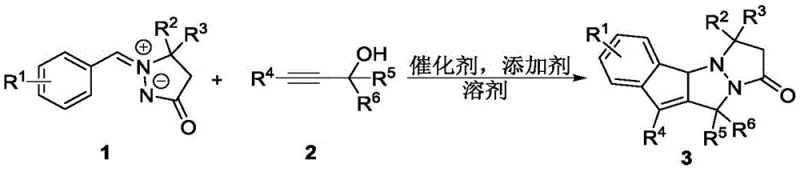
Scalable Ruthenium-Catalyzed Synthesis of Anticancer Indenopyrazolopyrazolone Intermediates
The pharmaceutical industry continuously seeks novel heterocyclic scaffolds that offer unique biological profiles, particularly in the realm of oncology. Patent CN112939988B introduces a groundbreaking class of indenopyrazolopyrazolone compounds, which fuse the structural motifs of indenopyrazoles and N,N-bicyclic pyrazolones into a single, potent pharmacophore. This innovation addresses a critical gap in medicinal chemistry by providing a streamlined synthetic route to these complex nitrogen-containing fused heterocycles. The disclosed methodology leverages a ruthenium-catalyzed cascade reaction between aryl azomethine imines and propargyl alcohols, enabling the rapid construction of the target skeleton in a single operational step. This technical breakthrough is not merely an academic exercise; it represents a viable pathway for generating high-purity pharmaceutical intermediates with demonstrated efficacy against aggressive cancer cell lines such as REC-1 and Ramos. For R&D teams focused on expanding their oncology pipeline, this patent offers a robust platform for developing next-generation antitumor agents with improved therapeutic indices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of related indenopyrazole structures faced significant hurdles that hindered their widespread adoption in drug discovery campaigns. Existing literature, such as the work by Kim et al., relied on ruthenium-catalyzed reactions involving aryl azomethine imines and allyl acetals. However, this conventional approach suffered from severe drawbacks, primarily concerning the stability and availability of the starting materials. Allyl acetals are often difficult to synthesize and exhibit poor stability under storage conditions, complicating supply chain logistics for large-scale manufacturing. Furthermore, the chemical outcome of these traditional methods was fundamentally limited; they produced hydrogenated indenopyrazolopyrazolones which lack a critical carbon-carbon double bond on the indene ring. This structural deficiency alters the electronic properties and three-dimensional conformation of the molecule, leading to biochemical performance that differs significantly from the desired unsaturated analogues. Consequently, the utility of these older methods for accessing the specific bioactive scaffold described in CN112939988B was effectively non-existent.
The Novel Approach
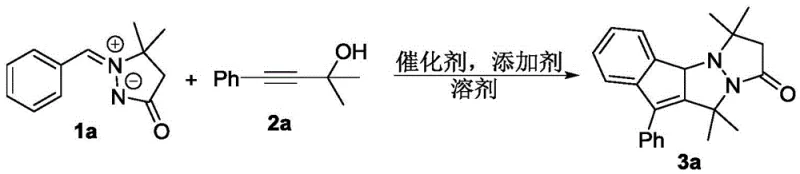
In stark contrast to the limitations of prior art, the novel methodology disclosed in this patent utilizes readily available and stable propargyl alcohol derivatives as coupling partners. This strategic shift in reactant selection allows for a direct, one-pot tandem reaction that efficiently constructs the fully unsaturated indenopyrazolopyrazolone core. The process operates under remarkably mild conditions, typically requiring temperatures around 60°C in hexafluoroisopropanol (HFIP) solvent, which minimizes energy consumption and reduces the formation of thermal degradation byproducts. By employing a [Ru(p-cymene)Cl2]2 catalyst system, the reaction achieves excellent regioselectivity and high yields, often exceeding 80% for a broad range of substrates. This efficiency translates directly into reduced waste generation and simplified downstream processing, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing. The ability to access diverse derivatives through simple modification of the aryl or alkyne components further enhances the versatility of this synthetic platform for medicinal chemists.
Mechanistic Insights into Ru-Catalyzed C-H Activation Cascade
The success of this transformation hinges on the precise orchestration of a ruthenium-catalyzed C-H activation followed by a cyclization cascade. The active catalytic species is generated in situ from the dimeric precursor [Ru(p-cymene)Cl2]2 upon treatment with the silver salt additive AgSbF6, which abstracts chloride ligands to create a cationic ruthenium center. This electrophilic metal center coordinates with the aryl azomethine imine, facilitating the cleavage of a proximal C-H bond on the aromatic ring. This step is critical as it forms a metallacycle intermediate that positions the substrate for subsequent nucleophilic attack by the alkyne moiety of the propargyl alcohol. The use of HFIP as a solvent is not incidental; its unique hydrogen-bonding donor capability stabilizes charged intermediates and enhances the acidity of the C-H bond, thereby accelerating the activation step. Understanding this mechanistic nuance is vital for R&D directors aiming to optimize reaction parameters or adapt the protocol for continuous flow processing.
Following the initial C-H activation and alkyne insertion, the reaction proceeds through a series of intramolecular cyclizations that forge the multiple rings of the target scaffold in a single sequence. The propargyl alcohol component undergoes dehydration and rearrangement, driven by the thermodynamic stability of the resulting conjugated system. This cascade nature of the reaction ensures that no isolable intermediates accumulate, which significantly simplifies the impurity profile of the crude reaction mixture. For quality control teams, this means fewer unknown peaks in HPLC analysis and a more straightforward purification strategy, typically achievable via standard silica gel chromatography. The robustness of the catalytic cycle is evidenced by the wide substrate scope tolerated by the system, including electron-rich and electron-deficient aryl groups, as well as various alkyl substitutions on the alkyne. This mechanistic resilience ensures consistent batch-to-batch reproducibility, a key requirement for reliable pharmaceutical intermediate supplier operations.
How to Synthesize Indenopyrazolopyrazolone Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators should adhere to the optimized conditions detailed in the patent examples. The standard protocol involves charging a reaction vessel with the aryl azomethine imine and propargyl alcohol in a molar ratio of approximately 1:1, along with 2.5 mol% of the ruthenium dimer catalyst and 10 mol% of the silver additive. The mixture is dissolved in HFIP and heated to 60°C for 2 hours with vigorous stirring. Upon completion, the reaction is cooled to ambient temperature, filtered to remove silver salts, and concentrated under reduced pressure. The resulting residue is purified by flash column chromatography using a petroleum ether and ethyl acetate gradient. Detailed standardized synthesis steps see the guide below.
- Mix aryl azomethine imine, propargyl alcohol, [Ru(p-cymene)Cl2]2 catalyst, and AgSbF6 additive in HFIP solvent.
- Heat the reaction mixture to 60°C and stir for 2 hours under sealed conditions.
- Cool to room temperature, filter, concentrate, and purify via silica gel column chromatography to obtain the target product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial strategic benefits driven by the accessibility and stability of the raw materials. Unlike previous methods that relied on specialized and unstable acetals, the propargyl alcohols and azomethine imines required here are commodity chemicals or easily synthesized from bulk precursors. This shift drastically simplifies the sourcing process, reducing lead times for high-purity pharmaceutical intermediates and mitigating the risk of supply disruptions caused by niche reagent shortages. Furthermore, the elimination of multi-step sequences in favor of a one-pot cascade reaction significantly reduces the total number of unit operations required. Fewer isolation and purification steps mean lower solvent consumption, reduced labor costs, and decreased equipment occupancy time, all of which contribute to a leaner and more cost-effective manufacturing process.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the high atom economy of the cascade reaction, where the majority of atoms from the starting materials are incorporated into the final product. By avoiding the use of expensive transition metals beyond the catalytic amount of ruthenium and eliminating the need for stoichiometric organometallic reagents, the direct material costs are significantly lowered. Additionally, the mild reaction conditions reduce energy expenditures associated with heating and cooling, while the high selectivity minimizes the loss of valuable materials to side products. These factors combine to create a manufacturing process that is inherently more economical than traditional multi-step syntheses, allowing for competitive pricing in the global market.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents ensures that inventory management is straightforward and predictable. Propargyl alcohols do not require the stringent storage conditions often necessary for sensitive organometallics or reactive acetals, reducing warehousing costs and spoilage risks. This stability extends to the reaction intermediates, which proceed rapidly to the final product without accumulating unstable species that could pose safety hazards or quality issues. For supply chain heads, this translates to a more resilient production schedule capable of meeting fluctuating demand without the bottlenecks associated with complex, fragile synthetic routes.
- Scalability and Environmental Compliance: The simplicity of the reaction setup, utilizing common solvents like HFIP and standard heating equipment, facilitates seamless scale-up from gram to kilogram scales. The reduced solvent usage and high yield inherently lower the E-factor (environmental factor) of the process, aligning with modern green chemistry principles and regulatory expectations for waste minimization. The absence of hazardous byproducts simplifies waste treatment protocols, ensuring compliance with increasingly strict environmental regulations. This scalability ensures that the transition from clinical trial material to commercial production can be achieved with minimal process re-engineering, securing long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic technology. These answers are derived directly from the experimental data and scope studies presented in the patent documentation, providing clarity on catalyst selection, substrate tolerance, and biological relevance. Understanding these details is essential for technical teams evaluating the feasibility of integrating this chemistry into their existing development pipelines.
Q: What are the key advantages of this ruthenium-catalyzed method over previous syntheses?
A: Unlike previous methods using unstable allyl acetals that yielded hydrogenated products, this novel approach utilizes stable propargyl alcohols to directly form the unsaturated indenopyrazolopyrazolone core in a single pot with high efficiency.
Q: What represents the optimal catalytic system for this transformation?
A: The patent identifies [Ru(p-cymene)Cl2]2 as the superior catalyst when paired with AgSbF6 as an additive in hexafluoroisopropanol (HFIP) solvent, achieving yields up to 92% under mild thermal conditions.
Q: Does this scaffold demonstrate significant biological activity?
A: Yes, the synthesized compounds exhibit potent anticancer activity against REC-1 and Ramos cell lines, with specific derivatives showing IC50 values in the nanomolar range, validating their potential as drug leads.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indenopyrazolopyrazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the indenopyrazolopyrazolone scaffold in modern oncology drug discovery. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from benchtop innovation to market-ready supply. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for pharmaceutical applications. We are committed to supporting your R&D efforts with reliable supply chains and technical expertise that accelerates your timeline to clinic.
We invite you to collaborate with us to leverage this efficient ruthenium-catalyzed technology for your specific drug candidates. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this streamlined synthesis can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Together, we can advance the next generation of anticancer therapeutics with speed, precision, and reliability.
