Advanced Synthesis of Polysubstituted Naphthalene Derivatives via HDDA Reaction for Commercial Scale-Up
Advanced Synthesis of Polysubstituted Naphthalene Derivatives via HDDA Reaction for Commercial Scale-Up
The landscape of organic synthesis for complex aromatic systems is undergoing a significant transformation, driven by the need for more efficient and atom-economical pathways. Patent CN110028409B introduces a groundbreaking methodology for the preparation of polysubstituted naphthalene derivatives, utilizing a sophisticated Hexadehydro-Diels-Alder (HDDA) reaction strategy. This technology moves beyond the limitations of classical electrophilic aromatic substitution, enabling the construction of highly functionalized naphthalene cores from acyclic precursors with exceptional precision. The disclosed method employs a tetraalkyne compound and para-substituted phenylacetylene as primary raw materials, reacting them in a toluene solvent system at controlled temperatures between 100-110 ℃ for a duration of 12-14 hours. This thermal cyclization process not only simplifies the synthetic route but also generates a series of novel derivatives with complex and diversified structures that were previously difficult to access. For R&D teams seeking reliable pharmaceutical intermediate supplier partnerships, this patent represents a critical advancement in accessing privileged scaffolds for medicinal chemistry.
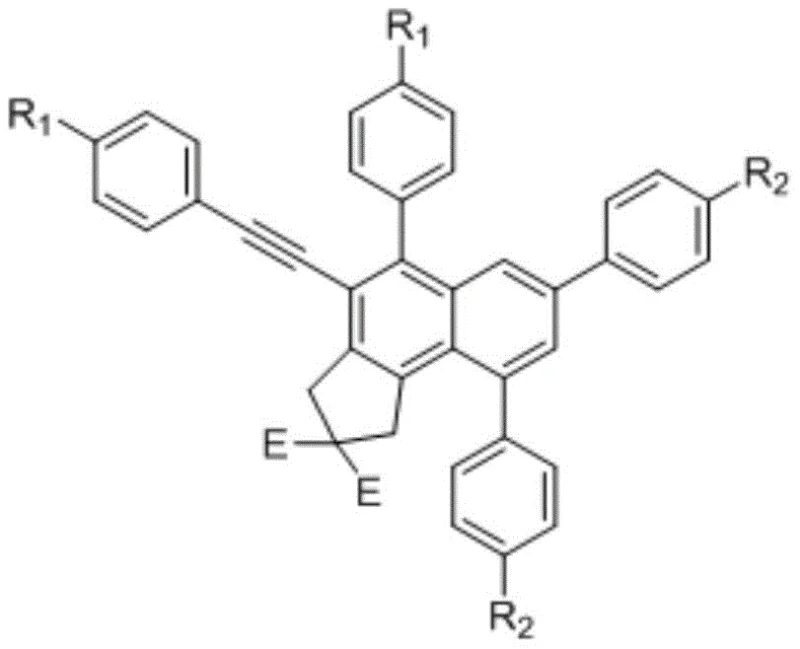
The versatility of this synthetic platform is evident in the general structural formula, where substituents such as esters, halogens, and alkyl groups can be precisely positioned on the aromatic core. This level of structural control is paramount for optimizing the biological activity or material properties of the final compound. By leveraging this patented technology, manufacturers can produce high-purity polysubstituted naphthalene derivatives that serve as vital building blocks for next-generation agrochemicals, dyes, and pharmaceutical agents. The ability to tune the electronic and steric properties of the naphthalene ring through simple variation of the starting alkynes opens up vast possibilities for structure-activity relationship (SAR) studies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of naphthalene has relied heavily on traditional electrophilic aromatic substitution reactions, such as sulfonation, nitration, and halogenation. These classical methods are often plagued by inherent issues regarding regioselectivity, frequently yielding mixtures of isomers that require tedious and costly separation processes. Furthermore, the harsh reaction conditions typically associated with these transformations, including the use of strong acids and oxidizing agents, pose significant safety risks and generate substantial amounts of hazardous waste. From a supply chain perspective, the low atom economy of these multi-step functionalization sequences results in higher raw material consumption and increased disposal costs. Additionally, introducing complex substituents onto a pre-formed naphthalene ring often requires protecting group strategies that further elongate the synthetic timeline and reduce overall yield. These inefficiencies create bottlenecks in the commercial scale-up of complex organic intermediates, limiting the speed at which new candidates can be brought to market.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a cascade cyclization strategy that constructs the naphthalene framework de novo from simpler alkyne precursors. By employing a tetraalkyne compound as a key intermediate, the process harnesses the power of the HDDA reaction to generate a reactive benzyne species in situ, which is subsequently trapped by a phenylacetylene partner. This convergent synthesis allows for the rapid assembly of molecular complexity in a single operational step, significantly reducing the number of unit operations required. The reaction conditions are remarkably mild, proceeding in toluene at 100-110 ℃, which eliminates the need for extreme temperatures or pressures often seen in traditional cyclizations. This methodological shift not only improves the overall yield but also enhances the purity profile of the final product by minimizing side reactions associated with harsh reagents. For procurement managers focused on cost reduction in fine chemical manufacturing, this streamlined approach offers a compelling value proposition through reduced processing time and simplified downstream purification.
Mechanistic Insights into HDDA-Mediated Cyclization
The core of this innovative synthesis lies in the Hexadehydro-Diels-Alder (HDDA) reaction, a powerful pericyclic transformation that converts a 1,3-diyne and a dienophile into a benzyne intermediate. In this specific application, the tetraalkyne precursor serves as the source of the 1,3-diyne moiety, which undergoes thermal cyclization to form a highly reactive aryne species. This transient intermediate is then intercepted by the para-substituted phenylacetylene acting as a trapping agent, leading to the formation of the polysubstituted naphthalene skeleton. The mechanism involves a concerted [4+2] cycloaddition followed by aromatization, a process that is thermodynamically driven by the formation of the stable aromatic system. Understanding this mechanistic pathway is crucial for R&D directors, as it highlights the importance of maintaining strict anhydrous conditions and precise temperature control to ensure the efficient generation of the benzyne intermediate without premature decomposition.
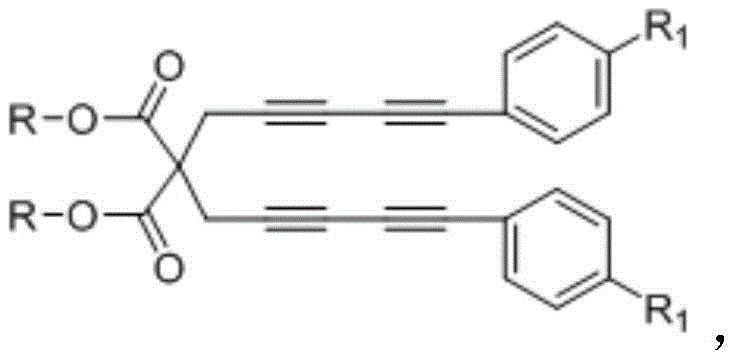
Impurity control in this process is inherently managed by the high specificity of the HDDA reaction and the subsequent nucleophilic addition step. Unlike electrophilic substitutions that can occur at multiple positions on the ring, the cyclization pathway dictates the position of the new substituents based on the connectivity of the starting alkynes. The use of para-substituted phenylacetylenes ensures that the resulting naphthalene derivative possesses a defined substitution pattern, minimizing the formation of regioisomers. Furthermore, the reaction produces minimal byproducts, primarily consisting of unreacted starting materials which can be easily removed during the workup phase. The purification protocol described, involving extraction with water and ethyl acetate followed by crystallization from petroleum ether, effectively removes trace impurities and catalyst residues. This robust impurity profile is essential for meeting the stringent quality standards required for pharmaceutical intermediates, ensuring that the final API or active ingredient is free from genotoxic or persistent organic contaminants.
How to Synthesize Polysubstituted Naphthalene Efficiently
The synthesis of these high-value derivatives follows a logical three-step sequence that begins with the preparation of the tetrayne precursor, followed by the key thermal cyclization, and concludes with a straightforward purification protocol. The initial step involves the alkylation of diisopropyl malonate with propargyl bromide to establish the carbon backbone, followed by a Sonogashira coupling to install the necessary alkyne arms. This modular approach allows for the easy introduction of diverse functional groups early in the synthesis, providing flexibility for library generation. The critical cyclization step is performed in toluene, where the tetrayne and phenylacetylene are heated to induce the HDDA reaction, forming the core naphthalene structure with high efficiency. Detailed standardized synthetic steps see the guide below.
- Preparation of the tetrayne precursor via alkylation of diisopropyl malonate with propargyl bromide followed by Sonogashira coupling.
- Thermal cyclization of the tetrayne compound with para-substituted phenylacetylene in toluene at 100-110°C for 12-14 hours.
- Purification of the crude reaction mixture via extraction, concentration, and crystallization using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical novelty. The transition from multi-step functionalization to a convergent cyclization strategy fundamentally alters the cost structure of producing these complex intermediates. By reducing the number of synthetic steps and eliminating the need for expensive protecting groups or harsh reagents, the overall manufacturing cost is significantly optimized. The use of commodity chemicals such as toluene, ethyl acetate, and petroleum ether as solvents ensures that raw material sourcing remains stable and cost-effective, mitigating the risks associated with supply chain disruptions for exotic reagents. Moreover, the high atom economy of the HDDA reaction means that a greater proportion of the starting mass is incorporated into the final product, reducing waste disposal fees and environmental compliance burdens.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in the final cyclization step is a major driver for cost savings, as it removes the need for expensive palladium or copper removal processes that are often required in cross-coupling reactions. The thermal nature of the reaction allows for the use of standard heating equipment rather than specialized cryogenic or high-pressure reactors, lowering capital expenditure requirements. Additionally, the high yields reported in the examples (exceeding 80%) indicate a robust process that minimizes material loss, directly contributing to a lower cost of goods sold (COGS). The simplified workup procedure, which relies on basic extraction and crystallization, reduces labor hours and solvent consumption compared to complex chromatographic separations.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including diisopropyl malonate, propargyl bromide, and various substituted phenylacetylenes, are commercially available in bulk quantities from multiple global suppliers. This diversity in sourcing options prevents single-source dependency, ensuring a continuous supply of raw materials even during market fluctuations. The stability of the intermediates and the final product allows for flexible inventory management, enabling manufacturers to produce in larger batches and store stock safely without significant degradation. This reliability is critical for maintaining consistent delivery schedules to downstream pharmaceutical or agrochemical clients who depend on just-in-time manufacturing models.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of standard solvents and moderate reaction temperatures that translate easily from laboratory to pilot and production scales. The absence of highly toxic reagents or explosive intermediates simplifies the safety assessment and regulatory approval process for new manufacturing facilities. Furthermore, the reduced generation of hazardous waste aligns with modern green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The ability to recycle solvents like toluene and petroleum ether further enhances the sustainability profile of the operation, appealing to environmentally conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these polysubstituted naphthalene derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production pipelines or R&D programs.
Q: What are the advantages of the HDDA reaction method over traditional naphthalene functionalization?
A: Traditional methods often rely on harsh sulfonation or nitration of pre-existing naphthalene rings, which suffer from poor regioselectivity and generate significant acidic waste. The HDDA (Hexadehydro-Diels-Alder) approach constructs the naphthalene core de novo under mild thermal conditions, offering superior atom economy and precise control over substitution patterns.
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Yes, the process utilizes common solvents like toluene and acetonitrile and operates at moderate temperatures (100-110°C), avoiding cryogenic conditions or ultra-high pressures. The workup involves standard extraction and crystallization techniques, making it highly amenable to kilogram-to-ton scale production.
Q: What represents the key structural feature of these derivatives?
A: The core structure features a highly substituted naphthalene backbone fused with a cyclopentane ring bearing ester groups. This unique architecture provides multiple handles for further functionalization, making it a versatile scaffold for drug discovery and material science applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Naphthalene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the HDDA-mediated synthesis pathway for generating complex aromatic scaffolds. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this process, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required for pharmaceutical and fine chemical applications. We are committed to delivering high-quality intermediates that empower your innovation.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain with reliable, high-performance chemical solutions.
