Advanced Ruthenium-Catalyzed Synthesis of Anticancer Indenopyrazolopyrazolone Intermediates for Commercial Scale-Up
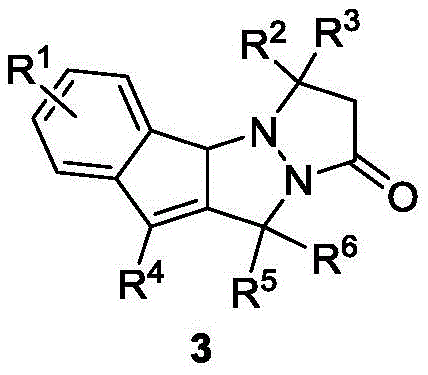
The pharmaceutical industry continuously seeks novel heterocyclic scaffolds that offer unique biological profiles, particularly in the realm of oncology. Patent CN112939988A discloses a groundbreaking class of indenopyrazolopyrazolone compounds, characterized by a fused nitrogen-containing heterocyclic system that merges the structural advantages of indenopyrazoles and N,N-bicyclic pyrazolones. These compounds are not merely theoretical constructs; they exhibit profound anticancer activity against critical cell lines such as REC-1 and Ramos, positioning them as high-value candidates for drug discovery pipelines. The core innovation lies in the efficient construction of this complex tetracyclic framework through a sophisticated cascade reaction, overcoming the synthetic bottlenecks that have historically plagued the production of such densely functionalized molecules.
For procurement managers and supply chain directors, the significance of this patent extends beyond biological efficacy to the realm of manufacturability. The disclosed method utilizes readily available starting materials—aryl azomethine imines and propargyl alcohol compounds—which simplifies the sourcing strategy and mitigates supply chain risks associated with exotic reagents. By establishing a reliable pharmaceutical intermediate supplier network for these specific precursors, manufacturers can secure a steady flow of high-quality inputs. Furthermore, the reaction operates under mild conditions, typically around 60 °C, which drastically reduces energy consumption compared to high-temperature protocols, directly contributing to cost reduction in API manufacturing and enhancing the overall sustainability profile of the production process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of related indenopyrazole structures faced significant hurdles that limited their commercial viability and biological utility. Existing literature, such as the work by Kim et al., described ruthenium-catalyzed reactions involving arylazomethines and allylic acetals. However, this conventional approach suffered from critical drawbacks: the allylic acetal raw materials are notoriously difficult to synthesize and possess poor stability, leading to inconsistent batch quality and increased waste. Moreover, the resulting products were hydrogenated indenopyrazolopyrazolones, lacking a crucial carbon-carbon double bond on the indene ring. This structural deficiency fundamentally alters the biochemical properties of the molecule, often rendering it less effective or entirely inactive for the intended therapeutic applications, thereby necessitating additional synthetic steps to reintroduce unsaturation.
The Novel Approach
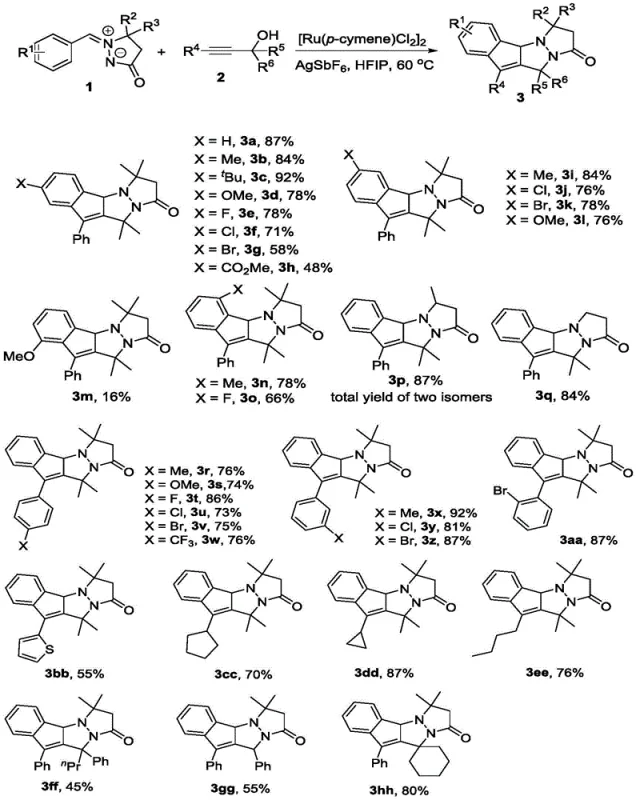
The methodology presented in CN112939988A represents a paradigm shift in constructing these fused heterocycles. By employing a tandem reaction between aryl azomethine imines and propargyl alcohol compounds, the process achieves a direct, one-pot formation of the target indenopyrazolopyrazolone skeleton. This strategy eliminates the need for unstable allylic acetals and bypasses the formation of the undesired hydrogenated byproducts. The reaction is catalyzed by robust transition metal complexes, specifically rhodium or ruthenium catalysts, in the presence of specific additives. This streamlined approach not only simplifies the operational workflow but also ensures the final product retains the essential conjugated system required for optimal anticancer activity, providing a superior route for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ruthenium-Catalyzed Cascade Cyclization
From an R&D perspective, understanding the mechanistic underpinnings of this transformation is vital for process optimization and impurity control. The reaction proceeds via a ruthenium-catalyzed C-H activation pathway, where the metal center coordinates with the aryl azomethine imine to facilitate the initial bond formation. The presence of the silver salt additive, such as AgSbF6, plays a pivotal role in generating the active cationic ruthenium species by abstracting chloride ligands, thereby enhancing the electrophilicity of the catalyst. This activation step is crucial for initiating the cascade sequence that eventually leads to the formation of the multiple rings in a single operation. The choice of solvent is equally critical; fluorinated alcohols like 2,2,2-trifluoroethanol (TFE) or hexafluoroisopropanol (HFIP) are preferred due to their unique hydrogen-bonding donor capabilities, which stabilize charged intermediates and accelerate the reaction kinetics without participating in side reactions.
Controlling the impurity profile in such complex cascade reactions is a major challenge, yet this method demonstrates remarkable selectivity. The specific coordination geometry imposed by the ruthenium catalyst directs the regioselective cyclization, ensuring that the propargyl alcohol moiety integrates correctly into the growing heterocyclic framework. This precision minimizes the formation of regioisomers or oligomeric byproducts that are common in uncatalyzed thermal reactions. The broad substrate scope demonstrated in the patent, accommodating various substituents on both the aryl ring and the alkyne component, indicates a robust catalytic cycle that tolerates electronic and steric variations. This tolerance is essential for medicinal chemists aiming to generate diverse libraries of analogs for structure-activity relationship (SAR) studies without needing to re-optimize conditions for every new derivative.
How to Synthesize Indenopyrazolopyrazolone Efficiently
The practical implementation of this synthesis is designed for scalability and ease of execution, making it highly attractive for process chemistry teams. The standard protocol involves charging a reaction vessel with the aryl azomethine imine, the propargyl alcohol compound, the ruthenium catalyst dimer [Ru(p-cymene)Cl2]2, and the silver hexafluoroantimonate additive. The mixture is then dissolved in hexafluoroisopropanol (HFIP), a solvent that has proven to deliver the highest yields, often exceeding 80% for model substrates. The reaction is heated to a moderate temperature of 60 °C and stirred for approximately 2 hours, after which the conversion is typically complete. Workup involves simple filtration and concentration, followed by purification via silica gel column chromatography, yielding the pure white solid product ready for biological evaluation.
- Mix aryl azomethine imine, propargyl alcohol compound, [Ru(p-cymene)Cl2]2 catalyst, AgSbF6 additive, and HFIP solvent in a reaction vessel.
- Heat the reaction mixture to 60 °C and stir for approximately 2 hours to facilitate the cascade cyclization.
- Cool the reaction to room temperature, filter, concentrate under reduced pressure, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, this patented technology offers compelling economic and logistical benefits. The shift from unstable, hard-to-source allylic acetals to commercially abundant propargyl alcohols and aryl azomethines fundamentally de-risks the supply chain. This availability ensures that production schedules are not held hostage by the lead times of niche reagents, facilitating reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the one-pot nature of the reaction consolidates what would traditionally be a multi-step synthesis into a single unit operation. This consolidation reduces the requirement for intermediate isolation, solvent swaps, and extensive purification between steps, leading to substantial savings in labor, equipment usage, and solvent waste disposal costs.
- Cost Reduction in Manufacturing: The elimination of expensive and unstable starting materials directly lowers the Bill of Materials (BOM) cost. Additionally, the use of a highly active catalyst system allows for low catalyst loading (typically around 2.5 mol%), minimizing the expense associated with precious metals like ruthenium. The mild reaction temperature of 60 °C significantly reduces energy overheads compared to processes requiring reflux or cryogenic conditions. By avoiding the formation of hydrogenated byproducts, the process also negates the need for downstream dehydrogenation steps, further streamlining the manufacturing cost structure and improving the overall atom economy of the synthesis.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as propargyl alcohols and simple substituted benzaldehydes (precursors to the imines) ensures a resilient supply base. These materials are produced globally by numerous chemical manufacturers, preventing single-source bottlenecks. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that minor variations in raw material quality are less likely to cause batch failures. This reliability is critical for maintaining consistent production output and meeting the rigorous delivery commitments required by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving basic filtration and crystallization or chromatography, translates well to large-scale reactors. The process avoids the use of highly toxic reagents or extreme pressures, aligning with modern green chemistry principles and easing regulatory compliance burdens. The high yields observed across a broad range of substrates indicate that the process is efficient, generating less chemical waste per kilogram of product. This efficiency supports sustainable manufacturing goals and reduces the environmental footprint associated with the production of these valuable anticancer intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these indenopyrazolopyrazolone compounds. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this technology for adoption.
Q: What are the primary advantages of this synthesis method over prior art?
A: Unlike previous methods utilizing unstable allylic acetals that yield hydrogenated products, this novel approach uses stable aryl azomethine imines and propargyl alcohols to produce fully conjugated indenopyrazolopyrazolones with superior biological potential.
Q: What specific biological activity do these compounds exhibit?
A: The synthesized indenopyrazolopyrazolone derivatives demonstrate significant anticancer activity, specifically showing potent inhibition against REC-1 and Ramos cancer cell lines, making them valuable leads for oncology drug development.
Q: What are the optimal reaction conditions for high yield?
A: The optimal protocol involves using [Ru(p-cymene)Cl2]2 as the catalyst, AgSbF6 as the additive, and hexafluoroisopropanol (HFIP) as the solvent at a temperature of 60 °C, achieving isolated yields up to 87%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indenopyrazolopyrazolone Supplier
As the demand for novel anticancer scaffolds grows, partnering with an experienced CDMO is essential for translating laboratory success into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from gram-scale discovery to industrial manufacturing is seamless. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of handling complex heterocyclic syntheses, guaranteeing that every batch of indenopyrazolopyrazolone intermediate meets the highest standards required for clinical and commercial applications.
We invite you to leverage our technical expertise to optimize this ruthenium-catalyzed route for your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project volume. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your drug development timeline while maximizing cost efficiency.
