoiding reagents with complex logistics (such as compressed gases), the supply chain becomes more resilient to disruptions. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without extensive re-validation, ensuring continuity of supply even in the face of regional instability. This flexibility is crucial for maintaining just-in-time delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction temperature of 60°C and the use of acetonitrile, a common and recyclable solvent, make this process highly scalable from kilogram to multi-ton quantities. The simplified workup procedure, involving filtration and standard chromatography, reduces the generation of hazardous waste streams. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations seeking sustainable partners, positioning this method as a future-proof solution for green manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-acylindole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous CO gas cylinders, thereby significantly improving operational safety and simplifying reactor requirements.
Q: What is the substrate compatibility of this palladium-catalyzed method?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents such as halogens (F, Cl, Br), alkyl groups (methyl, tert-butyl), and alkoxy groups (methoxy) on both the aniline and aryl iodide components without significant yield loss.
Q: How does the two-stage heating process improve product purity?
A: The sequential addition of silver oxide in the second stage ensures complete cyclization of the intermediate amide to the final N-acylindole structure, minimizing residual starting materials and side products, which simplifies downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acylindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the potential of this palladium-catalyzed carbonylation technology and is fully prepared to integrate it into our production capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of N-acylindole intermediate meets the highest international standards for quality and consistency.
We invite you to collaborate with us to leverage this innovative synthesis method for your specific drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this route can optimize your budget. Please contact us today to discuss your requirements, and we will promptly provide specific COA data and comprehensive route feasibility assessments to support your decision-making process.
