Advanced Synthesis of Polysubstituted Naphthalene Derivatives via HDDA Reaction for Commercial Scale-up
Advanced Synthesis of Polysubstituted Naphthalene Derivatives via HDDA Reaction for Commercial Scale-up
The landscape of organic synthesis for complex aromatic systems is undergoing a significant transformation, driven by the need for more efficient and atom-economical pathways to access highly functionalized scaffolds. Patent CN110028409B introduces a groundbreaking methodology for the preparation of polysubstituted naphthalene derivatives, utilizing a sophisticated Hexadehydro-Diels-Alder (HDDA) reaction strategy. This technology represents a paradigm shift from traditional electrophilic aromatic substitution methods, offering a robust route to generate diverse molecular architectures that are critical for the development of next-generation active pharmaceutical ingredients (APIs) and advanced agrochemical intermediates. By leveraging a tetraalkyne compound and para-substituted phenylacetylene as key building blocks, this process achieves high structural complexity under relatively mild thermal conditions, thereby addressing long-standing challenges in regioselectivity and functional group tolerance.
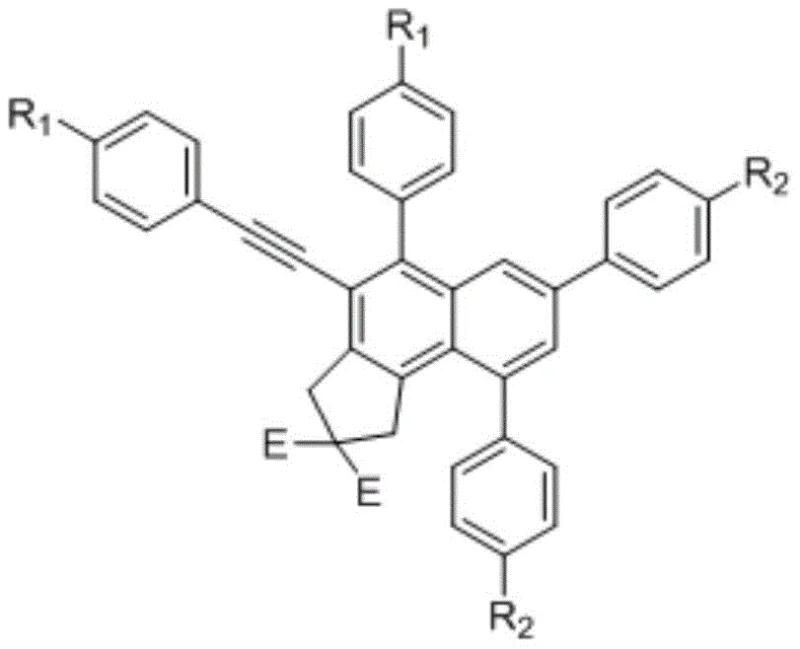
For procurement specialists and supply chain managers evaluating reliable polysubstituted naphthalene suppliers, this patent data underscores a viable pathway for cost reduction in organic synthesis manufacturing. The ability to construct the naphthalene core de novo from acyclic precursors allows for precise control over substitution patterns, which is often difficult to achieve with native naphthalene feedstocks. This level of precision translates directly into value for R&D directors who require high-purity OLED material or pharmaceutical intermediates with specific electronic or steric properties. The commercial implications are profound, as this method simplifies the supply chain by reducing the number of synthetic steps required to reach complex targets, ultimately enhancing the overall efficiency of the production lifecycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for functionalizing naphthalene rings have historically relied on classical electrophilic aromatic substitution reactions, such as sulfonation, nitration, and halogenation, which are fraught with significant limitations regarding regiocontrol and environmental impact. These conventional processes often yield complex mixtures of isomers that require energy-intensive and solvent-heavy separation techniques to isolate the desired product, leading to substantial material loss and increased operational costs. Furthermore, the harsh reaction conditions typically associated with these transformations, including the use of strong acids and oxidizers, pose safety risks and generate considerable hazardous waste, complicating compliance with increasingly stringent environmental regulations. The inability to easily introduce diverse functional groups at specific positions on the naphthalene core restricts the chemical space available to medicinal chemists, often forcing them to accept suboptimal lead candidates due to synthetic inaccessibility.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a cascade cyclization strategy initiated by the thermal activation of a tetrayne precursor, which undergoes an intramolecular HDDA reaction to generate a highly reactive benzyne intermediate in situ. This transient species is then immediately trapped by an external alkyne partner, specifically a para-substituted phenylacetylene, to forge the polysubstituted naphthalene skeleton with exceptional precision. This methodology bypasses the need for pre-functionalized naphthalene starting materials and avoids the use of transition metal catalysts in the final cyclization step, significantly simplifying the workup procedure and reducing the risk of metal contamination in the final product. The result is a streamlined synthetic route that offers superior atom economy and the flexibility to incorporate a wide range of substituents, enabling the rapid generation of diverse libraries for biological screening.
Mechanistic Insights into Hexadehydro-Diels-Alder (HDDA) Cyclization
The core of this innovative synthesis lies in the pericyclic nature of the Hexadehydro-Diels-Alder reaction, a [4+2] cycloaddition involving three alkyne units that proceeds through a concerted mechanism to form a benzyne intermediate. Under thermal conditions ranging from 100°C to 110°C in a toluene solvent, the tetrayne precursor undergoes a conformational change that aligns the reactive pi-systems, allowing for the formation of new sigma bonds and the expulsion of aromaticity in a controlled manner. This generates a highly strained aryne species that acts as a powerful dienophile or dipolarophile, ready to react with the added phenylacetylene trap. The elegance of this mechanism is its ability to construct two new carbon-carbon bonds and a fully aromatic ring system in a single operation, demonstrating high step efficiency that is rarely seen in traditional stepwise aromatic substitutions.
From an impurity control perspective, the mechanism inherently limits the formation of side products because the benzyne intermediate is extremely short-lived and reacts rapidly with the abundant alkyne trap present in the reaction mixture. This kinetic control ensures that the primary pathway dominates, minimizing the formation of oligomeric byproducts or alternative cyclization isomers that often plague radical-based aromatic couplings. The subsequent nucleophilic addition and aromatization steps proceed smoothly to yield the stable polysubstituted naphthalene derivative, which can be isolated as a high-purity white powder after simple crystallization. This mechanistic robustness is crucial for commercial scale-up of complex polymer additives or electronic chemicals, where batch-to-b一致性 consistency is paramount.
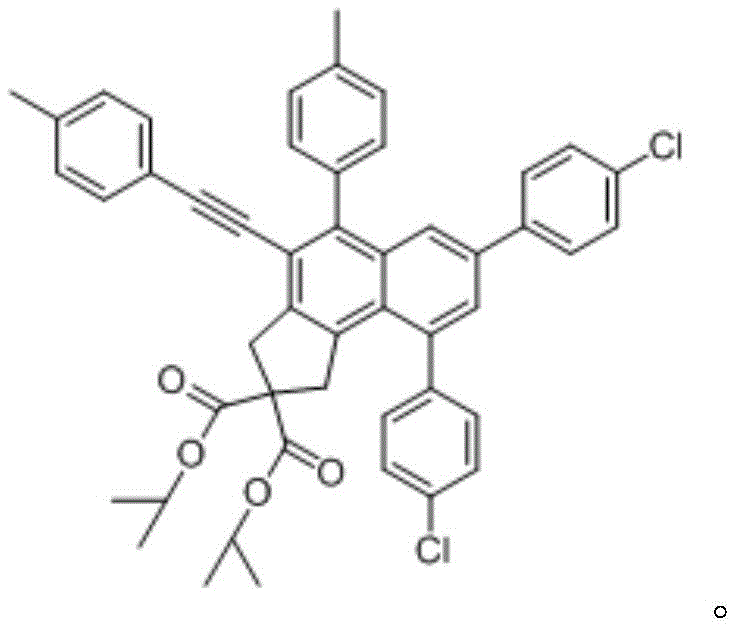
How to Synthesize Polysubstituted Naphthalene Derivatives Efficiently
The practical execution of this synthesis involves a multi-step sequence beginning with the preparation of the tetrayne precursor via alkylation and Sonogashira coupling, followed by the key thermal cyclization event. The process is designed to be operationally simple, utilizing standard laboratory glassware and commercially available reagents, which facilitates easy technology transfer from R&D to pilot plant operations. The reaction conditions are optimized to balance reaction rate with product stability, ensuring high yields while maintaining safety standards suitable for large-scale manufacturing. For detailed procedural specifics regarding stoichiometry, temperature ramps, and workup protocols, please refer to the standardized synthesis guide below.
- Preparation of Tetrayne Precursor: React diisopropyl malonate with propargyl bromide using sodium hydride in acetonitrile, followed by Sonogashira coupling with p-substituted phenylethynyl bromide.
- Thermal Cyclization (HDDA Reaction): Heat the tetrayne compound with para-substituted phenylacetylene in toluene solvent at 100-110°C for 12-14 hours to induce benzyne formation and trapping.
- Purification: Extract the reaction mixture with ethyl acetate and water, concentrate the organic phase, and crystallize the product using a petroleum ether and ethyl acetate system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this HDDA-based synthesis route offers compelling strategic advantages that extend beyond mere technical novelty. By shifting the synthesis paradigm from modification of existing aromatics to de novo construction, manufacturers can decouple their supply chains from the volatile pricing and availability fluctuations of specialized naphthalene derivatives. The reliance on commodity chemicals such as diisopropyl malonate, propargyl bromide, and simple phenylacetylenes ensures a stable and cost-effective raw material base, significantly mitigating supply risk. Furthermore, the elimination of transition metal catalysts in the final cyclization step removes the need for expensive scavenging resins and extensive metal testing, directly contributing to cost reduction in organic synthesis manufacturing.
- Cost Reduction in Manufacturing: The streamlined nature of the HDDA reaction eliminates multiple protection and deprotection steps often required in traditional aromatic functionalization, leading to a drastic simplification of the overall process flow. By avoiding the use of precious metal catalysts in the key ring-forming step, the process removes a major cost driver associated with catalyst recovery and residual metal analysis, resulting in substantial cost savings. Additionally, the high atom economy of the cycloaddition means that a greater proportion of the starting mass is incorporated into the final product, reducing waste disposal costs and improving the overall material efficiency of the production line.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are bulk commodities with well-established global supply networks, ensuring that production schedules are not held hostage by the scarcity of exotic reagents. The robustness of the thermal cyclization step allows for flexible manufacturing windows, as the reaction tolerates minor variations in heating rates without compromising product quality, thereby enhancing supply chain resilience. This reliability is critical for maintaining continuous production of high-purity OLED material or pharmaceutical intermediates, where interruptions can have cascading effects on downstream drug development timelines.
- Scalability and Environmental Compliance: The use of toluene as a solvent and moderate temperatures (100-110°C) makes this process highly scalable using standard stainless steel reactors without the need for specialized high-pressure or cryogenic equipment. The simplified workup procedure, which relies on standard extraction and crystallization techniques, minimizes the generation of complex aqueous waste streams, facilitating easier compliance with environmental discharge regulations. This environmental compatibility, combined with the inherent safety of avoiding energetic reagents, positions this technology as a sustainable choice for the commercial scale-up of complex polymer additives and fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polysubstituted naphthalene synthesis technology. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent literature, offering a transparent view of the process capabilities. Understanding these details is essential for partners looking to integrate this chemistry into their existing manufacturing portfolios or R&D pipelines.
Q: What are the key advantages of the HDDA reaction route for naphthalene synthesis?
A: The HDDA reaction offers superior atom economy and avoids the use of harsh sulfonation or nitration conditions typical of traditional naphthalene functionalization, resulting in cleaner reaction profiles and easier purification.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes common solvents like toluene and operates at moderate temperatures (100-110°C), making it highly amenable to scale-up from kilogram to multi-ton production without requiring specialized high-pressure equipment.
Q: What is the purity profile of the resulting derivatives?
A: The method yields white powdery products with high structural integrity, as confirmed by NMR spectroscopy, and the crystallization step effectively removes impurities, ensuring specifications suitable for pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the HDDA reaction technology for producing high-value aromatic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of polysubstituted naphthalene derivative meets the exacting standards required for pharmaceutical and electronic applications.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this methodology can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for advanced chemical intermediates.
