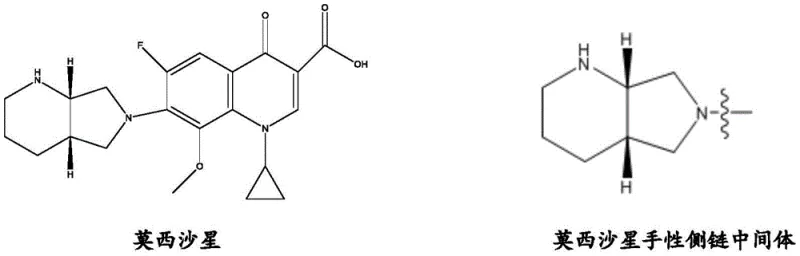
Advanced Eight-Step Synthesis of Moxifloxacin Chiral Side Chain Intermediate for Commercial Scale-up
The pharmaceutical industry is constantly seeking more efficient pathways for the production of fourth-generation quinolone antibiotics, specifically targeting the complex chiral side chains that define their efficacy. Patent CN110218210B introduces a groundbreaking methodology for synthesizing the moxifloxacin chiral side chain intermediate, fundamentally shifting the paradigm from expensive, resolution-dependent routes to a streamlined chiral pool approach. This novel process utilizes L-asparagine, a ubiquitous and cost-effective amino acid, as the foundational building block, thereby embedding the necessary stereochemistry at the very inception of the synthesis. By navigating through an meticulously designed eight-step reaction sequence, this technology successfully circumvents the traditional bottlenecks associated with chiral resolution, which typically result in a theoretical maximum yield of only 50% for the desired enantiomer. The strategic implementation of catalytic hydrogenation and selective protection groups ensures high optical purity without the need for downstream separation of racemic mixtures. For R&D directors and process chemists, this represents a significant leap forward in atom economy and process mass intensity, offering a robust alternative to legacy methods that rely on scarce or costly starting materials like 2,3-dipicolinic acid.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of moxifloxacin intermediates has been plagued by economic and technical inefficiencies inherent to the prevailing synthetic strategies. Traditional routes often commence with 2,3-dipicolinic acid, a substrate that not only commands a premium price in the global chemical market but also necessitates a cumbersome series of acid anhydrization and ammonolysis reactions. Furthermore, these conventional pathways invariably require a chiral resolution step, frequently employing tartaric acid or similar resolving agents, to isolate the biologically active enantiomer from the racemic mixture. This resolution process is inherently wasteful, discarding half of the synthesized material unless a dynamic kinetic resolution is employed, which adds further complexity and catalyst costs. Another common approach involves Diels-Alder reactions using N-dimethylamino propylene imine and N-benzyl maleimide, which, while effective, generate significant quantities of byproducts that complicate purification and reduce overall throughput. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for high-purity pharmaceutical intermediates, creating supply chain vulnerabilities for manufacturers of generic quinolones.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages the intrinsic chirality of L-asparagine to dictate the stereochemical outcome of the entire synthesis, effectively rendering chiral resolution obsolete. The route is characterized by its logical progression, where the amino acid backbone is systematically transformed into the fused bicyclic structure required for moxifloxacin. By initiating the synthesis with a naturally occurring chiral source, the process guarantees the correct absolute configuration from the first step, thereby maximizing the theoretical yield potential. The design incorporates robust protecting group strategies, such as the use of trimethylacetyl groups, which offer stability during subsequent transformations while being easily removable under mild conditions. This novel approach not only simplifies the operational workflow by reducing the total number of unit operations but also enhances the environmental profile of the manufacturing process by minimizing solvent usage and waste generation associated with resolution salts. The result is a highly practical and industrially viable pathway that aligns perfectly with the goals of green chemistry and sustainable manufacturing.
Mechanistic Insights into MsCl/NaH-Mediated Cyclization
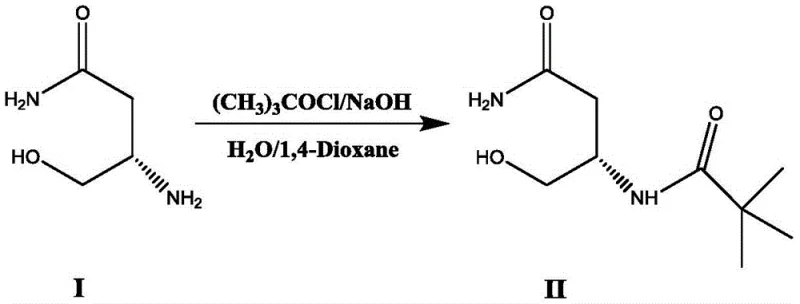
A critical juncture in this synthetic pathway is the formation of the five-membered pyrrolidinone ring, a structural motif essential for the biological activity of the final antibiotic. The patent details a sophisticated solution to a common mechanistic pitfall: the competitive formation of alicyclic byproducts where oxygen atoms substitute nitrogen atoms in the ring structure. In the conversion of Compound II to Compound III, the inventors identified that standard cyclization conditions often led to O-alkylation rather than the desired N-alkylation. To overcome this, the process employs methanesulfonyl chloride (MsCl) to activate the hydroxyl group, converting it into a superior leaving group (mesylate). Subsequent treatment with sodium hydride (NaH) in tetrahydrofuran generates the nucleophilic amide anion, which then attacks the activated carbon center intramolecularly. This specific sequence of activation followed by base-mediated cyclization kinetically favors the formation of the N-heterocycle, drastically suppressing the formation of oxygen-containing impurities. This level of mechanistic control is vital for maintaining high purity standards without resorting to extensive chromatographic purification, which is often impractical on a multi-ton scale.
Furthermore, the final stages of the synthesis demonstrate a keen understanding of orthogonal deprotection strategies to reveal the free amine functionalities required for coupling with the quinolone core. The removal of the benzyl and acyl protecting groups is achieved through a combination of catalytic hydrogenation and hydrazinolysis. Specifically, the use of Pd-C under a hydrogen atmosphere allows for the clean cleavage of the benzyl group without affecting the sensitive lactam ring. Following this, the reaction with hydrazine hydrate in dimethyl sulfoxide (DMSO) effectively removes the trimethylacetyl group via nucleophilic acyl substitution, yielding the target diamine intermediate. The choice of DMSO as a solvent in this step is particularly noteworthy, as it facilitates the solubility of the polar intermediates and stabilizes the transition state for the hydrazine attack. This careful orchestration of deprotection steps ensures that the final product emerges with the precise structural integrity needed for the subsequent coupling reaction, minimizing the risk of epimerization or degradation that could compromise the efficacy of the final drug substance.
How to Synthesize Moxifloxacin Chiral Side Chain Intermediate Efficiently
The execution of this eight-step synthesis requires precise control over reaction parameters to maximize yield and purity at each stage. The process begins with the catalytic hydrogenation of L-asparagine, followed by sequential protection, cyclization, alkylation, and deprotection steps. Each transformation has been optimized to balance reaction kinetics with selectivity, ensuring that the chiral information encoded in the starting material is preserved throughout the sequence. For process chemists looking to implement this route, attention to detail regarding reagent stoichiometry and temperature control is paramount, particularly during the cyclization and ring-closing steps where side reactions can occur. The detailed standardized synthesis steps below outline the specific conditions and workup procedures required to achieve the high yields reported in the patent documentation.
- Catalytic hydrogenation of L-asparagine using Ru-C/H2 in dilute HCl to form Compound I.
- Protection of the amine group with trimethylacetyl chloride to form Compound II, followed by cyclization using MsCl and NaH to form the pyrrolidinone ring (Compound III).
- N-benzylation, alkylation with 1,3-dibromopropane, ring closure with t-BuOK, and final deprotection/hydrazinolysis to yield the target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers transformative advantages that directly impact the bottom line and operational resilience of pharmaceutical manufacturing. The primary driver of value is the substitution of expensive, specialty starting materials with L-asparagine, a commodity chemical produced in massive volumes for the food and feed industries. This shift decouples the production of the intermediate from the volatile pricing dynamics of niche fine chemicals, providing a stable and predictable cost base. Moreover, the elimination of chiral resolution steps removes the need for purchasing costly resolving agents and the associated infrastructure for recycling or disposing of the unwanted enantiomer. This simplification of the bill of materials (BOM) translates into substantial cost savings in raw material procurement and inventory management. Additionally, the robust nature of the reaction conditions, which avoid cryogenic temperatures and pyrophoric reagents in favor of ambient or mild heating, reduces the energy consumption and specialized equipment requirements of the manufacturing facility.
- Cost Reduction in Manufacturing: The economic benefits of this process are profound, primarily driven by the high atom efficiency and the avoidance of yield-limiting resolution steps. By preserving the chirality of the starting material throughout the synthesis, the theoretical yield is effectively doubled compared to racemic synthesis followed by resolution. This efficiency gain means that less raw material is required to produce the same amount of active intermediate, directly lowering the variable cost per kilogram. Furthermore, the use of common solvents like water, ethyl acetate, and THF, along with non-precious metal catalysts where possible, minimizes the expenditure on specialized reagents. The reduction in byproduct formation also lowers the cost associated with waste treatment and disposal, contributing to a leaner and more cost-effective manufacturing operation that can withstand margin pressures in the generic drug market.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the reliance on L-asparagine, which is sourced from a mature and diversified global supply network. Unlike specialized heterocyclic building blocks that may have single-source suppliers or long lead times, L-asparagine is readily available from multiple vendors, mitigating the risk of supply disruptions. The simplified synthetic route also reduces the number of intermediate handoffs and quality control checkpoints, shortening the overall production cycle time. This agility allows manufacturers to respond more rapidly to fluctuations in market demand for moxifloxacin, ensuring that downstream formulation teams have a consistent supply of high-quality API. The robustness of the process against minor variations in reaction conditions further enhances reliability, reducing the incidence of batch failures that can stall production lines and delay shipments to customers.
- Scalability and Environmental Compliance: The scalability of this route is evidenced by the replacement of hazardous reagents like lithium diisopropylamide (LDA), which requires strict temperature control at minus 40 degrees Celsius, with safer alternatives like potassium tert-butoxide. This modification removes a significant safety barrier to scale-up, allowing the reaction to be performed in standard stainless steel reactors without the need for specialized cryogenic cooling systems. From an environmental standpoint, the process generates fewer hazardous wastes and avoids the use of heavy metals in the final steps, aligning with increasingly stringent regulatory requirements for pharmaceutical manufacturing. The high selectivity of the cyclization step minimizes the generation of difficult-to-separate impurities, simplifying the purification process and reducing the volume of solvent waste. These factors collectively make the process highly attractive for commercial scale-up, enabling the production of complex pharmaceutical intermediates with a reduced environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific challenges and solutions detailed in the patent literature, providing clarity on the feasibility and advantages of the technology. Understanding these nuances is essential for stakeholders evaluating the potential for technology transfer or licensing.
Q: Why is L-asparagine preferred over 2,3-dipicolinic acid for this synthesis?
A: L-asparagine is a readily available, low-cost chiral pool starting material that eliminates the need for expensive resolution steps required when using achiral precursors like 2,3-dipicolinic acid.
Q: How does the new process control byproducts during the cyclization step?
A: The process utilizes methanesulfonyl chloride and sodium hydride under specific temperature conditions to favor N-cyclization over O-substitution, significantly reducing alicyclic byproducts.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the route avoids hazardous reagents like LDA at cryogenic temperatures, opting for safer bases like potassium tert-butoxide, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Moxifloxacin Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of antibiotic manufacturing. Our team of expert process chemists has thoroughly analyzed the technology described in patent CN110218210B and is fully prepared to adapt this innovative eight-step sequence for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant and finally to full-scale manufacturing is seamless and risk-mitigated. Our facilities are equipped with state-of-the-art reactor systems capable of handling the specific conditions required for this synthesis, including hydrogenation units and precise temperature control systems. We are committed to delivering moxifloxacin chiral side chain intermediates that meet stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify identity and assay at every stage of production.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and robust synthesis technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this L-asparagine-based route for your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data from our pilot batches and to discuss route feasibility assessments tailored to your project timelines. Let us help you optimize your supply chain for moxifloxacin production with a partner dedicated to quality, innovation, and reliability.
