Revolutionizing Chiral Cephalotaxine Production: A Scalable 10-Step Synthetic Route
Revolutionizing Chiral Cephalotaxine Production: A Scalable 10-Step Synthetic Route
The pharmaceutical landscape for anti-leukemia agents continues to evolve, driven by the need for more efficient and cost-effective manufacturing processes for complex alkaloids. Patent CN109776506B, published in June 2022, introduces a groundbreaking synthetic methodology for chiral (-)-cephalotaxine and its intermediates. This technology addresses long-standing challenges in the total synthesis of cephalotaxine esters, which are potent inhibitors of protein synthesis effective against various forms of leukemia, including acute myelocytic leukemia. Unlike previous methods that relied on extraction from plant sources or lengthy semi-synthetic routes, this invention proposes a concise 10-step total synthesis starting from the industrial raw material furan. The strategic design of this pathway not only simplifies the construction of the unique [4.4.0]-azaspiro benzocycloheptamine skeleton but also ensures high atom economy and operational simplicity, positioning it as a superior candidate for commercial scale-up.
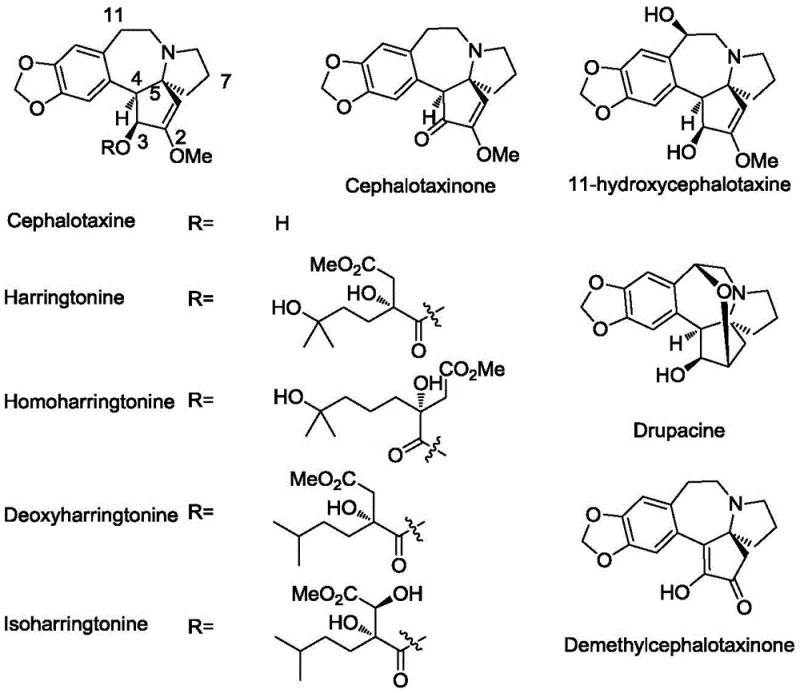
The structural complexity of cephalotaxine derivatives, such as homoharringtonine (Synribo), demands precise stereochemical control. The disclosed method achieves this through a novel rearrangement reaction catalyzed by Lewis acids, transforming a linear precursor into the requisite spirocyclic core with high fidelity. By leveraging robust chemical transformations and avoiding excessive protecting group manipulations, this route offers a compelling alternative to historical syntheses. For procurement and supply chain leaders, this represents a potential shift towards more reliable sourcing of high-purity pharmaceutical intermediates, reducing dependency on variable natural extraction yields.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cephalotaxine has been fraught with inefficiencies that hinder large-scale commercial viability. Early efforts, such as the Weinreb group's racemic synthesis in the 1970s, established the foundational chemistry but lacked stereocontrol, necessitating resolution steps that halve theoretical yield. Subsequent chiral syntheses, like those by Mori and Gin, while scientifically elegant, often suffered from excessive step counts and low overall atom economy. These traditional routes frequently require multiple protection and deprotection sequences to manage the reactivity of sensitive functional groups, leading to increased waste generation and higher production costs. Furthermore, reliance on chiral pool starting materials like proline or sugar derivatives can introduce supply chain bottlenecks and price volatility. The cumulative effect of these factors is a manufacturing process that is difficult to scale, expensive to operate, and environmentally burdensome due to the high volume of solvent and reagent consumption per kilogram of final product.
The Novel Approach
In stark contrast, the methodology described in CN109776506B streamlines the synthesis into a mere 10 steps from furan, a commodity chemical. The core innovation lies in the construction of the chiral center early in the sequence using (R)-tert-butylsulfinamide, which directs the stereochemistry throughout the subsequent transformations without the need for intermediate resolutions. The pivotal step involves a Lewis acid-catalyzed rearrangement of an acyclic precursor (Compound A) to the cyclic ketone (Compound B), effectively building the complex spiro-framework in a single operation. This approach eliminates the need for the cumbersome ring-closing strategies seen in prior art, such as Friedel-Crafts cyclizations under harsh conditions. By minimizing the number of unit operations and utilizing robust catalysts like stannous triflate or binaphthol phosphate, the new route significantly enhances process robustness. This efficiency translates directly into reduced manufacturing lead times and lower cost of goods sold (COGS), making it an attractive option for generic API manufacturers seeking to optimize their supply chains.
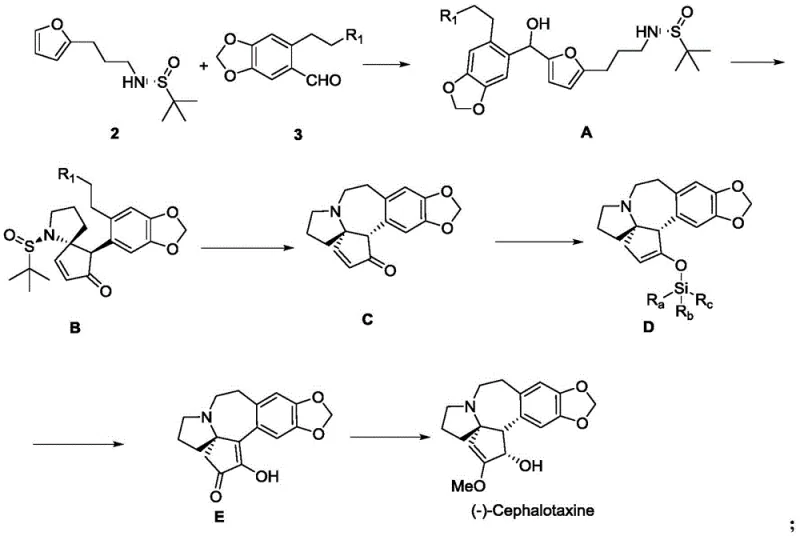
Mechanistic Insights into Lewis Acid-Catalyzed Rearrangement
The heart of this synthetic strategy is the transformation of Compound A into Compound B, a process that constructs the critical five-membered ring and establishes the quaternary stereocenter simultaneously. This reaction proceeds under the influence of a Lewis acid, which activates the leaving group on the side chain, facilitating an intramolecular nucleophilic attack or rearrangement cascade. The patent specifies a range of effective catalysts, including strong metal salts like stannous trifluoromethanesulfonate (Sn(OTf)2) and copper trifluoromethanesulfonate, as well as organic acids like binaphthol phosphate. The choice of catalyst allows for fine-tuning of reaction conditions; for instance, strong metal salts enable the reaction to proceed at room temperature in solvents like ethylene glycol dimethyl ether, whereas weaker Lewis acids may require elevated temperatures. This mechanistic flexibility is crucial for process chemists aiming to optimize reaction kinetics and minimize side reactions. The high yields reported for this step, reaching up to 76% with Sn(OTf)2, underscore the efficiency of this bond-forming event.
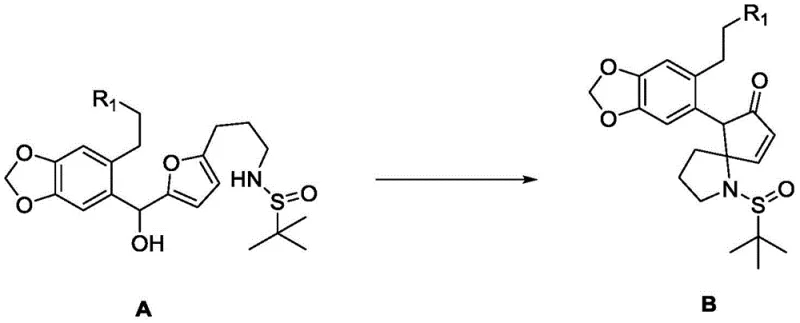
Impurity control is inherently managed by the stereospecific nature of the sulfinamide auxiliary and the concerted mechanism of the rearrangement. The chiral information encoded in the starting amine is faithfully transferred to the product, minimizing the formation of diastereomeric impurities that would otherwise require difficult chromatographic separations. Furthermore, the use of mild reaction conditions helps preserve the integrity of other sensitive functionalities within the molecule, such as the methylenedioxy group on the aromatic ring. Post-reaction work-up involves standard aqueous quenching and extraction, followed by purification via column chromatography, which is scalable to industrial standards. The ability to monitor reaction progress via TLC or HPLC ensures precise endpoint determination, preventing over-reaction or decomposition. This level of control is essential for maintaining the stringent purity specifications required for pharmaceutical intermediates intended for clinical use.
How to Synthesize (-)-Cephalotaxine Efficiently
The synthesis of (-)-cephalotaxine via this patented route is designed for operational simplicity and high throughput. The process begins with the preparation of a chiral amine building block from furan, followed by coupling with a functionalized aldehyde to generate the rearrangement precursor. The subsequent Lewis acid-mediated cyclization forms the core scaffold, which is then elaborated through hydrosilylation, oxidation, and final reductive functionalization. Each step has been optimized for yield and scalability, utilizing common laboratory reagents and solvents. For detailed procedural specifics, including exact stoichiometry, temperature profiles, and work-up protocols, please refer to the standardized synthesis guide below which outlines the critical operational parameters derived from the patent examples.
- Preparation of chiral amine intermediate via iodination and reaction with (R)-tert-butylsulfinamide.
- Coupling with aldehyde derivative followed by Lewis acid catalyzed rearrangement to form the core spiro-cycle.
- Final functionalization including hydrosilylation, oxidation, and reductive methoxylation to yield (-)-cephalotaxine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical elegance. The shift from extraction-dependent or multi-step semi-synthetic methods to a concise 10-step total synthesis fundamentally alters the cost structure and risk profile of cephalotaxine production. By utilizing furan as a starting material, manufacturers can bypass the agricultural variability and seasonal constraints associated with plant extraction. This transition to petrochemical-derived feedstocks ensures a consistent, year-round supply of raw materials, insulating the supply chain from environmental disruptions. Moreover, the reduction in step count directly correlates with a reduction in capital expenditure on equipment and labor, as fewer batch cycles are required to produce the same amount of final product. This efficiency gain allows for more competitive pricing models in the global marketplace for anti-leukemia therapeutics.
- Cost Reduction in Manufacturing: The elimination of multiple protection and deprotection steps significantly reduces the consumption of reagents and solvents, which are major cost drivers in fine chemical manufacturing. Additionally, the high yields observed in key steps, such as the rearrangement and hydrosilylation reactions, minimize material loss and waste disposal costs. The use of commercially available catalysts like Karstedt's catalyst and tin salts further avoids the expense of proprietary or exotic reagents. Consequently, the overall cost of goods is drastically simplified and lowered, enabling more aggressive pricing strategies for generic versions of cephalotaxine esters.
- Enhanced Supply Chain Reliability: Relying on industrial raw materials like furan and standard reagents such as sodium borohydride and triethylsilane mitigates the risk of supply shortages. Unlike specialized chiral pool materials that may have limited suppliers, the inputs for this route are commoditized and widely available from multiple global vendors. This diversification of the supply base enhances resilience against geopolitical or logistical disruptions. Furthermore, the robustness of the chemical transformations ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed batches or out-of-specification results.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, utilizing reaction conditions that are readily transferable from laboratory to pilot and commercial scales. The avoidance of hazardous reagents and the minimization of waste streams align with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance. The simplified work-up procedures, often involving straightforward extractions and crystallizations, reduce the burden on wastewater treatment facilities. This environmental stewardship not only lowers compliance costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN109776506B, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing supplier capabilities. The clarity provided here aims to bridge the gap between academic innovation and industrial application.
Q: What is the primary advantage of this new cephalotaxine synthesis route?
A: The primary advantage is the significant reduction in synthetic steps to just 10, starting from inexpensive industrial raw materials like furan, compared to traditional routes that require extensive protection and deprotection sequences.
Q: Which catalysts are used in the key rearrangement step?
A: The process utilizes Lewis acids such as stannous trifluoromethanesulfonate (Sn(OTf)2), copper trifluoromethanesulfonate, or binaphthol phosphate to facilitate the critical rearrangement of the intermediate compound A to compound B.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the patent explicitly states the method features high reaction yields, simple operations, and uses industrially available starting materials, making it highly expected for application in industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-Cephalotaxine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this 10-step synthetic route for the global supply of cephalotaxine intermediates. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this patented methodology into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised in the lab are realized on the factory floor. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the quality of every batch. Our commitment to excellence extends beyond mere synthesis; we offer comprehensive process optimization services to further enhance yield and reduce environmental impact.
We invite pharmaceutical companies and generic manufacturers to collaborate with us to secure a stable and cost-effective supply of high-purity cephalotaxine. By leveraging our capabilities, you can accelerate your drug development timelines and bring life-saving treatments to market faster. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing prowess can support your supply chain objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
