Advanced Manufacturing of Ezetimibe Intermediates via Non-Silylated Mannich Reaction
Advanced Manufacturing of Ezetimibe Intermediates via Non-Silylated Mannich Reaction
The pharmaceutical industry continuously seeks robust and cost-effective pathways for the production of high-value active pharmaceutical ingredients (APIs), and Ezetimibe remains a cornerstone in lipid-lowering therapy. A significant technological breakthrough in this domain is detailed in patent CN102850390A, which discloses a novel preparation method for Formula VI compounds and their precursors. This innovation addresses long-standing inefficiencies in the synthesis of Ezetimibe intermediates by introducing a streamlined route that bypasses the need for silyl protecting groups during the critical Mannich reaction step. By fundamentally altering the synthetic strategy, this method not only simplifies the operational workflow but also enhances the overall economic viability of the process, making it highly attractive for industrial-scale adoption.
For R&D directors and process chemists, the significance of this patent lies in its ability to generate the key intermediate, Formula V, with high yield and purity without the cumbersome protection-deprotection sequences typical of legacy methods. The direct utilization of chiral alcohol intermediates in the presence of titanium catalysts represents a sophisticated approach to stereocontrol and impurity management. This report analyzes the technical merits of this novel pathway, contrasting it with conventional methods to highlight its potential for reducing manufacturing costs and improving supply chain reliability for global pharmaceutical partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ezetimibe has been dominated by routes established by major pharmaceutical entities, such as the methods disclosed in patents CN1050830C and CN1130342C. The earlier route (CN1050830C) involves a multi-step sequence starting with the reaction of (S)-4-phenyl-2-oxazolidone and 4-chloroformyl methyl-butyrate, followed by a Mannich reaction. However, this pathway is fraught with challenges, particularly the requirement for anhydrous conditions during the zinc-mediated ketone formation step, which is highly sensitive and prone to generating impurities. Furthermore, the necessity for column chromatography purification in these older methods creates a significant bottleneck for scale-up, driving up both time and material costs substantially.
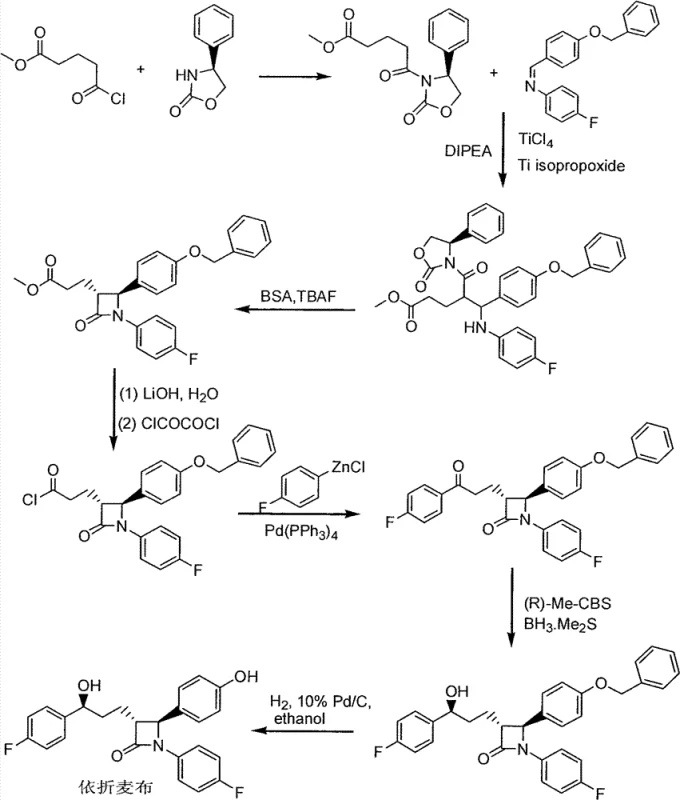
Even the improved methods, such as those in CN1130342C, which utilize chiral reduction followed by silyl protection, present distinct disadvantages. While they offer some improvements, they rely heavily on the use of hazardous silyl protecting agents like trimethylsilyl chloride and expensive chiral catalysts in large quantities. The reliance on these reagents introduces safety hazards related to toxicity and handling, while the additional steps for installing and removing the silyl groups add unnecessary complexity to the manufacturing process. These factors collectively render the conventional routes less suitable for modern, green, and cost-efficient industrial production environments.
The Novel Approach
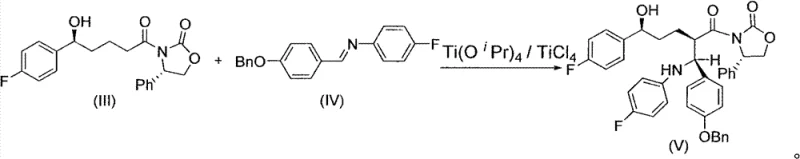
In stark contrast, the method described in CN102850390A introduces a paradigm shift by synthesizing the Formula V intermediate directly from the chiral alcohol precursor without prior silylation. This approach leverages a titanium-catalyzed Mannich reaction that tolerates the free hydroxyl group, thereby eliminating an entire class of reagents and processing steps. The result is a significantly shortened synthetic sequence that maintains high stereochemical integrity. By avoiding the use of dangerous silyl protective materials in the initial stages, the new route not only reduces the chemical footprint but also simplifies the downstream processing, allowing for more straightforward isolation techniques such as crystallization rather than chromatography.
This novel methodology effectively overcomes the technological prejudice that silyl protection is mandatory for successful Mannich reactions in this specific chemical space. The ability to proceed with unprotected intermediates demonstrates a deep understanding of the reaction mechanism and catalyst compatibility. For procurement and supply chain managers, this translates to a process that is inherently more robust and less prone to the variability associated with multi-step protection strategies. The streamlined nature of this synthesis positions it as a superior candidate for commercial scale-up, offering a clear path to reducing the cost of goods sold (COGS) for Ezetimibe production.
Mechanistic Insights into Titanium-Catalyzed Mannich Reaction
The core of this innovative synthesis lies in the precise execution of the Mannich reaction between the chiral alcohol intermediate (III) and the imine derivative (IV). The reaction is facilitated by a dual titanium catalyst system comprising Titanium Tetrachloride (TiCl4) and Titanium Isopropoxide (Ti(OiPr)4). Mechanistically, the titanium species likely coordinate with both the carbonyl oxygen of the amide side chain and the nitrogen of the imine, creating a rigid transition state that enforces high diastereoselectivity. This coordination is crucial for establishing the correct stereochemistry at the newly formed carbon-carbon bond, which is essential for the biological activity of the final Ezetimibe molecule. The reaction is typically conducted at low temperatures, ranging from -20°C to -30°C, to further suppress side reactions and ensure optimal kinetic control.
From an impurity control perspective, this mechanism offers distinct advantages over zinc-mediated alternatives. The absence of organozinc reagents eliminates the risk of over-addition or competing nucleophilic attacks that often plague metal-mediated couplings. Furthermore, the workup procedure involves a controlled quench with acetic acid and sulfuric acid, which effectively decomposes the titanium complexes and facilitates the separation of the organic product. The resulting intermediate (V) can be isolated as a white solid through crystallization from methanol, achieving a purity profile that negates the need for extensive chromatographic purification. This level of control over the reaction environment and product isolation is critical for meeting the stringent quality standards required for pharmaceutical intermediates.
How to Synthesize Ezetimibe Intermediate Efficiently
The synthesis of the key Formula VI intermediate begins with the preparation of the unprotected Mannich base (Formula V), followed by a cyclization step to form the beta-lactam ring. The process is designed to be operationally simple, utilizing common solvents like dichloromethane and toluene, and standard laboratory equipment such as three-necked bottles equipped with addition funnels and thermometers. The initial reaction requires careful temperature control to maintain the integrity of the chiral centers, while the subsequent cyclization utilizes BSA and TBAF to promote ring closure. Detailed standardized operating procedures for this synthesis are provided below to guide process implementation.
- Perform a titanium-catalyzed Mannich reaction between chiral alcohol intermediate (III) and imine (IV) using TiCl4 and Ti(OiPr)4 at low temperatures (-20 to -30°C) to obtain intermediate (V).
- Subject intermediate (V) to cyclization conditions using BSA and TBAF in toluene to form the beta-lactam ring structure of compound (VI).
- Complete the synthesis of Ezetimibe via acid hydrolysis and catalytic hydrogenation to remove protecting groups.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere technical elegance. The primary advantage lies in the substantial reduction of raw material costs and processing complexity. By eliminating the need for expensive silylating agents and the associated reagents for their removal, the overall material bill is significantly lowered. Additionally, the simplification of the workflow reduces the consumption of solvents and energy, contributing to a more sustainable and cost-effective manufacturing profile. These efficiencies are critical in a competitive market where margin optimization is a key driver of business success.
- Cost Reduction in Manufacturing: The elimination of silyl protection steps removes the cost burden of purchasing hazardous silyl chlorides and the specialized handling equipment they require. Furthermore, the avoidance of column chromatography in favor of crystallization drastically reduces silica gel consumption and solvent usage, leading to significant operational savings. The higher yield consistency of the titanium-catalyzed route ensures that less starting material is wasted, optimizing the atom economy of the entire process and lowering the effective cost per kilogram of the intermediate.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route reduces the number of potential failure points in the manufacturing process, thereby enhancing supply continuity. The reagents used, such as titanium tetrachloride and common organic solvents, are widely available commodities, reducing the risk of supply disruptions associated with specialized or proprietary catalysts. This robustness allows for more accurate demand forecasting and inventory management, ensuring that production schedules can be met reliably even in fluctuating market conditions.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations like mixing, cooling, and filtration, which are easily transferred from pilot plant to commercial scale. The reduction in hazardous waste generation, particularly from silyl byproducts and heavy metal residues, simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. This environmental stewardship not only mitigates regulatory risk but also aligns with the corporate sustainability goals of major pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of this Ezetimibe intermediate synthesis. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: How does this new method improve upon the traditional Schering routes?
A: The new method eliminates the need for silyl protection groups prior to the Mannich reaction, significantly reducing reagent costs and operational complexity compared to prior art which required hazardous silylating agents and additional protection/deprotection steps.
Q: What are the key catalysts used in this synthesis?
A: The process utilizes a combination of Titanium Tetrachloride (TiCl4) and Titanium Isopropoxide (Ti(OiPr)4) to facilitate the stereoselective Mannich reaction, ensuring high purity of the chiral intermediate.
Q: Is this process suitable for large-scale industrial production?
A: Yes, by avoiding column chromatography purification and utilizing crystallization for isolation, the process is designed for scalability, offering better yield consistency and easier waste management for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN102850390A. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory processes into robust commercial realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel route are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Ezetimibe intermediate meets the highest global standards.
We invite pharmaceutical partners to explore how this optimized synthesis can enhance your supply chain efficiency and reduce overall production costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can support your long-term strategic goals in the lipid-lowering therapeutic market.
