Advanced Palladium-Catalyzed Synthesis of N-Acylindole Compounds for Commercial Pharmaceutical Production
Advanced Palladium-Catalyzed Synthesis of N-Acylindole Compounds for Commercial Pharmaceutical Production
The structural motif of indole is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules including anti-inflammatory agents, anti-tumor drugs, and antiviral compounds. As depicted in the reference structures of well-known pharmaceuticals such as Indomethacin and Delavirdine, the functionalization of the indole nitrogen is a critical transformation for modulating biological activity. However, traditional methods for constructing N-acylindole frameworks often suffer from harsh reaction conditions, poor atom economy, or the requirement for hazardous gaseous reagents. Addressing these industrial pain points, the Chinese patent CN112898192A discloses a highly efficient and operationally simple preparation method for N-acylindole compounds. This innovative protocol leverages a palladium-catalyzed carbonylation cyclization strategy that not only streamlines the synthetic route but also significantly enhances the safety profile by utilizing a solid carbon monoxide surrogate.
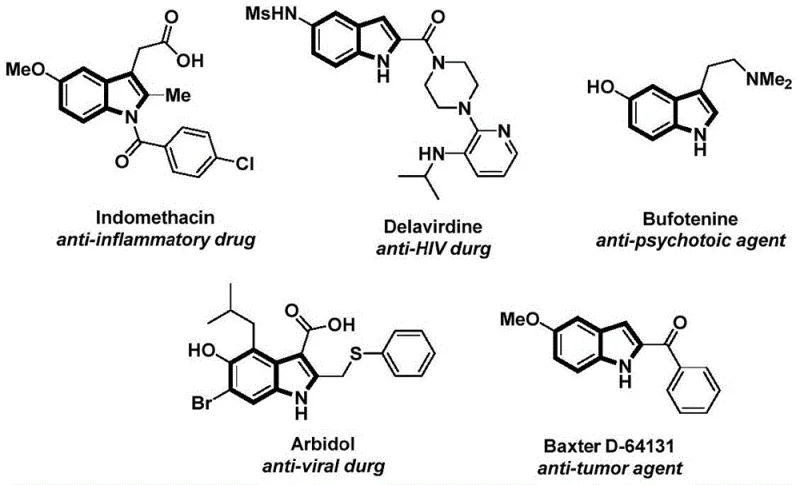
For procurement managers and supply chain directors seeking a reliable pharmaceutical intermediate supplier, this technology represents a substantial opportunity for cost reduction in API manufacturing. By replacing dangerous gases with stable solids and employing widely available aryl iodides and alkynyl anilines, the process mitigates supply chain risks associated with specialized reagent sourcing. Furthermore, the mild reaction temperatures and high substrate compatibility ensure that this method can be seamlessly integrated into existing production lines, facilitating the commercial scale-up of complex pharmaceutical intermediates without the need for exotic high-pressure equipment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acylated heterocycles has relied heavily on direct acylation using acid chlorides or anhydrides, which often generates stoichiometric amounts of corrosive waste and requires strict moisture control. Alternatively, transition-metal catalyzed carbonylation using carbon monoxide gas has been explored, but this approach presents severe logistical and safety challenges for large-scale operations. The handling of high-pressure CO cylinders necessitates specialized infrastructure, rigorous safety protocols, and expensive containment systems, all of which drive up the capital expenditure and operational costs for chemical manufacturers. Additionally, traditional carbonylation reactions frequently require elevated temperatures and pressures that can lead to decomposition of sensitive functional groups, thereby limiting the scope of substrates that can be effectively utilized in drug discovery and development pipelines.
The Novel Approach
The methodology described in patent CN112898192A offers a transformative solution by employing 1,3,5-tricarboxylic acid phenol ester (TFBen) as a safe and efficient solid carbon monoxide substitute. This strategic substitution allows the reaction to proceed under atmospheric pressure and mild thermal conditions, specifically at 60°C, which is remarkably gentle compared to conventional high-pressure carbonylations. The process involves a tandem reaction where 2-alkynyl aniline and aryl iodide are coupled in the presence of a palladium catalyst and potassium carbonate. As illustrated in the general reaction scheme, this one-pot procedure efficiently constructs the N-acylindole core through an initial carbonylation followed by an oxidative cyclization promoted by silver oxide. This novel approach not only simplifies the operational workflow by eliminating the need for gas handling but also broadens the practical applicability of the synthesis for diverse chemical libraries.
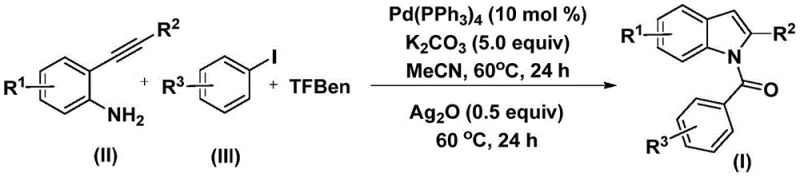
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
From a mechanistic perspective, this transformation is a sophisticated example of palladium-catalyzed C-N bond formation coupled with carbonyl insertion. The catalytic cycle initiates with the oxidative addition of the aryl iodide to the zero-valent palladium species, generating an aryl-palladium(II) intermediate. Subsequently, carbon monoxide, which is released in situ from the thermal decomposition of the TFBen additive, inserts into the palladium-carbon bond to form an acyl-palladium complex. This acyl species then undergoes nucleophilic attack by the amine group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide intermediate. The final crucial step involves the silver oxide-mediated intramolecular cyclization, which closes the indole ring and regenerates the active catalyst or facilitates the final aromatization. Understanding this mechanism is vital for R&D directors aiming to optimize purity and minimize side products, as the precise control of the CO release rate from TFBen is key to preventing homocoupling of the aryl iodide.
Impurity control in this system is inherently managed by the choice of reagents and the stepwise addition of silver oxide. The use of TFBen ensures a steady, low-concentration supply of CO, which favors the desired carbonyl insertion over competing pathways such as direct Suzuki-type coupling. Furthermore, the addition of silver oxide in the second stage acts as both an oxidant and a halide scavenger, driving the cyclization equilibrium forward and ensuring high conversion rates. The patent data indicates excellent functional group tolerance, accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing halogens like fluorine and chlorine on the aromatic rings. This robustness is evidenced by the successful synthesis of various derivatives, such as compounds I-1 through I-5, which demonstrate that the catalytic system remains active even with sterically hindered or electronically deactivated substrates.
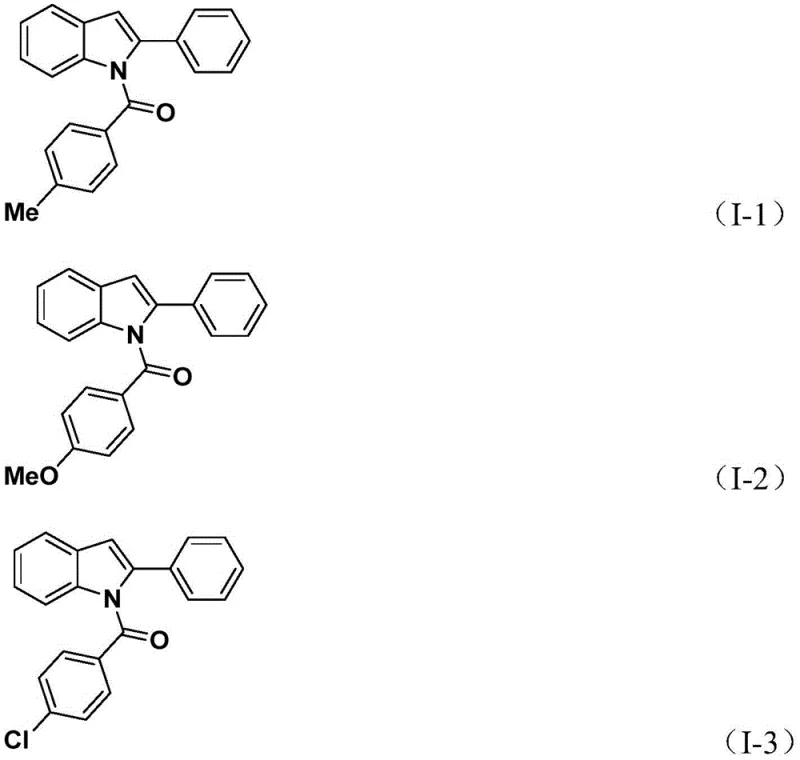
How to Synthesize N-Acylindole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The procedure typically begins by charging a reaction vessel with the palladium catalyst, base, solid CO source, and the two primary organic substrates in an acetonitrile solvent system. The mixture is heated to facilitate the initial coupling and carbonylation events before the oxidant is introduced to trigger the final ring closure. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical steps for achieving optimal yields.
- Combine palladium catalyst (Pd(PPh3)4), potassium carbonate, solid CO source (TFBen), 2-alkynyl aniline, and aryl iodide in acetonitrile solvent.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and coupling steps.
- Add silver oxide (Ag2O) to the mixture and continue heating at 60°C for another 24 hours to promote cyclization, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals evaluating this technology, the shift from gaseous CO to a solid surrogate like TFBen represents a significant logistical advantage that translates directly into cost reduction in pharmaceutical intermediate manufacturing. The elimination of high-pressure gas cylinders reduces the regulatory burden and insurance costs associated with hazardous material storage, while the use of common solvents like acetonitrile ensures compatibility with standard recovery and recycling systems. Moreover, the starting materials, specifically 2-alkynyl anilines and aryl iodides, are commodity chemicals that are readily available from multiple global suppliers, thereby reducing lead time for high-purity raw materials and mitigating the risk of single-source dependency. This supply chain resilience is critical for maintaining continuous production schedules in the fast-paced pharmaceutical industry.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction setup and the high atom efficiency of the carbonylation step. By avoiding the need for specialized autoclaves or high-pressure reactors, manufacturers can utilize standard glass-lined or stainless steel vessels, significantly lowering capital investment requirements. Additionally, the moderate reaction temperature of 60°C minimizes energy consumption compared to processes requiring reflux or cryogenic conditions. The high yields reported, reaching up to 82% for certain substrates, further enhance the cost-effectiveness by maximizing the output per batch and reducing the volume of waste solvent that requires treatment and disposal.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents rather than pressurized gases fundamentally improves the reliability of the supply chain. TFBen and silver oxide have long shelf lives and do not require special transportation permits, unlike carbon monoxide. This stability allows for bulk purchasing and inventory buffering, protecting the manufacturing schedule from short-term market fluctuations or delivery delays. Furthermore, the broad substrate scope means that the same catalytic platform can be adapted to produce a wide variety of N-acylindole analogues without needing to requalify entirely new reaction conditions, providing flexibility to respond quickly to changing customer demands for different API intermediates.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the homogeneous nature of the catalytic system and the absence of exothermic gas evolution hazards. The post-treatment process described involves simple filtration and column chromatography, which are unit operations well-understood and easily optimized in GMP facilities. From an environmental perspective, the use of a solid CO source prevents the venting of toxic gases, aligning with increasingly stringent environmental regulations. The ability to run the reaction in a one-pot fashion also reduces the number of isolation steps, thereby minimizing solvent usage and the overall environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for potential partners.
Q: What is the primary advantage of using TFBen over carbon monoxide gas in this synthesis?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute eliminates the safety hazards and specialized equipment required for handling toxic CO gas, significantly simplifying the operational procedure and enhancing workplace safety.
Q: What is the typical yield range for this N-acylindole preparation method?
A: According to the experimental data in patent CN112898192A, the reaction demonstrates high efficiency with isolated yields ranging from 44% to 82% across various substrates, indicating robust performance even with different electronic substituents.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process utilizes mild reaction conditions (60°C), commercially available starting materials, and a straightforward two-step one-pot procedure, making it highly amenable to commercial scale-up for producing complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acylindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has extensively evaluated the technology disclosed in CN112898192A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to commercial supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-acylindole intermediate we deliver meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a secure and efficient supply chain for your critical pharmaceutical intermediates.
