Advanced Metal-Free Synthesis of Sulfonylacetonitriles: A Breakthrough for Scalable API Intermediate Manufacturing
Advanced Metal-Free Synthesis of Sulfonylacetonitriles: A Breakthrough for Scalable API Intermediate Manufacturing
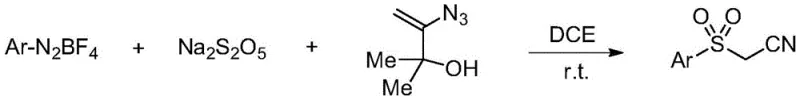
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the urgent need for greener, safer, and more cost-effective methodologies. A pivotal advancement in this domain is documented in Chinese Patent CN111517904A, which discloses a highly efficient preparation method for sulfonylacetonitrile compounds. These compounds serve as critical building blocks in the construction of complex bioactive molecules, including UT-B inhibitors and various agrochemical agents. The patented technology distinguishes itself by employing a metal-free radical addition strategy that utilizes aryl diazonium salts and sodium metabisulfite as key reagents. This approach fundamentally shifts the paradigm away from traditional, hazardous sulfonylation methods, offering a robust pathway for the production of high-purity API intermediates. By leveraging in situ generated arylsulfonyl radicals, this process achieves excellent functional group tolerance under remarkably mild conditions, positioning it as a superior choice for modern chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonylacetonitrile derivatives has relied heavily on the use of sulfonyl chlorides or strong sulfonic acids as the primary sulfur sources. These traditional reagents present significant challenges in an industrial setting, primarily due to their corrosive nature, high toxicity, and the generation of substantial acidic waste streams. Furthermore, conventional protocols often necessitate the use of transition metal catalysts or harsh oxidants to drive the reaction forward, which introduces complex downstream processing requirements. The removal of trace metal residues to meet stringent pharmaceutical purity standards adds considerable time and cost to the manufacturing process. Additionally, many classical methods require elevated temperatures or strictly anhydrous conditions, limiting their operational window and increasing energy consumption. These factors collectively create a bottleneck for the scalable production of sulfonyl-containing fine chemicals, prompting the industry to seek alternative, more sustainable synthetic routes.
The Novel Approach
In stark contrast to legacy techniques, the methodology outlined in CN111517904A introduces a transformative strategy that circumvents the use of toxic sulfonyl chlorides entirely. By utilizing sodium metabisulfite as a solid, easy-to-handle surrogate for sulfur dioxide, the process enables the in situ generation of arylsulfonyl radicals directly from aryl diazonium salts. This radical species then undergoes a selective addition reaction with 3-azido-2-methylbut-3-en-2-ol, followed by a unique fragmentation sequence that installs the nitrile functionality. The entire transformation proceeds at room temperature in 1,2-dichloroethane, eliminating the need for external heating or cooling systems. This metal-free protocol not only simplifies the reaction setup but also drastically reduces the environmental footprint by avoiding heavy metal contamination. The result is a streamlined, high-yielding process that delivers sulfonylacetonitrile compounds with exceptional purity, making it ideally suited for the demands of modern pharmaceutical supply chains.
Mechanistic Insights into Metal-Free Radical Sulfonylation
The core innovation of this technology lies in its elegant radical mechanism, which bypasses the need for exogenous catalysts. The reaction initiates with the interaction between the aryl diazonium tetrafluoroborate and sodium metabisulfite, leading to the homolytic cleavage that generates the reactive arylsulfonyl radical species. This radical intermediate is highly electrophilic and selectively attacks the electron-rich double bond of the azido-alkene substrate. Following the initial addition, the resulting carbon-centered radical undergoes a rapid beta-scission process, expelling a molecule of nitrogen gas and a hydroxyisopropyl radical. This fragmentation step is thermodynamically driven and serves as the driving force for the formation of the final carbon-sulfur bond. The single-electron transfer processes involved are finely balanced to ensure high conversion rates without the accumulation of unwanted byproducts. Understanding this mechanistic pathway is crucial for optimizing reaction parameters and ensuring consistent quality in large-scale batches.
Furthermore, the impurity profile of this reaction is exceptionally clean due to the specificity of the radical addition and fragmentation steps. Unlike transition-metal catalyzed cross-couplings which often suffer from homocoupling side reactions or incomplete conversions, this radical pathway demonstrates high chemoselectivity. The use of sodium metabisulfite ensures that sulfur dioxide is released in a controlled manner, preventing the formation of polysulfonated impurities that can plague other sulfonylation methods. The stability of the aryl diazonium salt precursors allows for precise stoichiometric control, further minimizing the presence of unreacted starting materials in the crude mixture. For R&D teams focused on impurity control, this mechanism offers a predictable and manageable profile, facilitating easier purification via standard column chromatography. The ability to tolerate diverse electronic environments on the aromatic ring—from electron-deficient halides to electron-rich alkoxy groups—underscores the robustness of this radical manifold.
How to Synthesize Sulfonylacetonitrile Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational protocols to maximize yield and safety. The process begins with the careful preparation of the reaction vessel under an inert atmosphere to prevent premature oxidation of the radical intermediates. Reagents are added sequentially to ensure proper mixing and heat dissipation, although the reaction is exothermic enough to proceed spontaneously at ambient temperatures. Monitoring the reaction progress via thin-layer chromatography (TLC) is essential to determine the optimal endpoint, typically achieved within a few hours depending on the substrate electronics. Once completion is confirmed, the workup procedure is straightforward, involving simple concentration and purification steps that do not require specialized equipment. For a detailed, step-by-step guide on executing this synthesis with precision, please refer to the standardized protocol below.
- Mix aryl diazonium salt, sodium metabisulfite, and 3-azido-2-methylbut-3-en-2-ol in an inert atmosphere.
- Add 1,2-dichloroethane (DCE) solvent and stir the mixture at room temperature until TLC indicates completion.
- Concentrate the reaction mixture under reduced pressure and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that directly address the pain points of procurement managers and supply chain directors. The elimination of transition metal catalysts represents a significant cost saving, as it removes the necessity for expensive ligands and the subsequent rigorous purification steps required to meet residual metal specifications. Moreover, the reliance on commodity chemicals like sodium metabisulfite and aryl diazonium salts ensures a stable and reliable supply of raw materials, mitigating the risk of shortages associated with exotic reagents. The mild reaction conditions translate to lower energy costs and reduced wear on reactor equipment, enhancing the overall economic viability of the process. These factors combine to create a manufacturing route that is not only technically superior but also commercially resilient in a volatile market.
- Cost Reduction in Manufacturing: The absence of precious metal catalysts such as palladium or copper significantly lowers the direct material costs associated with the synthesis. Additionally, the simplified workup procedure reduces solvent consumption and labor hours required for purification, leading to substantial operational savings. By avoiding the use of hazardous sulfonyl chlorides, the facility also saves on costs related to specialized corrosion-resistant equipment and hazardous waste disposal. The overall process efficiency allows for higher throughput without proportional increases in overhead, driving down the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The key reagents utilized in this protocol, including sodium metabisulfite and various aryl diazonium salts, are widely available from multiple global suppliers, ensuring supply continuity. This diversification of the supply base reduces dependency on single-source vendors and minimizes the risk of production delays due to raw material scarcity. The stability of the starting materials allows for bulk purchasing and long-term storage, further securing the supply chain against market fluctuations. Consequently, manufacturers can maintain consistent production schedules and meet delivery commitments with greater confidence.
- Scalability and Environmental Compliance: The metal-free nature of this reaction aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge. Scaling this process from gram to ton scale does not introduce new safety hazards, as the reaction operates at room temperature and atmospheric pressure. The generation of nitrogen gas as a byproduct is benign and easily managed, avoiding the release of toxic sulfur oxides common in other sulfonylation routes. This environmental compatibility facilitates smoother regulatory approvals and supports the company's sustainability goals, making it an attractive option for green chemistry initiatives.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the practical application and scope of this synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent literature. Addressing these questions helps clarify the operational boundaries and potential integration of this route into existing manufacturing workflows. We encourage stakeholders to review these details to fully appreciate the versatility and robustness of the described protocol.
Q: Does this synthesis method require toxic sulfonyl chlorides?
A: No, this novel method avoids the use of hazardous sulfonyl chlorides or strong sulfonic acids by utilizing sodium metabisulfite as a safe sulfur dioxide surrogate.
Q: Is a transition metal catalyst needed for this radical reaction?
A: No, the process is completely metal-free, operating via a radical mechanism initiated by aryl diazonium salts, which eliminates the need for expensive catalyst removal steps.
Q: What is the substrate scope for the aryl group in this reaction?
A: The method tolerates a wide range of substituents, including electron-withdrawing groups like halogens and esters, as well as electron-donating alkyl and alkoxy groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonylacetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free radical sulfonylation technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of sulfonylacetonitrile delivered meets the highest quality standards required by global regulatory bodies. We are committed to leveraging our technical expertise to optimize this route for maximum efficiency and yield.
We invite you to collaborate with us to explore how this advanced synthesis method can enhance your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
