Advanced Green Synthesis of 2-Methyl-1,4-Naphthoquinone for Industrial Pharmaceutical Applications
The global demand for high-purity Vitamin K3, chemically known as 2-methyl-1,4-naphthoquinone, continues to surge across the pharmaceutical and animal nutrition sectors, driving an urgent need for sustainable and scalable manufacturing technologies. Patent CN109384659B introduces a groundbreaking preparation method that fundamentally shifts the synthetic paradigm from hazardous oxidation processes to a green, base-catalyzed condensation strategy. This innovation addresses critical pain points faced by R&D Directors and Supply Chain Heads, specifically the elimination of heavy metal contaminants and the reliance on expensive, unstable intermediates. By utilizing phthalic diesters and 3-cyanobutyrate as starting materials, the disclosed technology offers a robust pathway to achieve yields exceeding 90% while maintaining exceptional product purity. For procurement professionals seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in process safety and economic viability, ensuring a stable supply chain for this essential coagulant agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-methyl-1,4-naphthoquinone has been plagued by severe environmental and operational inefficiencies inherent to traditional oxidation and cyclization routes. One prevalent method involves the direct oxidation of 2-methylnaphthalene using stoichiometric amounts of chromium trioxide or manganese dioxide, which inevitably generates vast quantities of wastewater contaminated with toxic heavy metals, posing immense disposal costs and regulatory risks. Another common approach, depicted in the reaction scheme below, utilizes m-cresol as a raw material to generate 2-methylbenzoquinone, followed by a Diels-Alder reaction with 1,3-butadiene; however, this route suffers from the poor stability of the benzoquinone intermediate and low selectivity during the cycloaddition step, leading to complex impurity profiles that are difficult to purge.
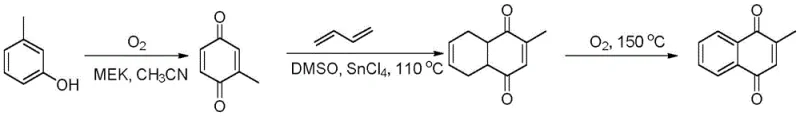
Furthermore, alternative pathways starting from 1-naphthol require harsh methylation and subsequent oxidation conditions that often result in over-oxidation byproducts and reduced overall yields. These conventional methodologies not only burden the cost reduction in pharmaceutical intermediate manufacturing initiatives due to expensive raw materials and waste treatment but also compromise the continuity of supply due to stringent environmental restrictions on heavy metal discharge. The reliance on unstable intermediates and toxic oxidants creates a fragile production ecosystem that is increasingly untenable in the modern green chemistry landscape.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN109384659B leverages a sophisticated condensation-hydrolysis-decarboxylation sequence that bypasses the need for transition metal oxidants entirely. This innovative route initiates with the condensation of phthalic diesters and 3-cyanobutyrate under the catalysis of strong alkali, forming a key cyclic intermediate that is subsequently hydrolyzed and aromatized. The strategic selection of phthalic acid derivatives as the aromatic core ensures high structural fidelity and minimizes side reactions, while the use of 3-cyanobutyrate introduces the necessary methyl group and carbon framework with high atom economy. This methodology effectively transforms the synthesis into a cleaner, more predictable process that aligns perfectly with the requirements for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Base-Catalyzed Condensation and Cyclization
The core of this technological breakthrough lies in the precise mechanistic orchestration of a Claisen-type condensation followed by intramolecular cyclization, driven by strong bases such as sodium methoxide or potassium tert-butoxide. In the initial stage, the strong base deprotonates the alpha-carbon of the 3-cyanobutyrate ester, generating a highly nucleophilic enolate species that attacks the carbonyl carbon of the phthalic diester. This nucleophilic acyl substitution is carefully controlled within a temperature range of 90-110°C in solvents like toluene or xylene, facilitating the removal of alcohol byproducts to drive the equilibrium forward. The resulting intermediate undergoes a rapid intramolecular cyclization to form the naphthalene backbone, setting the stage for the subsequent functional group transformations required to achieve the quinone structure.
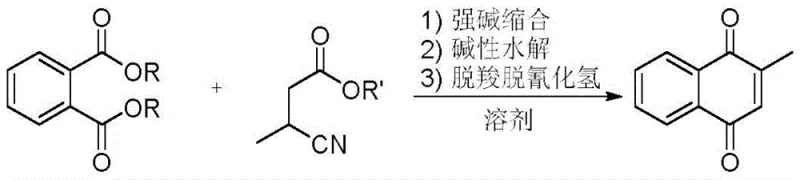
Following the condensation, the process employs a mild alkaline hydrolysis step using sodium hydroxide at ambient temperatures (20-30°C) to convert the ester functionalities into carboxylate salts without degrading the sensitive ring system. The final transformation is achieved through acidification with hydrochloric acid, which triggers a simultaneous decarboxylation and dehydrocyanation cascade. This dual elimination reaction is thermodynamically favorable and proceeds efficiently at moderate temperatures (50-80°C), releasing carbon dioxide and hydrogen cyanide. Crucially, the evolved hydrogen cyanide can be captured and recycled to synthesize more 3-cyanobutyrate, creating a closed-loop system that enhances the overall sustainability and reducing lead time for high-purity pharmaceutical intermediates by minimizing raw material procurement cycles.
How to Synthesize 2-Methyl-1,4-Naphthoquinone Efficiently
The operational protocol for this synthesis is designed for seamless integration into existing multipurpose reactors, requiring standard equipment for heating, distillation, and phase separation. The process begins by charging a solvent such as toluene into a reactor and heating it to the optimal condensation temperature before the controlled addition of the ester substrates and the base catalyst. Detailed standard operating procedures regarding reagent ratios, addition rates, and specific workup techniques are critical for maximizing yield and ensuring safety during the handling of cyanide-containing intermediates.
- Condense phthalic diester with 3-cyanobutyrate in a solvent like toluene using a strong base such as sodium methoxide at 90-110°C.
- Perform alkaline hydrolysis on the condensation product using an inorganic base solution (e.g., NaOH) at mild temperatures (20-30°C).
- Acidify the mixture with hydrochloric acid to induce simultaneous decarboxylation and dehydrocyanation, yielding the final quinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patent-protected methodology offers transformative advantages that extend far beyond simple yield improvements. By fundamentally altering the chemical architecture of the synthesis, the process eliminates the most costly and logistically challenging aspects of traditional Vitamin K3 production, specifically the management of hazardous heavy metal waste and the sourcing of volatile oxidants. This shift translates directly into a more resilient supply chain capable of withstanding regulatory pressures and fluctuating raw material markets, ensuring consistent delivery of high-purity pharmaceutical intermediates to downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic metal oxidants such as chromium and manganese removes the substantial costs associated with their purchase, storage, and the complex wastewater treatment required for their disposal. Furthermore, the use of commodity chemicals like phthalic esters and the potential for recycling hydrogen cyanide byproducts significantly lowers the raw material cost basis, driving down the overall cost of goods sold without compromising quality.
- Enhanced Supply Chain Reliability: Unlike routes dependent on unstable intermediates like 2-methylbenzoquinone or scarce natural precursors, this method utilizes robust, shelf-stable esters that are widely available in the global chemical market. This abundance of feedstock ensures that production schedules are not disrupted by raw material shortages, providing a dependable source of supply for long-term contracts and just-in-time manufacturing requirements.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently in batch reactors with straightforward workup procedures involving phase separation and distillation. The absence of heavy metal discharge simplifies environmental permitting and reduces the risk of production shutdowns due to non-compliance, making it an ideal candidate for large-scale industrial expansion in regions with strict ecological regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their current manufacturing portfolios.
Q: How does this new synthesis method improve environmental compliance compared to traditional routes?
A: Unlike traditional methods that utilize chromic acid or manganese dioxide oxidants which generate toxic heavy metal wastewater, this patented process relies on organic ester condensation and avoids transition metal catalysts entirely, significantly reducing hazardous waste discharge.
Q: What are the primary raw materials required for this 2-methyl-1,4-naphthoquinone preparation?
A: The process utilizes readily available and cost-effective phthalic acid diesters (such as dimethyl phthalate) and 3-cyanobutyrate esters, which can be synthesized via the addition of hydrogen cyanide to 2-butenoate, ensuring high atom economy.
Q: What yield and purity levels can be expected from this industrial scale-up process?
A: Experimental data from the patent indicates that the method consistently achieves high reaction selectivity with product yields exceeding 90% and gas chromatography purity levels reaching up to 99.7%, making it highly suitable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-1,4-Naphthoquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, green synthesis technologies to meet the evolving demands of the global pharmaceutical and agrochemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-methyl-1,4-naphthoquinone adheres to the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this cutting-edge preparation method for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and product quality.
