Advanced Two-Step Synthesis of Chiral 1,2-Bis(2,5-Diphenylphosphino)Ethane for Industrial Catalysis
The landscape of asymmetric catalysis is continually evolving, driven by the demand for more efficient and safer synthetic routes for high-value chiral ligands. A significant breakthrough in this domain is documented in patent CN110878104A, which discloses a robust preparation method for chiral 1,2-bis(2,5-diphenylphosphino)ethane, a critical precursor for rhodium-based catalysts used in the synthesis of non-natural chiral amino acids. This technology represents a paradigm shift from hazardous multi-step sequences to a streamlined, industrially viable two-step protocol. By leveraging a Grignard coupling strategy followed by a mild silane-mediated reduction, the process addresses long-standing safety concerns while maintaining exceptional stereochemical integrity. For R&D directors and procurement specialists alike, understanding the nuances of this patented methodology is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 1,2-bis(2,5-diphenylphosphino)ethane has been plagued by significant operational hazards and inefficiencies that hinder large-scale adoption. Traditional literature routes, such as those described in Organic Letters and the European Journal of Organic Chemistry, typically rely on a convoluted four-step sequence involving extremely dangerous reagents. These legacy methods often necessitate the use of phenyltrihydrosilane or lithium aluminum hydride, both of which are highly pyrophoric and pose severe fire risks during handling and storage. Furthermore, the requirement for borane reagents and butyllithium introduces additional layers of complexity, demanding cryogenic conditions and rigorous exclusion of moisture and oxygen. The cumulative effect of these hazardous steps not only escalates the cost of goods sold due to specialized safety infrastructure but also creates bottlenecks in production scheduling, making it difficult for suppliers to guarantee consistent delivery timelines for bulk orders.
The Novel Approach
In stark contrast, the innovative process outlined in the patent data offers a dramatically simplified pathway that mitigates these risks while enhancing overall yield and purity. By condensing the synthesis into just two primary chemical transformations, the new method eliminates the need for the most volatile reagents associated with the older protocols. The initial step utilizes a controlled Grignard coupling in tetrahydrofuran, a standard solvent with well-understood handling properties, to construct the carbon-phosphorus backbone. This is followed by a deoxygenation step using trichlorosilane and a tertiary amine in toluene, which proceeds under reflux conditions rather than the extreme低温 required by butyllithium. This strategic redesign of the synthetic route not only improves process safety but also facilitates cost reduction in pharmaceutical intermediate manufacturing by reducing solvent consumption, waste generation, and reactor occupancy time, thereby offering a compelling value proposition for commercial scale-up.
Mechanistic Insights into Grignard Coupling and Silane Reduction
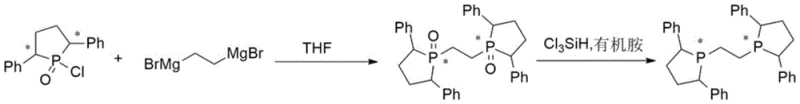
The core of this technological advancement lies in the precise orchestration of nucleophilic substitution and subsequent reductive deoxygenation. In the first stage, the reaction exploits the nucleophilicity of the 1,2-dibromoethane magnesium salt to attack the electrophilic phosphorus center of the chiral 2,5-diphenylphosphonyl chloride. This coupling event is critical for establishing the ethane bridge between the two phosphine oxide moieties while preserving the stereocenters located on the pyrrolidine rings. The patent specifies a molar ratio of approximately 1:2.2 to 1:3.3 between the Grignard reagent and the phosphonyl chloride, ensuring complete conversion while minimizing side reactions. Maintaining the addition temperature between 0°C and 10°C is paramount to controlling the exotherm and preventing racemization, a detail that underscores the importance of precise thermal management in achieving the reported 98.5% to 98.8% chemical purity in the intermediate stage.
Following the isolation of the bis-phosphine oxide, the second mechanistic phase involves the reduction of the phosphoryl (P=O) bond to the phosphine (P-H/P-R) state using a trichlorosilane-amine system. This transformation proceeds through the formation of a silyl-phosphonium intermediate, which is subsequently reduced to the target phosphine. The use of triethylamine or N,N-dimethylaniline serves to scavenge the hydrochloric acid generated during the reaction, driving the equilibrium forward and preventing the protonation of the sensitive phosphine product. This method is superior to metal hydride reductions because it avoids the introduction of metallic impurities that are notoriously difficult to remove from catalytic ligands. The result is a final product with chiral purity exceeding 99%, as confirmed by NMR data, making it ideally suited for high-turnover asymmetric hydrogenation reactions where trace impurities can poison expensive rhodium catalysts.
How to Synthesize Chiral 1,2-Bis(2,5-Diphenylphosphino)Ethane Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding solvent selection and stoichiometry to ensure reproducibility at scale. The process begins with the preparation of the Grignard reagent in tetrahydrofuran, which is then carefully dosed into the chiral phosphonyl chloride solution under an inert atmosphere. Following the coupling and workup, the resulting oxide is dissolved in toluene for the reduction step, where the ratio of trichlorosilane to amine is carefully balanced to maximize conversion without generating excessive salts. While the general chemistry is straightforward, the devil is in the details regarding temperature control and quenching procedures. For a comprehensive breakdown of the specific operational parameters, including exact addition rates and crystallization conditions, please refer to the standardized synthesis guide below.
- Perform Grignard coupling by dropwise adding 1,2-dibromoethane magnesium salt to chiral 2,5-diphenylphosphonyl chloride in THF at 0-10°C, then reflux to form the bis-phosphine oxide intermediate.
- Dissolve the isolated phosphine oxide intermediate in toluene and add trichlorosilane along with an organic amine base such as triethylamine.
- Heat the mixture to reflux for deoxygenation, quench with aqueous sodium hydroxide, and crystallize the final product from ethanol to achieve over 99% chiral purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented two-step process offers tangible benefits that extend far beyond simple chemical yield improvements. The elimination of high-risk reagents like butyllithium and lithium aluminum hydride fundamentally alters the risk profile of the manufacturing operation, leading to substantial cost savings in terms of insurance, safety equipment, and waste disposal. Moreover, the use of commodity solvents such as THF and toluene ensures that raw material sourcing remains stable and unaffected by the supply volatility often seen with specialty reagents. This stability translates directly into enhanced supply chain reliability, allowing manufacturers to commit to longer-term contracts with confidence. Additionally, the reduction in unit operations from four steps to two significantly decreases the lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands and reducing the working capital tied up in work-in-progress inventory.
- Cost Reduction in Manufacturing: The streamlined nature of this process drives down manufacturing costs through multiple mechanisms, primarily by removing the need for expensive and hazardous reagents that require specialized handling protocols. By avoiding cryogenic conditions and pyrophoric materials, the facility overheads related to safety infrastructure are drastically reduced, allowing for more competitive pricing structures. Furthermore, the higher overall yield achieved through fewer purification steps means less raw material is wasted, directly improving the atom economy and lowering the cost per kilogram of the final active ligand. These efficiencies compound over large production runs, resulting in significant margin improvements for downstream users of these catalytic precursors.
- Enhanced Supply Chain Reliability: Dependence on niche, high-danger reagents often creates single points of failure in a supply chain, whereas this new method relies on widely available bulk chemicals. The robustness of the Grignard and silane reduction chemistry ensures that production campaigns are less likely to be interrupted by reagent shortages or quality deviations. This reliability is crucial for pharmaceutical clients who require consistent batch-to-batch quality to support their own regulatory filings. By stabilizing the upstream production of these critical ligands, suppliers can offer more predictable delivery schedules, reducing the need for customers to hold excessive safety stock and freeing up their own logistical resources.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the reduction in step count inherently reduces the volume of solvent waste and energy consumption per unit of product. The avoidance of heavy metal hydrides simplifies the effluent treatment process, making it easier to comply with increasingly stringent environmental regulations. This green chemistry advantage is not just a compliance checkbox but a strategic asset, as it future-proofs the manufacturing site against regulatory tightening. The process is designed to be scalable from pilot plant quantities to multi-ton commercial production without requiring fundamental changes to the reaction engineering, ensuring a smooth transition from R&D to full-scale manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy routes. Understanding these details is vital for technical teams evaluating the feasibility of adopting this new standard for ligand production.
Q: What are the safety advantages of this new synthesis route compared to traditional methods?
A: The novel process eliminates the use of highly pyrophoric reagents like phenyltrihydrosilane and lithium aluminum hydride, replacing them with safer Grignard reagents and trichlorosilane systems that are easier to handle on an industrial scale.
Q: What level of chiral purity can be achieved with this manufacturing method?
A: According to the patent data, the optimized process consistently yields chiral 1,2-bis(2,5-diphenylphosphino)ethane with a chiral purity exceeding 99%, meeting the stringent requirements for asymmetric catalysis applications.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reduction of reaction steps from four to two, combined with the use of common solvents like THF and toluene, significantly simplifies the workflow and enhances the feasibility of scaling up to multi-ton production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful deployment of advanced catalytic technologies depends on the availability of high-quality ligands produced via robust and scalable processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of chiral 1,2-bis(2,5-diphenylphosphino)ethane meets the exacting standards required for asymmetric hydrogenation applications. Our infrastructure is designed to handle complex organometallic chemistries safely, providing our partners with a secure and dependable source for their critical raw materials.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this safer, more efficient manufacturing method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your supply chain is built on a foundation of technical excellence and commercial viability.
