Revolutionizing Ezetimibe Intermediate Production: A Deep Dive into Nitrone-Based Asymmetric Synthesis
Revolutionizing Ezetimibe Intermediate Production: A Deep Dive into Nitrone-Based Asymmetric Synthesis
The pharmaceutical industry continuously seeks more efficient pathways for high-value cardiovascular drugs, and the synthesis of Ezetimibe intermediates stands as a prime example of process innovation. Patent CN102219803A introduces a groundbreaking preparation method that fundamentally alters the conventional approach to constructing the chiral azetidinone core. By shifting from traditional imine chemistry to a highly reactive nitrone-based condensation strategy, this technology addresses critical bottlenecks in yield and stereoselectivity. The method involves reacting a specific nitrone compound with a chiral carbonyl derivative in the presence of a Lewis acid catalyst, followed by a reductive workup. This technical breakthrough not only simplifies the operational steps but also drastically reduces the stoichiometric burden of expensive chiral starting materials, positioning it as a vital asset for manufacturers aiming to optimize their supply chains for cholesterol absorption inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ezetimibe intermediates has relied heavily on asymmetric aldol reactions involving imine substrates, as disclosed in earlier patents like EP720599 and WO0034240. These conventional routes face significant chemical hurdles, primarily stemming from the electronic nature of the imine functional group. The delocalization of conjugated electrons within the imine structure inherently lowers its nucleophilic activity, resulting in sluggish reaction kinetics and often necessitating harsh conditions or extended reaction times. Furthermore, imines generated from the condensation of benzaldehyde and aniline derivatives exist as a mixture of cis and trans isomers. Since the critical Evans-type transition state mechanism typically favors reaction with only the trans-isomer, manufacturers are forced to use more than two equivalents of the expensive chiral oxazolidinone auxiliary to drive the reaction to completion. This inefficiency leads to substantial waste of high-value chiral pool materials and complicates downstream purification processes due to the presence of unreacted isomers and byproducts.
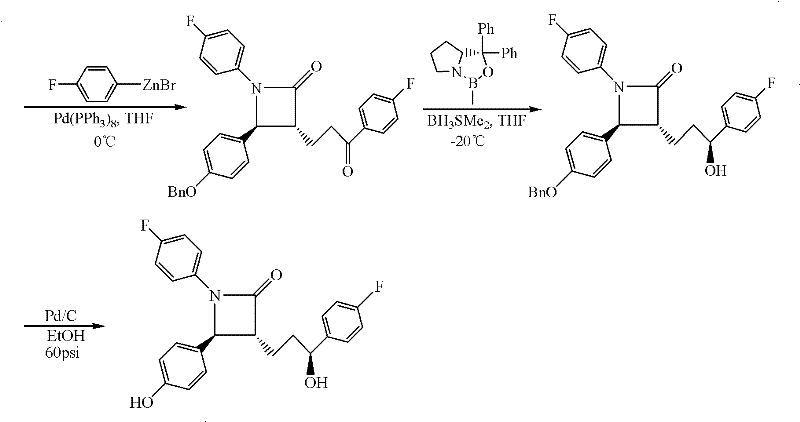
The Novel Approach
In stark contrast to the limitations of imine chemistry, the novel approach detailed in CN102219803A leverages the superior reactivity of nitrone compounds to overcome these kinetic and thermodynamic barriers. By employing a nitrone substrate, the reaction bypasses the cis-trans isomerism issues that plague imine-based routes, thereby eliminating the need for a large excess of chiral reagents. The nitrone functionality exhibits higher electrophilicity and better orbital alignment for the cycloaddition or condensation steps, allowing the reaction to proceed rapidly even at moderate low temperatures ranging from -20°C to 35°C. This method streamlines the synthetic sequence by integrating the condensation and subsequent reduction into a cohesive workflow that minimizes intermediate isolation steps. The result is a process that not only accelerates the overall production timeline but also significantly enhances the atom economy of the transformation, making it an economically superior choice for large-scale manufacturing of high-purity pharmaceutical intermediates.
Mechanistic Insights into Lewis Acid-Catalyzed Nitrone Condensation
The core of this innovative synthesis lies in the precise coordination chemistry facilitated by strong Lewis acids, specifically Titanium Tetrachloride (TiCl4) or Diisopropoxy Titanium Dichloride. When the nitrone compound and the chiral carbonyl compound are introduced into an anhydrous organic solvent such as dichloromethane, the addition of an organic base like diisopropylethylamine serves to scavenge protons and activate the nucleophile. Upon the introduction of the Lewis acid at controlled low temperatures, a rigid cyclic transition state is formed. This transition state locks the conformation of the reacting species, ensuring that the incoming nucleophile attacks the electrophilic center from the least hindered face, thus dictating the stereochemical outcome with high fidelity. The use of titanium-based catalysts is particularly effective due to their ability to coordinate simultaneously with the oxygen atoms of the nitrone and the carbonyl groups, creating a highly organized molecular architecture that favors the formation of the desired syn-aldol adduct precursor.
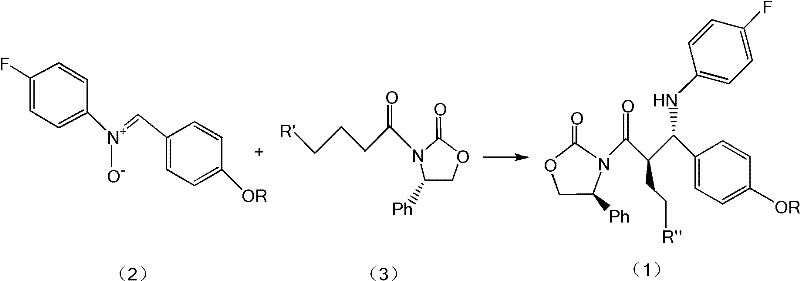
Following the condensation phase, the reaction mixture undergoes a carefully controlled quenching and reduction sequence to finalize the intermediate structure. The quenching step typically utilizes mild acidic conditions, such as glacial acetic acid or aqueous tartaric acid, to decompose the titanium complexes without degrading the sensitive product. Subsequently, the reduction is carried out using zinc powder in the presence of acetate buffers, which selectively reduces the N-O bond of the nitrone-derived adduct to form the secondary amine linkage required for the final azetidinone ring closure in later steps. This reductive workup is crucial for establishing the correct oxidation state of the nitrogen atom while preserving the stereochemical integrity of the adjacent chiral centers. The entire mechanistic pathway is designed to minimize side reactions such as epimerization or hydrolysis, ensuring that the final product meets the stringent purity specifications required for active pharmaceutical ingredient (API) synthesis.
How to Synthesize Ezetimibe Intermediate Efficiently
The practical execution of this synthesis requires strict adherence to anhydrous conditions and temperature control to maximize yield and stereoselectivity. The process begins with the dissolution of the nitrone and carbonyl precursors in dry dichloromethane under an inert nitrogen atmosphere to prevent moisture interference. Detailed standardized operating procedures for reagent addition rates, stirring speeds, and crystallization protocols are essential for reproducibility. For a comprehensive guide on the specific molar ratios, solvent volumes, and workup techniques described in the patent embodiments, please refer to the structured synthesis guide below.
- Combine nitrone compound (2) and carbonyl compound (3) in anhydrous dichloromethane under nitrogen atmosphere.
- Add organic base (e.g., DIPEA) at -10°C to 5°C, followed by Lewis acid (e.g., TiCl4) at -20°C to 35°C for condensation.
- Quench the reaction with acetic acid or tartaric acid solution, followed by zinc powder reduction to yield the target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nitrone-based technology translates into tangible strategic advantages regarding cost stability and supply reliability. The most significant economic driver is the drastic reduction in raw material consumption, specifically the elimination of the need for double equivalents of expensive chiral auxiliaries. In traditional processes, the waste of chiral pool materials represents a major cost center; by optimizing the stoichiometry to near 1:1 ratios, this new method substantially lowers the direct material cost per kilogram of the intermediate. Furthermore, the simplified reaction profile reduces the demand for complex purification infrastructure, as the cleaner reaction crude allows for more straightforward crystallization processes. This efficiency gain means that manufacturing facilities can achieve higher throughput with existing equipment, effectively increasing capacity without requiring capital-intensive expansion projects.
- Cost Reduction in Manufacturing: The primary financial benefit stems from the improved atom economy and the removal of excess chiral reagent requirements. By avoiding the use of more than two equivalents of the chiral oxazolidinone, the direct cost of goods sold is significantly optimized. Additionally, the use of common, commodity-grade solvents like dichloromethane and ethyl acetate, combined with readily available Lewis acids, ensures that the variable costs remain predictable and low. The reduction in waste generation also lowers the operational expenses associated with hazardous waste disposal and environmental compliance, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: From a sourcing perspective, this route relies on stable, commercially available starting materials that are less prone to supply disruptions compared to specialized imine precursors. The robustness of the reaction conditions, which tolerate a broader temperature window compared to cryogenic imine reactions, reduces the risk of batch failures due to minor thermal fluctuations. This reliability ensures consistent delivery schedules for downstream API manufacturers, mitigating the risk of production stoppages. The ability to source key reagents from multiple global suppliers further strengthens the supply chain resilience against geopolitical or logistical shocks.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory scale to multi-kilogram batches without loss of efficiency. The use of zinc powder for reduction is a well-established, safe industrial practice that avoids the need for high-pressure hydrogenation equipment in the early stages, simplifying the safety profile of the plant. Moreover, the aqueous workup procedures utilizing tartaric acid generate biodegradable waste streams, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing site. This compliance facilitates easier regulatory approvals and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrone-based synthesis route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction parameters and product quality. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer.
Q: Why is the nitrone-based route superior to traditional imine condensation for Ezetimibe?
A: Traditional imine routes suffer from cis-trans isomerism and low reactivity, often requiring over 2 equivalents of chiral auxiliary. The nitrone route eliminates these isomerism issues, significantly improving reaction speed and atom economy.
Q: What catalysts are used in this novel synthesis method?
A: The process utilizes strong Lewis acids such as Titanium Tetrachloride (TiCl4) or Diisopropoxy Titanium Dichloride to facilitate the asymmetric aldol-type condensation at low temperatures.
Q: How is the stereochemistry controlled in this reaction?
A: Stereocontrol is achieved through the use of chiral oxazolidinone auxiliaries on the carbonyl component and the specific transition state geometry enforced by the Lewis acid coordination.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving cardiovascular medications. Our technical team has extensively analyzed the nitrone-based synthesis route and possesses the expertise to implement this advanced chemistry at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of Ezetimibe intermediate we produce meets the highest international standards for chirality and chemical purity.
We invite you to collaborate with us to leverage this cutting-edge technology for your pharmaceutical projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this novel route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to drive efficiency and innovation in your supply chain.
