Scalable Metal-Free Synthesis of Trifluoromethyl Tertiary Alcohols for Advanced Agrochemical Applications
Scalable Metal-Free Synthesis of Trifluoromethyl Tertiary Alcohols for Advanced Agrochemical Applications
The development of efficient synthetic routes for fluorinated organic molecules remains a cornerstone of modern medicinal and agricultural chemistry. Patent CN115197200B introduces a groundbreaking methodology for the synthesis of trifluoromethyl tertiary alcohols, addressing critical limitations in existing technologies regarding catalyst dependency and environmental impact. This innovation leverages the unique reactivity of trifluoroacetyl-substituted coumarins and nitrogen heterocycles to construct complex scaffolds in a single step. The significance of this chemical motif cannot be overstated, as trifluoromethyl-substituted tertiary alcohols serve as pivotal intermediates in the creation of high-value bioactive agents. As illustrated in the structural diversity of known pharmaceuticals, these scaffolds are integral to glucocorticoid receptor agonists, HIV inhibitors, and anti-inflammatory agents, highlighting their broad utility across therapeutic areas.
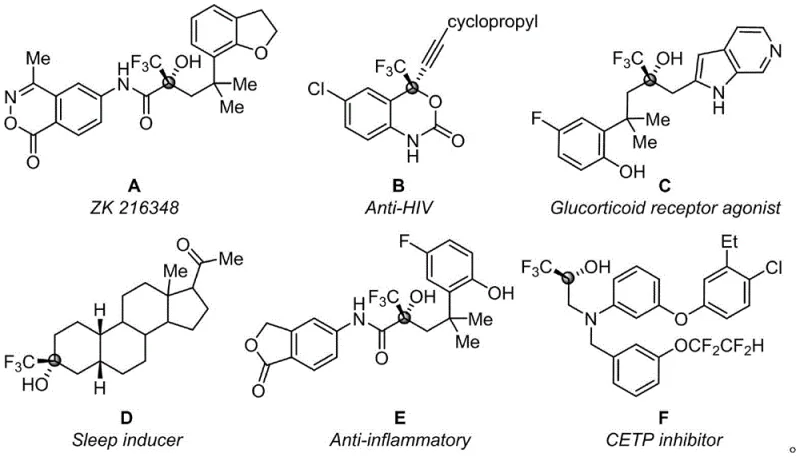
Furthermore, the patent explicitly demonstrates the potential of these novel compounds in the agrochemical sector, where they exhibit potent fungicidal properties against prevalent plant pathogens. For procurement managers and supply chain directors, this represents a dual opportunity: accessing a versatile intermediate for pharmaceutical pipelines while simultaneously securing a robust supply of active agrochemical ingredients. The ability to produce these high-purity intermediates without the burden of heavy metal residues aligns perfectly with the increasing regulatory pressures facing the global chemical industry. By adopting this technology, manufacturers can position themselves as a reliable agrochemical intermediate supplier capable of meeting stringent purity specifications required for next-generation crop protection solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl tertiary alcohol skeletons via the coupling of trifluoromethyl ketones and methylquinolines has been fraught with significant technical and economic challenges. Traditional protocols predominantly rely on the use of Lewis acid catalysts, such as indium or iron salts, to activate the carbonyl group and facilitate the nucleophilic attack. While effective in laboratory settings, these metal-mediated processes introduce substantial complications for industrial scale-up. The presence of metal salts necessitates rigorous downstream purification steps to ensure the final product meets residual metal limits, often requiring expensive scavenging resins or complex extraction procedures. Moreover, the generation of metal-containing waste streams poses a severe environmental liability, conflicting with the principles of green chemistry and sustainable manufacturing that modern enterprises strive to uphold.
Beyond the environmental concerns, the operational complexity of metal-catalyzed reactions often leads to inconsistent batch-to-batch reproducibility and lower overall yields due to side reactions promoted by the Lewis acids. The sensitivity of these catalysts to moisture and oxygen further complicates the reaction setup, requiring inert atmospheres and anhydrous conditions that drive up energy and infrastructure costs. For a procurement manager focused on cost reduction in agrochemical intermediate manufacturing, these hidden costs associated with catalyst procurement, waste disposal, and extended processing times can erode profit margins significantly. Consequently, there is an urgent industry demand for a streamlined, catalyst-free alternative that maintains high efficiency while eliminating the logistical burdens of metal handling.
The Novel Approach
The methodology disclosed in CN115197200B offers a transformative solution by enabling the direct C(sp3)-H functionalization of methylquinolines with trifluoroacetylcoumarins without any external catalyst or additive. This metal-free approach represents a paradigm shift in synthetic efficiency, achieving 100% atom economy by incorporating all atoms from the starting materials into the final product. The reaction proceeds smoothly under air atmosphere in common organic solvents such as acetonitrile, demonstrating exceptional functional group tolerance and operational simplicity. By eliminating the need for metal catalysts, this process inherently removes the risk of heavy metal contamination, thereby simplifying the purification workflow to basic filtration and chromatography. This not only accelerates the production timeline but also drastically reduces the consumption of auxiliary materials and solvents typically required for metal removal.
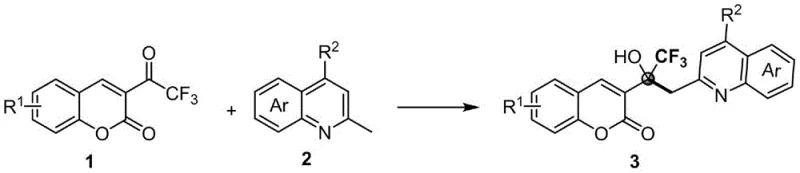
From a commercial perspective, this novel route provides a distinct competitive advantage by lowering the barrier to entry for high-quality intermediate production. The mild reaction conditions, ranging from 50°C to 120°C, allow for the use of standard reactor equipment without the need for specialized high-pressure or cryogenic setups. This accessibility facilitates rapid technology transfer from laboratory to pilot plant, ensuring a stable and continuous supply chain for downstream customers. For supply chain heads concerned with lead time reliability, the robustness of this catalyst-free system means fewer production delays caused by catalyst deactivation or sensitivity issues. Ultimately, this approach delivers a high-purity agrochemical intermediate that is both economically viable and environmentally sustainable, meeting the evolving demands of global regulatory bodies.
Mechanistic Insights into Metal-Free C-H Functionalization
The success of this catalyst-free transformation lies in the intrinsic electronic properties of the reactants and the specific activation of the C-H bond adjacent to the nitrogen heterocycle. The trifluoroacetyl group on the coumarin ring acts as a powerful electron-withdrawing moiety, significantly enhancing the electrophilicity of the carbonyl carbon. This activation allows the relatively weakly acidic methyl C-H bond of the quinoline derivative to undergo deprotonation or radical abstraction under thermal conditions, generating a reactive nucleophilic species in situ. Unlike traditional methods that require Lewis acids to coordinate with the carbonyl oxygen, this thermal pathway leverages the polarity of the solvent and the stability of the resulting trifluoromethyl carbinol center to drive the reaction forward. The formation of the quaternary carbon center bearing the hydroxyl and trifluoromethyl groups is thermodynamically favorable, ensuring high conversion rates even in the absence of catalytic promotion.
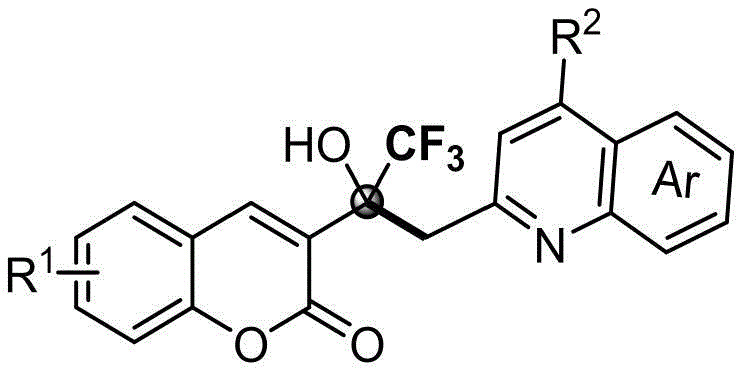
Regarding impurity control, the absence of metal catalysts inherently minimizes the formation of metal-complexed byproducts that are notoriously difficult to separate. The primary side reactions in this system are typically limited to over-oxidation or solvent interactions, which are easily managed by optimizing the reaction temperature and time. The patent data indicates that varying the substituents on the coumarin and quinoline rings (such as halogens or alkoxy groups) does not significantly hinder the reaction, suggesting a broad substrate scope with consistent impurity profiles. This predictability is crucial for R&D directors who need to validate analytical methods and establish strict quality control parameters. The clean reaction profile ensures that the final crystalline products can be obtained with high purity after simple recrystallization or column chromatography, reducing the need for extensive preparative HPLC purification.
How to Synthesize Trifluoromethyl Tertiary Alcohol Efficiently
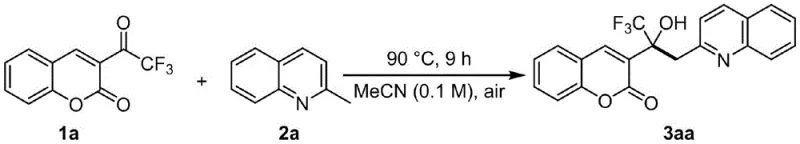
To implement this advanced synthesis in a production environment, precise control over reaction parameters is essential to maximize yield and minimize impurities. The protocol involves combining the trifluoroacetylcoumarin precursor with the methylquinoline derivative in a polar aprotic solvent, with acetonitrile being the preferred medium due to its optimal solubility and boiling point characteristics. The reaction mixture is heated in a sealed vessel to maintain solvent integrity and prevent evaporation losses during the extended heating period. Following the reaction, the workup procedure is remarkably straightforward, involving filtration through a pad of diatomaceous earth to remove any insoluble particulates, followed by solvent evaporation under reduced pressure. The crude residue is then subjected to silica gel column chromatography using a petroleum ether and ethyl acetate gradient to isolate the pure target compound.
- Mix trifluoroacetylcoumarin compound and methylquinoline compound in a solvent such as acetonitrile at a molar ratio of 1: 2.
- Heat the reaction mixture to 90°C in a sealed tube under air atmosphere for 9 hours to ensure complete conversion.
- Filter the mixture through diatomaceous earth, remove solvent under reduced pressure, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this metal-free synthetic route offers profound commercial benefits that extend far beyond the laboratory bench, directly impacting the bottom line and operational resilience of chemical manufacturing enterprises. For procurement teams, the elimination of expensive and often volatile metal catalysts translates into immediate raw material cost savings and reduced supply chain vulnerability. Since the process relies on readily available organic starting materials and common solvents, sourcing becomes more predictable and less susceptible to geopolitical fluctuations that often affect the supply of specialty metals. Furthermore, the simplified purification process reduces the consumption of secondary reagents and consumables, such as metal scavengers and specialized filtration media, leading to a leaner and more cost-effective production model. These efficiencies collectively contribute to substantial cost savings in pharmaceutical intermediates manufacturing, allowing companies to offer more competitive pricing without compromising on quality.
- Cost Reduction in Manufacturing: The removal of metal catalysts from the process equation eliminates the need for costly downstream purification steps designed to reduce metal residues to ppm levels. This streamlining of the workflow reduces labor hours, energy consumption, and waste disposal fees associated with hazardous metal waste. Additionally, the high atom economy of the reaction ensures that raw materials are utilized efficiently, minimizing waste generation and maximizing the output per batch. By avoiding the use of precious or toxic metals, manufacturers also mitigate the regulatory compliance costs associated with environmental monitoring and reporting. This holistic reduction in operational overhead creates a more robust financial structure for long-term production contracts.
- Enhanced Supply Chain Reliability: Relying on a catalyst-free system significantly de-risks the supply chain by removing a potential single point of failure. Metal catalysts can suffer from batch-to-batch variability or supply shortages, which can halt production lines; this new method bypasses those risks entirely. The use of stable, commodity-grade solvents and reagents ensures that production can continue uninterrupted even during market disruptions. Moreover, the mild reaction conditions reduce wear and tear on reactor equipment, extending asset life and reducing maintenance downtime. For supply chain heads, this translates to improved on-time delivery performance and the ability to scale production volumes rapidly in response to market demand without complex process re-validation.
- Scalability and Environmental Compliance: The green chemistry credentials of this process provide a strategic advantage in markets with strict environmental regulations. The absence of heavy metals simplifies wastewater treatment and solid waste management, ensuring compliance with increasingly stringent discharge standards. The reaction's scalability is proven by its tolerance to air and moisture, meaning that expensive inert gas blanketing systems are not strictly required for large-scale batches. This ease of scale-up facilitates the transition from kilogram to ton-scale production with minimal technical barriers. Companies adopting this technology can market their products as sustainably produced, appealing to eco-conscious partners and fulfilling corporate social responsibility goals related to carbon footprint reduction and waste minimization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the practical advantages of the metal-free approach and its alignment with modern industry standards for safety and efficiency.
Q: Does this synthesis require expensive metal catalysts?
A: No, the patented method described in CN115197200B operates without any metal catalysts or additives, utilizing a direct C-H functionalization strategy that significantly simplifies the purification process and reduces heavy metal contamination risks.
Q: What is the antifungal activity of the synthesized compounds?
A: The synthesized trifluoromethyl tertiary alcohols exhibit notable bactericidal activity against common agricultural pathogens, with specific derivatives showing up to 63.32% inhibition rates against Rhizoctonia solani.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction conditions are mild (50-120°C), use common solvents like acetonitrile, and achieve high atom economy (100%), making it highly scalable and environmentally compliant for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Tertiary Alcohol Supplier
The technological breakthroughs detailed in CN115197200B underscore the immense potential of metal-free C-H functionalization in producing high-value fluorinated intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes are optimized for safety, efficiency, and cost-effectiveness. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of trifluoromethyl tertiary alcohol meets the highest international standards. Our commitment to quality assurance ensures that our clients receive intermediates that are ready for immediate use in downstream API or agrochemical synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this catalyst-free technology can be tailored to your specific project requirements. By collaborating with us, you can leverage our process development capabilities to conduct a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener synthetic route. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our materials in your own applications. Together, we can drive innovation in the agrochemical and pharmaceutical sectors by delivering superior intermediates that combine scientific excellence with commercial viability.
