Advanced Metal-Free Synthesis of Sulfonylacetonitrile Intermediates for Commercial Scale-Up
Advanced Metal-Free Synthesis of Sulfonylacetonitrile Intermediates for Commercial Scale-Up
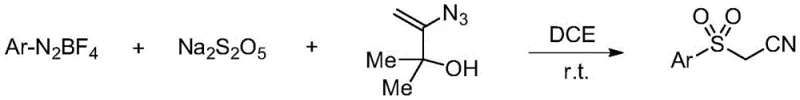
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for constructing complex molecular scaffolds, particularly those containing sulfonyl functionalities which are ubiquitous in bioactive molecules. A significant breakthrough in this domain is documented in patent CN111517904B, which discloses a novel preparation method for sulfonylacetonitrile compounds that fundamentally shifts the paradigm from traditional harsh synthetic routes to a mild, metal-free radical process. This innovation leverages the in situ generation of arylsulfonyl radicals from aryl diazonium salts and sodium metabisulfite, enabling the direct functionalization of alkenes without the need for transition metal catalysts or extreme thermal conditions. For R&D directors and procurement managers alike, this represents a critical opportunity to streamline supply chains for high-purity pharmaceutical intermediates while drastically reducing the environmental footprint associated with heavy metal waste disposal and hazardous reagent handling. The versatility of this method allows for a broad substrate scope, accommodating various electron-withdrawing and electron-donating groups, thereby ensuring its applicability across a wide range of drug discovery and development programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonylacetonitrile compounds has relied heavily on the use of sulfonyl chlorides or sulfonic acids as the primary sulfur sources, which present significant challenges in terms of safety, cost, and operational complexity. These traditional reagents are often highly corrosive, toxic, and moisture-sensitive, requiring specialized equipment and rigorous safety protocols that inflate the overall cost of manufacturing and increase the risk of workplace accidents. Furthermore, conventional methods frequently necessitate the use of stoichiometric amounts of strong oxidants or transition metal catalysts to drive the reaction forward, leading to complicated downstream processing steps to remove residual metal impurities that could compromise the purity of the final active pharmaceutical ingredient. The harsh reaction conditions often associated with these legacy processes, such as high temperatures or strong acidic environments, can also lead to the decomposition of sensitive functional groups, limiting the scope of substrates that can be effectively utilized and resulting in lower overall yields and higher waste generation.
The Novel Approach
In stark contrast to these legacy methodologies, the novel approach outlined in the patent utilizes a radical addition strategy that operates under exceptionally mild conditions, specifically at room temperature in a common organic solvent like 1,2-dichloroethane. By employing sodium metabisulfite as a solid surrogate for sulfur dioxide, the process eliminates the need for handling gaseous SO2, which is hazardous and difficult to control on a large scale, while simultaneously avoiding the use of any metal catalysts that could contaminate the product stream. This metal-free protocol not only simplifies the reaction setup by removing the need for inert atmosphere gloveboxes or specialized pressure vessels but also significantly enhances the safety profile of the manufacturing process, making it more attractive for cost reduction in fine chemical manufacturing. The direct generation of arylsulfonyl radicals allows for a highly efficient one-step construction of the sulfonyl group, bypassing multiple synthetic steps and purification stages that are typically required in conventional routes, thereby improving the overall atom economy and reducing the time to market for new drug candidates.
Mechanistic Insights into Radical-Mediated Sulfonylation
The core of this technological advancement lies in the intricate radical mechanism that facilitates the formation of the carbon-sulfur bond without the assistance of transition metals. The process initiates with the reaction between the aryl diazonium salt and sodium metabisulfite, which generates an arylsulfonyl radical species in situ under ambient conditions. This highly reactive radical intermediate then undergoes a selective addition to the double bond of 3-azido-2-methylbut-3-en-2-ol, forming a new carbon-centered radical that is stabilized by the adjacent functional groups. Subsequent fragmentation of this intermediate releases a molecule of nitrogen gas and a 2-hydroxypropyl radical, driving the reaction equilibrium forward and yielding the desired sulfonylacetonitrile product with high selectivity. This mechanism is particularly advantageous because it avoids the formation of stable metal-complex intermediates that often require harsh conditions to break down, thus preserving the integrity of sensitive functional groups on the aromatic ring.
From an impurity control perspective, this radical pathway offers distinct advantages over ionic mechanisms that are prone to side reactions such as over-oxidation or hydrolysis. The mild reaction conditions minimize the thermal degradation of reagents and products, ensuring that the impurity profile remains clean and manageable throughout the synthesis. Since no metal catalysts are involved, there is no risk of metal leaching into the final product, which is a critical quality attribute for pharmaceutical intermediates intended for human consumption. The use of sodium metabisulfite as a sulfur source also ensures that the byproducts are largely inorganic salts that can be easily removed during the aqueous workup or column chromatography purification steps. This high level of purity reduces the burden on downstream processing teams and ensures that the final material meets the stringent specifications required by regulatory agencies, thereby facilitating a smoother path through clinical trials and commercial approval processes.
How to Synthesize Sulfonylacetonitrile Efficiently
To implement this synthesis route effectively in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that maximize yield and safety while minimizing waste. The process begins with the careful preparation of the reaction mixture under an inert atmosphere to prevent unwanted oxidation of the radical intermediates by atmospheric oxygen. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and consistency across different batches and scales of production.
- Mix aryl diazonium salt, sodium metabisulfite, and 3-azido-2-methylbut-3-en-2-ol in 1,2-dichloroethane under inert atmosphere.
- Stir the reaction mixture at room temperature until TLC monitoring indicates complete conversion of starting materials.
- Concentrate the reaction solution under reduced pressure and purify the crude product via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route translates into tangible strategic benefits that extend far beyond the laboratory bench. The elimination of expensive transition metal catalysts and the use of commodity chemicals like sodium metabisulfite significantly lower the raw material costs, which is a primary driver for cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the simplicity of the workup procedure, which involves direct concentration and column chromatography, reduces the consumption of solvents and energy, contributing to a more sustainable and economically viable production model. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality or environmental factors, ensuring a more reliable supply of critical intermediates for downstream drug synthesis.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts from the synthesis protocol eliminates the need for costly metal scavenging resins and specialized filtration equipment, which are often required to meet strict residual metal limits in pharmaceutical products. Additionally, the use of sodium metabisulfite as a sulfur source is significantly more economical than purchasing pre-functionalized sulfonyl chlorides, which often carry a high price premium due to their complex synthesis and hazardous nature. The mild reaction conditions also reduce energy consumption by eliminating the need for heating or cooling systems, further driving down the operational expenditures associated with large-scale production. These cumulative savings allow for a more competitive pricing structure for the final intermediate, enhancing the margin potential for the entire drug development value chain.
- Enhanced Supply Chain Reliability: The raw materials required for this process, such as aryl diazonium salts and sodium metabisulfite, are widely available from multiple global suppliers, reducing the risk of supply disruptions caused by single-source dependencies. The stability of these reagents under standard storage conditions simplifies logistics and inventory management, allowing for longer shelf lives and reduced waste due to expiration. Moreover, the room temperature operation of the reaction reduces the dependency on specialized utility infrastructure, such as high-capacity chillers or steam lines, making it easier to transfer the technology between different manufacturing sites or contract manufacturing organizations. This flexibility ensures that production can be scaled up or shifted geographically with minimal friction, securing the continuity of supply for critical drug programs.
- Scalability and Environmental Compliance: The absence of toxic gases like sulfur dioxide and the use of solid sulfur surrogates make this process inherently safer and easier to scale from gram to ton quantities without significant engineering redesign. The reduced generation of hazardous waste streams aligns with increasingly stringent environmental regulations, minimizing the costs associated with waste disposal and environmental compliance reporting. The high atom economy of the radical addition reaction ensures that a larger proportion of the starting materials are incorporated into the final product, reducing the overall material intensity of the process. This green chemistry profile not only enhances the corporate sustainability image but also future-proofs the manufacturing process against evolving regulatory landscapes regarding chemical safety and environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the detailed data provided in the patent documentation. These answers are designed to clarify the operational parameters and potential benefits for stakeholders evaluating this route for their specific supply chain needs.
Q: What are the advantages of this metal-free synthesis method?
A: This method avoids the use of toxic sulfonyl chlorides and expensive metal catalysts, operating under mild room temperature conditions with high product purity and easy separation.
Q: What solvents are compatible with this reaction protocol?
A: 1,2-dichloroethane (DCE) is the preferred solvent for optimal yield, while dichloromethane, acetonitrile, and tetrahydrofuran can be used as alternatives with potentially reduced efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of readily available raw materials like sodium metabisulfite and the absence of harsh conditions make this process highly scalable and environmentally compliant for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonylacetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route and have integrated similar advanced methodologies into our CDMO service offerings to support global pharmaceutical innovation. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of sulfonylacetonitrile intermediates meets the highest quality standards required for drug substance synthesis. Our commitment to technical excellence allows us to adapt this patent-protected chemistry to meet the specific volume and timeline requirements of our partners, providing a secure and scalable source for your critical building blocks.
We invite you to engage with our technical procurement team to discuss how this novel synthesis method can be leveraged to optimize your current supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this metal-free route for your specific target molecules. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your drug development programs. Let us partner with you to bring your next generation of therapeutics to market faster and more sustainably.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
