Advanced Metal-Free Synthesis of Trifluoromethyl Tertiary Alcohols for Commercial Scale Production
Introduction to Next-Generation Fluorinated Scaffolds
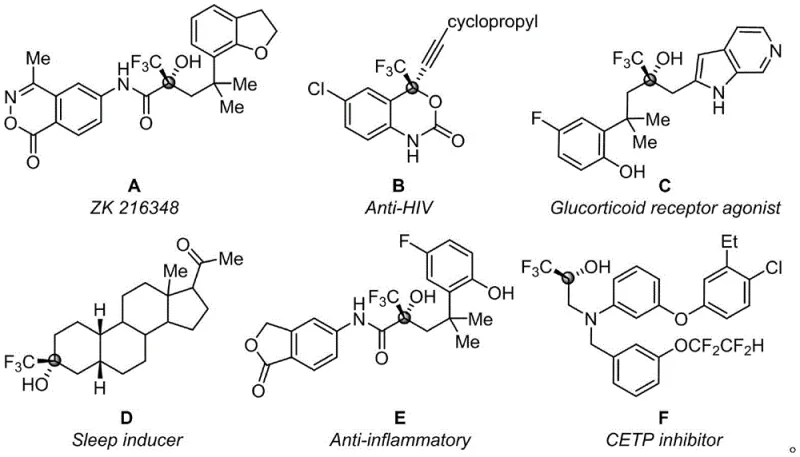
The strategic incorporation of fluorine atoms into organic molecules has revolutionized modern medicinal chemistry and agrochemical design, primarily due to the unique electronic and lipophilic properties that fluorine imparts to biological targets. As detailed in the recent patent documentation CN115197200B, there is a pressing industrial demand for efficient methodologies to construct trifluoromethyl-substituted tertiary alcohol backbones, which are ubiquitous motifs found in high-value therapeutic agents ranging from HIV reverse transcriptase inhibitors to glucocorticoid receptor agonists. This groundbreaking technology addresses the critical limitations of existing synthetic routes by introducing a novel, one-step assembly strategy that utilizes simple and readily available starting materials, specifically trifluoroacetyl group substituted coumarin compounds and nitrogen heterocyclic 2-methylquinoline derivatives. The significance of this development cannot be overstated for supply chain stakeholders, as it promises to streamline the production of complex fluorinated intermediates while simultaneously enhancing the environmental profile of the manufacturing process through the elimination of hazardous catalytic systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of azaarene-substituted trifluoromethyl tertiary alcohol compounds has relied heavily on the participation of stoichiometric or catalytic amounts of Lewis acids, such as indium or iron salts, to activate the carbonyl species for nucleophilic attack. These traditional protocols introduce significant operational complexities and downstream processing burdens, as the removal of residual metal contaminants often necessitates expensive purification steps like chelating resin treatment or extensive recrystallization to meet stringent pharmaceutical purity specifications. Furthermore, the reliance on moisture-sensitive catalysts frequently mandates the use of inert atmosphere techniques, such as gloveboxes or Schlenk lines, which drastically increases the capital expenditure and operational overhead required for commercial scale-up. From a sustainability perspective, the generation of heavy metal waste streams poses a substantial environmental liability, conflicting with the global industry shift towards greener, more sustainable chemical manufacturing practices that minimize ecological footprints and regulatory compliance costs.
The Novel Approach
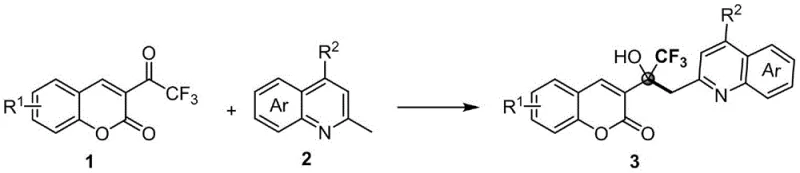
In stark contrast to these legacy methods, the innovative process disclosed in the patent data achieves the direct C(sp3)–H functionalization of methylquinolines coupled with trifluoromethyl ketones without the requirement for any external metal catalyst or additive. This metal-free paradigm shift simplifies the reaction setup to merely mixing the raw materials in a common organic solvent under ambient air conditions, thereby removing the need for specialized inert gas infrastructure and reducing the overall process mass intensity. The reaction demonstrates exceptional functional group tolerance, accommodating various substituents on both the coumarin and quinoline rings, which allows for the rapid generation of diverse molecular libraries for structure-activity relationship studies without the fear of catalyst poisoning or side reactions. By achieving 100% atom economy in the coupling step, this approach not only maximizes the utilization of raw materials but also significantly reduces the volume of chemical waste generated per kilogram of product, aligning perfectly with the principles of green chemistry and offering a robust pathway for cost-effective industrial production.
Mechanistic Insights into Metal-Free C-H Functionalization
The underlying mechanism of this transformation represents a sophisticated interplay of electronic activation and thermal energy that bypasses the traditional need for Lewis acid coordination. It is hypothesized that the electron-deficient nature of the trifluoroacetyl group on the coumarin scaffold sufficiently activates the carbonyl carbon towards nucleophilic attack by the methyl group of the quinoline derivative under thermal conditions. The absence of a metal catalyst suggests that the reaction proceeds through a concerted or radical-mediated pathway facilitated by the inherent polarity of the reactants and the stabilizing effect of the adjacent nitrogen atom in the heterocyclic ring. This mechanistic simplicity is a major advantage for process chemists, as it eliminates the variability associated with catalyst batch-to-batch consistency and removes the risk of trace metal leaching which can compromise the stability of the final active pharmaceutical ingredient during long-term storage. Understanding this catalyst-free trajectory allows R&D teams to confidently predict reaction outcomes across a broad substrate scope, knowing that the driving force is thermodynamic rather than dependent on fragile catalytic cycles that might be disrupted by minor impurities in the feedstock.
Regarding impurity control, the high selectivity of this metal-free system inherently minimizes the formation of complex byproduct profiles that are often observed in transition-metal catalyzed cross-couplings. Without the presence of metal centers that can facilitate multiple competing pathways such as homocoupling or over-oxidation, the reaction channel is narrowly focused on the desired C-C bond formation between the methylene carbon and the carbonyl carbon. This intrinsic selectivity translates directly to a cleaner crude reaction mixture, which significantly eases the burden on downstream purification units and reduces the loss of yield typically associated with aggressive purification protocols needed to remove metal residues. For quality assurance departments, this means a more consistent impurity profile that is easier to characterize and control, ensuring that the final trifluoromethyl tertiary alcohol intermediates meet the rigorous specifications required for subsequent elaboration into final drug substances or agrochemical active ingredients.
How to Synthesize Trifluoromethyl Tertiary Alcohol Efficiently
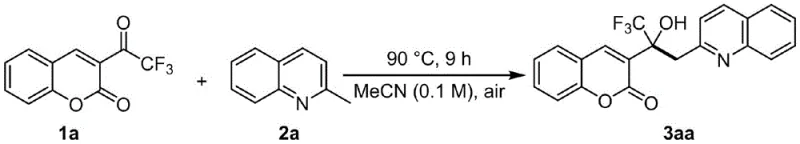
To implement this synthesis in a laboratory or pilot plant setting, operators should follow the generalized protocol which emphasizes simplicity and robustness under aerobic conditions. The process begins by charging a reaction vessel with the trifluoroacetyl coumarin substrate and the methylquinoline derivative in a molar ratio that favors complete consumption of the limiting reagent, typically utilizing a slight excess of the quinoline component to drive the equilibrium forward. The choice of solvent is critical, with acetonitrile identified as the optimal medium due to its ability to dissolve both polar and non-polar components effectively while maintaining stability at elevated temperatures, although other solvents like DMF or DMSO can be employed depending on specific substrate solubility requirements. Following the reaction period, the workup procedure is remarkably straightforward, involving simple filtration to remove any insoluble particulates followed by solvent evaporation, which stands in sharp contrast to the aqueous quenching and extraction sequences often required for metal-catalyzed reactions.
- Combine trifluoroacetyl coumarin compound and methylquinoline compound in a suitable solvent such as acetonitrile within a sealed reaction vessel.
- Heat the reaction mixture to a temperature range between 50°C and 120°C under an air atmosphere for a duration of 3 to 15 hours to ensure complete conversion.
- Upon completion, filter the mixture through a pad of diatomaceous earth, remove the solvent under reduced pressure, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis technology offers transformative benefits that extend far beyond the laboratory bench, impacting the total cost of ownership and supply security for critical fluorinated intermediates. The elimination of expensive transition metal catalysts such as palladium, rhodium, or even cheaper Lewis acids like indium triflate results in a direct reduction in raw material costs, while simultaneously removing the logistical complexity associated with sourcing and storing sensitive catalytic reagents that often have limited shelf lives. Furthermore, the ability to run the reaction under air atmosphere rather than requiring nitrogen or argon blanketing significantly lowers utility costs and infrastructure requirements, making this process highly adaptable for manufacturing facilities in regions where inert gas supplies may be costly or unreliable. These operational efficiencies compound to create a more resilient supply chain that is less vulnerable to fluctuations in the global market prices of specialty metals and gases.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of metal catalysts, which eradicates the need for costly metal scavenging resins and the associated validation testing required to prove residual metal levels are below ppm thresholds in the final product. This simplification of the purification train reduces the consumption of solvents and stationary phases during chromatography, leading to substantial savings in consumable costs and waste disposal fees. Additionally, the high atom economy of the reaction ensures that a greater proportion of the purchased raw materials ends up in the final product, minimizing the effective cost per kilogram of the active intermediate and improving the overall gross margin for the manufacturing operation.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals like coumarins and methylquinolines which are produced on a multi-ton scale globally, this process mitigates the risk of supply disruptions often associated with bespoke or highly specialized catalytic reagents. The robustness of the reaction conditions, which tolerate air and moisture, further enhances reliability by reducing the likelihood of batch failures due to environmental excursions or operator error during setup. This stability allows for more accurate production planning and shorter lead times, as the process does not require the extended equilibration times or strict anhydrous conditions that can bottleneck production schedules in traditional fine chemical manufacturing environments.
- Scalability and Environmental Compliance: The green chemistry credentials of this method provide a significant advantage in navigating increasingly stringent environmental regulations, as the absence of heavy metals simplifies wastewater treatment and reduces the regulatory burden associated with hazardous waste classification. The scalability is proven by the successful demonstration of the reaction across a wide range of substrates with consistent yields, indicating that the kinetics are well-suited for transfer from gram-scale laboratory synthesis to multi-kilogram pilot and commercial production without significant re-optimization. This ease of scale-up ensures that supply partners can rapidly respond to increased demand from downstream pharmaceutical or agrochemical clients without the need for prolonged process development campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis technology, providing clarity for technical teams evaluating its potential integration into their existing workflows. These answers are derived directly from the experimental data and claims presented in the patent literature, ensuring that the information provided is accurate and reflective of the actual capabilities of the method. Understanding these nuances is essential for project managers who need to assess the feasibility of adopting this route for specific target molecules within their pipeline.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method described in CN115197200B operates under metal-free conditions, eliminating the need for indium, iron, or other Lewis acid catalysts typically required in conventional routes.
Q: What is the primary application of these trifluoromethyl tertiary alcohols?
A: These compounds serve as critical scaffolds in pharmaceutical drug discovery and exhibit significant fungicidal activity against agricultural pathogens such as Rhizoctonia solani.
Q: What are the optimal reaction conditions for high yield?
A: Optimal results are achieved using acetonitrile as the solvent at 90°C for 9 hours under an air atmosphere, providing excellent yields without inert gas protection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Tertiary Alcohol Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to translate this innovative metal-free synthesis from patent literature into a robust commercial reality for your organization. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of metal contaminants and ensure the structural integrity of every batch of trifluoromethyl tertiary alcohol intermediates we produce. Our commitment to quality and consistency makes us the ideal partner for companies seeking to secure a stable supply of these high-value fluorinated building blocks.
We invite you to engage with our technical procurement team to discuss how this novel synthetic route can be tailored to your specific project needs and timeline requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this metal-free process for your specific target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate the tangible value our manufacturing capabilities can bring to your supply chain.
