Advanced Catalyst-Free Synthesis of 1,4-Naphthoquinone for Commercial Scale-Up
The chemical landscape for producing vital fine chemical intermediates is constantly evolving, driven by the dual demands of regulatory compliance and economic efficiency. A pivotal advancement in this domain is documented in patent CN102391091A, which outlines a novel synthetic method for 1,4-naphthoquinone, a critical building block in the manufacture of pharmaceuticals, agrochemicals, and functional dyes. Traditional manufacturing routes often rely on harsh oxidants or expensive catalytic systems that introduce significant environmental burdens and supply chain complexities. This specific intellectual property proposes a streamlined, catalyst-free oxidation pathway that utilizes hydrogen peroxide and acetic anhydride to generate peracetic acid in situ. For R&D Directors and Procurement Managers seeking a reliable 1,4-naphthoquinone supplier, understanding the mechanistic elegance and operational safety of this patent is essential for evaluating long-term sourcing strategies. The technology promises not only a reduction in raw material costs but also a substantial mitigation of process safety risks associated with handling unstable peroxy-compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 1,4-naphthoquinone has been plagued by inefficiencies inherent to older oxidation technologies. Conventional methods frequently employ stoichiometric amounts of heavy metal oxidants such as chromium trioxide or sodium dichromate, which generate vast quantities of toxic heavy metal waste that require costly disposal and remediation procedures. Furthermore, catalytic approaches utilizing transition metals like vanadium or ruthenium, while potentially more efficient, introduce the risk of metal contamination in the final API intermediate, necessitating rigorous and expensive purification steps to meet stringent pharmaceutical impurity profiles. Another significant bottleneck in traditional processes is the handling of peracids; isolating and distilling peracetic acid is notoriously dangerous due to its thermal instability and potential for explosive decomposition at elevated temperatures. These factors collectively inflate the cost of goods sold (COGS) and create fragile supply chains vulnerable to regulatory crackdowns on environmental emissions and workplace safety incidents.
The Novel Approach
The methodology disclosed in CN102391091A represents a paradigm shift by eliminating the need for external catalysts and avoiding the isolation of hazardous intermediates. By reacting hydrogen peroxide and acetic anhydride directly in the reaction vessel, the process generates the active oxidizing species, peracetic acid, exactly where and when it is needed. This in-situ generation strategy ensures that the concentration of the unstable peracid remains low throughout the reaction, drastically enhancing the safety profile of the operation. The absence of transition metal catalysts means there is no risk of metal leaching into the product stream, simplifying the downstream purification workflow and ensuring a cleaner impurity profile from the outset. This approach not only aligns with green chemistry principles by reducing waste but also offers a compelling economic advantage by removing the cost of catalyst procurement and recovery, making it an attractive option for cost reduction in fine chemical manufacturing.
Mechanistic Insights into In-situ Peracetic Acid Oxidation
The core of this innovative synthesis lies in the controlled formation and immediate consumption of peracetic acid. The initial step involves the equilibrium reaction between hydrogen peroxide and acetic anhydride. Under mild conditions, typically maintained between 1°C and 40°C, these reagents combine to form peracetic acid and acetic acid as a byproduct. This step is crucial because it avoids the accumulation of high-energy peroxy species. The reaction kinetics are managed by controlling the molar ratio of hydrogen peroxide to acetic anhydride, typically ranging from 1:1 to 4:1, ensuring complete conversion of the anhydride while maintaining a safe excess of oxidant. The absence of an acid catalyst in this step is particularly noteworthy, as it demonstrates that the reaction proceeds efficiently under neutral conditions, further reducing the corrosive load on reactor equipment and minimizing the formation of side products that often arise from acid-catalyzed degradation pathways.
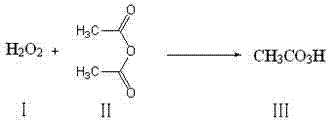
Once the peracetic acid is generated, the second phase of the reaction involves the oxidation of alpha-naphthol. The alpha-naphthol, dissolved in a suitable organic solvent such as methanol or ethanol, is added dropwise to the reaction mixture. The temperature is carefully regulated, usually between 30°C and 80°C, to facilitate the electrophilic attack of the peracid on the naphthalene ring system. This oxidation converts the phenolic hydroxyl group and the adjacent aromatic carbon into the quinone carbonyl functionalities. The mechanism likely proceeds through a Baeyer-Villiger type oxidation intermediate or a direct electron transfer process, resulting in the formation of the 1,4-dicarbonyl structure characteristic of 1,4-naphthoquinone. The simplicity of this two-step one-pot sequence minimizes unit operations, reducing both capital expenditure on equipment and operational labor costs.
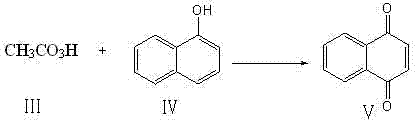
Controlling the impurity profile in this synthesis is achieved through precise stoichiometry and temperature management. Excess hydrogen peroxide can lead to over-oxidation or ring cleavage, while insufficient oxidant results in unreacted starting material. The patent specifies optimal molar ratios, such as 1:6 to 1:10 for alpha-naphthol to acetic anhydride, to maximize yield while minimizing byproduct formation. Following the reaction, the workup procedure is designed to remove acidic byproducts and unreacted materials efficiently. The use of aqueous bicarbonate washes neutralizes residual acetic acid and peracetic acid, while solvent extraction separates the organic product from the aqueous phase. Final recrystallization from cyclohexane ensures that the high-purity 1,4-naphthoquinone meets the rigorous specifications required for high-purity pharmaceutical intermediates, effectively addressing the concerns of R&D teams regarding product quality and consistency.
How to Synthesize 1,4-Naphthoquinone Efficiently
Implementing this synthesis route requires careful attention to the order of addition and thermal control to ensure safety and reproducibility. The process begins with the preparation of the oxidizing solution, followed by the controlled addition of the substrate. Detailed standard operating procedures (SOPs) are critical for scaling this chemistry from the laboratory to commercial production. The following guide outlines the fundamental steps derived from the patent examples to assist technical teams in evaluating the feasibility of this route for their specific manufacturing needs.
- Generate peracetic acid in situ by mixing hydrogen peroxide and acetic anhydride at controlled temperatures.
- Add alpha-naphthol solution directly to the peracetic acid mixture and maintain reaction temperature between 30-80°C.
- Perform workup via extraction, washing, and recrystallization to isolate high-purity 1,4-naphthoquinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free technology offers tangible strategic benefits beyond mere technical feasibility. The elimination of expensive transition metal catalysts directly translates to a reduction in raw material costs, which is a primary driver for cost reduction in electronic chemical manufacturing and pharma intermediate production. Furthermore, the simplified process flow, which avoids the distillation of hazardous peracids, reduces the energy consumption and specialized equipment requirements typically associated with high-risk chemical processes. This operational simplicity enhances supply chain reliability by minimizing the potential for unplanned shutdowns due to safety incidents or equipment failures. The use of commodity chemicals like hydrogen peroxide and acetic anhydride ensures a stable supply of raw materials, mitigating the risk of shortages that often plague specialty catalyst markets.
- Cost Reduction in Manufacturing: The most significant economic advantage of this process is the complete removal of catalyst costs. Traditional methods often rely on precious metals or complex organocatalysts that represent a substantial portion of the variable cost. By utilizing a catalyst-free system, manufacturers can achieve substantial cost savings without compromising on reaction efficiency. Additionally, the avoidance of metal removal steps, such as scavenger resin treatment or extensive filtration, reduces the consumption of auxiliary materials and shortens the overall batch cycle time. This streamlined workflow allows for higher throughput in existing facilities, effectively increasing capacity without the need for significant capital investment in new infrastructure.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of sourcing specialized reagents. This synthesis method relies on bulk commodities that are widely available from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, adds another layer of resilience to the manufacturing process. In the event of supply disruptions for a specific solvent, the patent indicates flexibility in solvent selection, including alcohols, esters, and hydrocarbons. This adaptability ensures that production schedules can be maintained even when specific raw material grades are temporarily unavailable, thereby reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges related to heat transfer and mass transfer, particularly with exothermic oxidations. The in-situ generation of peracetic acid in this method inherently manages heat release by controlling the addition rate of reagents, making the process safer and easier to scale from pilot plant to full commercial production. From an environmental perspective, the absence of heavy metals simplifies wastewater treatment and reduces the regulatory burden associated with hazardous waste disposal. This alignment with green chemistry principles not only lowers compliance costs but also enhances the corporate sustainability profile, which is increasingly important for multinational corporations aiming to meet ESG (Environmental, Social, and Governance) targets.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is crucial for stakeholders evaluating its implementation. The following questions address common concerns regarding safety, purity, and scalability, providing clarity based on the data presented in the patent documentation. These insights are intended to facilitate informed decision-making for technical and commercial teams alike.
Q: What are the primary safety advantages of this synthesis method?
A: This method eliminates the need to distill peracetic acid, which is prone to explosion at high temperatures (110°C). By generating it in situ and consuming it immediately, the process significantly reduces thermal risks.
Q: Does this process require expensive transition metal catalysts?
A: No, the patent CN102391091A explicitly describes a catalyst-free approach. This removes the cost of precious metals and the complex downstream processing required to remove metal residues from the final product.
Q: How is product purity ensured without chromatography?
A: The process utilizes a robust workup procedure involving solvent extraction, aqueous washing to remove acidic byproducts, and final recrystallization using cyclohexane to achieve high purity suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Naphthoquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust manufacturing capabilities. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative chemistries like the catalyst-free synthesis of 1,4-naphthoquinone can be realized efficiently. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards. We understand that for R&D Directors, consistency is key, and our advanced analytical capabilities ensure that impurity profiles are tightly controlled, meeting the exacting requirements of global pharmaceutical and fine chemical clients.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality expectations. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make data-driven decisions about integrating this high-efficiency intermediate into your production portfolio. Partnering with NINGBO INNO PHARMCHEM means gaining access to not just a product, but a comprehensive technical solution designed to enhance your competitive edge in the global market.
