Advanced Synthesis of Trimethoxy-Substituted Arabinose Triazole Spiroisoxazole-Pyrrolizine Derivatives for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds that combine high biological potency with synthetic feasibility, and patent CN112028955B presents a significant breakthrough in this domain by disclosing a trimethoxy-substituted arabinose triazole structure spiroisoxazole-pyrrolizine derivative. This specific chemical entity represents a sophisticated fusion of three distinct pharmacophores: the pyrrolizine alkaloid core, the bioactive 1,2,3-triazole moiety, and the solubility-enhancing arabinose glycoside, all linked through a rigid spiroisoxazole junction. The strategic design of this molecule addresses critical challenges in oncology drug development, particularly regarding water solubility and target specificity, while the disclosed preparation method offers a robust pathway for industrial production. By leveraging a convergent synthetic strategy centered around a 1,3-dipolar cycloaddition, the inventors have established a route that is not only chemically elegant but also practically viable for the manufacture of high-purity pharmaceutical intermediates. This report analyzes the technical nuances of this patent to provide R&D directors, procurement managers, and supply chain heads with actionable insights into adopting this technology for next-generation anticancer agent production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing complex spirocyclic heterocycles often suffer from poor atom economy, harsh reaction conditions, and a lack of stereochemical control, which can severely impact the scalability and cost-effectiveness of the final active pharmaceutical ingredient. Conventional routes frequently rely on linear syntheses where the spiro center is formed late in the sequence, leading to cumulative yield losses and difficult purification steps due to the formation of regioisomers. Furthermore, the introduction of sensitive glycosidic units typically requires extensive protecting group manipulations under aggressive acidic or basic conditions that risk hydrolyzing the sugar moiety or epimerizing chiral centers. These limitations create significant bottlenecks for supply chain reliability, as batch-to-batch consistency becomes difficult to maintain when the chemistry is inherently fragile. Additionally, the use of expensive transition metal catalysts or exotic reagents in older methodologies drives up the cost of goods sold, making the resulting therapeutic candidates less commercially attractive in a competitive generic and biosimilar market landscape.
The Novel Approach
In stark contrast, the methodology described in patent CN112028955B employs a highly efficient convergent strategy that constructs the complex spiroisoxazole-pyrrolizine core in a single, high-yielding transformation. The process begins with the straightforward preparation of a key dipolarophile, (E)-2-(3,4,5-trimethoxybenzylidene)-2,3-dihydropyrrolizin-1-one, via a simple aldol condensation that utilizes commodity chemicals like 3,4,5-trimethoxybenzaldehyde.
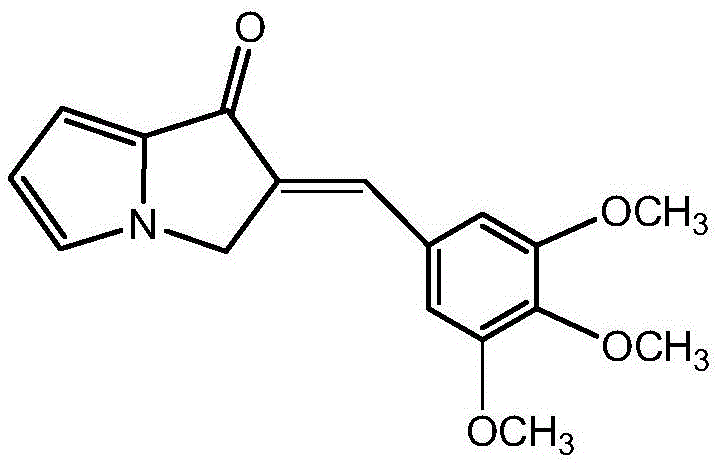
This intermediate is then coupled with a pre-functionalized arabinose triazole oxime through a 1,3-dipolar cycloaddition, a reaction known for its excellent regioselectivity and ability to generate molecular complexity rapidly. This approach minimizes the number of isolation steps and avoids the use of heavy metal catalysts, thereby simplifying the downstream purification process and reducing the environmental footprint. The final deprotection step is conducted under mild alkaline conditions at room temperature, preserving the integrity of the sensitive glycosidic linkage and ensuring the final product meets stringent purity specifications required for clinical applications without the need for extensive recrystallization cycles.
Mechanistic Insights into 1,3-Dipolar Cycloaddition and Spiro Formation
The cornerstone of this synthetic route is the 1,3-dipolar cycloaddition reaction between the nitrile oxide dipole, generated in situ from the acetyl arabinose triazole salicylaldoxime, and the electron-deficient exocyclic double bond of the pyrrolizine ketone. In the presence of Chloramine-T as an oxidant, the oxime undergoes dehydration to form the reactive nitrile oxide species, which immediately engages in a [3+2] cycloaddition with the alkene. This pericyclic reaction proceeds through a concerted mechanism that ensures high stereospecificity, locking the relative configuration of the newly formed isoxazole ring and the spiro center. The electronic nature of the trimethoxyphenyl group on the pyrrolizine ring enhances the electrophilicity of the double bond, facilitating the attack by the dipole and driving the reaction to completion with minimal formation of byproducts. This mechanistic precision is critical for R&D teams, as it guarantees a consistent impurity profile and eliminates the need for challenging chiral separations that would otherwise be required if the spiro center were formed via non-stereoselective alkylation.
Following the cycloaddition, the acetyl protecting groups on the arabinose sugar are removed via transesterification using sodium methoxide in methanol. This step is mechanistically straightforward yet vital for the biological activity of the final molecule, as the free hydroxyl groups on the sugar are essential for hydrogen bonding interactions with biological targets such as enzymes or receptors. The use of a strong acid cation exchange resin to neutralize the reaction mixture post-deprotection is a clever engineering choice that avoids the introduction of inorganic salts, which can be difficult to remove from polar glycosylated products. This attention to mechanistic detail ensures that the final trimethoxy-substituted arabinose triazole structure spiroisoxazole-pyrrolizine derivative is obtained as a pale yellow solid with high chemical purity, ready for biological evaluation or further formulation development.
How to Synthesize Trimethoxy-Substituted Arabinose Triazole Spiroisoxazole-Pyrrolizine Derivative Efficiently
The synthesis of this high-value intermediate is designed for operational simplicity and robustness, making it an ideal candidate for technology transfer from laboratory to pilot plant scales. The process flow involves four distinct stages: the initial condensation to form the pyrrolizine core, the preparation of the glycosylated oxime dipole, the key cycloaddition step to fuse the rings, and the final global deprotection. Each step utilizes standard unit operations such as reflux, filtration, and column chromatography, which are well-understood by process chemists and easily implemented in GMP facilities. The detailed standardized synthesis steps, including precise molar ratios, solvent volumes, and temperature profiles, are outlined in the structured guide below to facilitate immediate replication and optimization by your technical teams.
- Synthesize (E)-2-(3,4,5-trimethoxybenzylidene)-2,3-dihydropyrrolizin-1-one via aldol condensation of 2,3-dihydropyrrolizin-1-one and 3,4,5-trimethoxybenzaldehyde in ethanol with NaOH.
- Prepare acetyl arabinose triazole salicylaldoxime through the dehydration reaction of acetyl arabinose triazole salicylaldehyde with hydroxylamine hydrochloride.
- Perform 1,3-dipolar cycloaddition by reacting the pyrrolizine ketone with the oxime in the presence of Chloramine-T in refluxing ethanol to form the spiroisoxazole core.
- Execute deprotection by treating the acetylated intermediate with sodium methoxide in methanol at room temperature, followed by neutralization and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route disclosed in this patent offers substantial advantages over legacy methods, primarily driven by the availability of starting materials and the simplicity of the reaction conditions. The key building blocks, such as 3,4,5-trimethoxybenzaldehyde and 2,3-dihydropyrrolizin-1-one, are commodity chemicals available from multiple global suppliers, which mitigates the risk of single-source dependency and ensures stable pricing dynamics. Furthermore, the elimination of precious metal catalysts removes the need for expensive scavenging resins and rigorous heavy metal testing, directly translating to reduced manufacturing costs and shorter release times for quality control. The mild conditions employed throughout the synthesis, particularly the room temperature deprotection, reduce energy consumption and minimize the thermal stress on equipment, enhancing the overall sustainability and safety profile of the production process.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing Chloramine-T, a low-cost and readily available oxidant, instead of expensive hypervalent iodine reagents or transition metal catalysts often required for similar oxidative cyclizations. The high atom economy of the 1,3-dipolar cycloaddition means that the majority of the mass of the reactants ends up in the final product, minimizing waste disposal costs associated with low-yielding side reactions. Additionally, the purification strategy relies on standard recrystallization and column chromatography with common solvent systems like chloroform and methanol, avoiding the need for specialized preparative HPLC or complex extraction protocols that drive up operational expenditures in large-scale facilities.
- Enhanced Supply Chain Reliability: By designing a convergent synthesis where two major fragments are coupled in the final stages, the supply chain becomes more resilient to disruptions in the availability of any single raw material. The intermediates generated in the early steps, such as the pyrrolizine ketone, are stable solids that can be stockpiled, allowing for flexible production scheduling and buffer inventory management. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without compromising yield, ensures consistent batch quality even when scaling up from kilograms to metric tons, thereby guaranteeing reliable delivery timelines to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable as it avoids hazardous reagents and extreme pressure or temperature conditions that pose engineering challenges in large reactors. The use of ethanol and methanol as primary solvents aligns with green chemistry principles, as these are Class 3 solvents with low toxicity and are easily recovered and recycled through distillation. The absence of heavy metals simplifies the environmental compliance burden, reducing the cost of wastewater treatment and hazardous waste disposal. This eco-friendly profile not only lowers the total cost of ownership but also aligns with the increasingly strict environmental, social, and governance (ESG) criteria demanded by top-tier pharmaceutical companies when selecting contract development and manufacturing organizations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel spiroisoxazole-pyrrolizine derivative, based on the specific data and embodiments provided in the patent literature. These answers are designed to clarify the feasibility of the process and the potential value proposition for stakeholders involved in oncology drug development.
Q: What represents the key pharmacological advantage of this spiroisoxazole-pyrrolizine derivative?
A: The compound exhibits potent inhibitory effects against multiple tumor cell lines, including SW620 (colon), A549 (lung), and SGC7901 (gastric) cancers, due to the synergistic integration of the glycosylated triazole and spiroisoxazole pharmacophores which enhance water solubility and receptor binding affinity.
Q: How does the 1,3-dipolar cycloaddition strategy improve synthetic efficiency?
A: This method utilizes a highly regioselective 1,3-dipolar cycloaddition between a nitrile oxide and an exocyclic double bond, allowing for the rapid construction of the complex spirocyclic skeleton in a single step with excellent stereocontrol, bypassing the need for tedious multi-step ring closures.
Q: Is the deprotection step compatible with large-scale manufacturing?
A: Yes, the deprotection utilizes mild conditions (sodium methoxide in methanol at room temperature) which avoids the use of harsh acidic or basic environments that could degrade the sensitive glycosidic bonds, ensuring high yield and purity suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trimethoxy-Substituted Arabinose Triazole Spiroisoxazole-Pyrrolizine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this trimethoxy-substituted arabinose triazole structure spiroisoxazole-pyrrolizine derivative as a critical intermediate for next-generation antitumor therapies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency and quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, including advanced analytical methods to characterize complex spirocyclic structures and verify the absence of genotoxic impurities. We understand that time-to-market is crucial in the oncology sector, and our dedicated process chemistry team is ready to optimize this patented route to maximize yield and minimize cost, providing you with a competitive edge in the global marketplace.
We invite you to engage with our technical procurement team to discuss your specific requirements for this high-purity pharmaceutical intermediate. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic viability of this synthesis at your desired scale. We encourage you to request specific COA data and route feasibility assessments to validate how our manufacturing capabilities can support your R&D and commercial goals. Let us collaborate to bring this promising anticancer candidate from the laboratory bench to the patients who need it most, leveraging our expertise in complex heterocyclic chemistry and supply chain management.
