Advanced Acidic Boron-Complex Route for Commercial Curcumin Manufacturing
Advanced Acidic Boron-Complex Route for Commercial Curcumin Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient, scalable, and environmentally compliant pathways for producing high-value bioactive compounds. A significant breakthrough in this domain is documented in patent CN111116339B, which details a novel method for artificially synthesizing curcumin and its derivatives. Unlike traditional extraction methods that suffer from low natural abundance (approximately 1% in turmeric rhizomes) and difficult separation of structural analogues, this synthetic approach offers a robust alternative. The core innovation lies in utilizing a weak acid condition to facilitate the reaction between acetylacetone and boron oxide, forming a protective complex that reacts with benzaldehyde derivatives. This strategic modification not only enhances reaction selectivity far beyond that of conventional alkaline systems but also drives macroscopic yields from a historical average of roughly 60% to an impressive 80-90%. For a reliable curcumin supplier, mastering this technology represents a pivotal shift towards cost-effective and high-purity manufacturing capabilities.
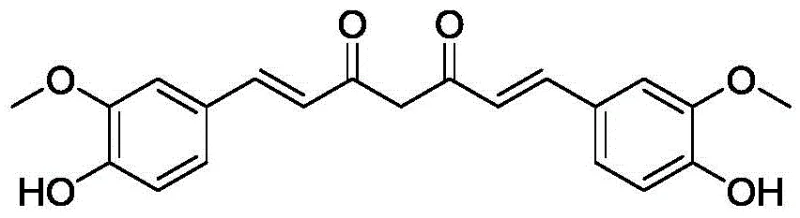
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of curcumin has been plagued by significant operational inefficiencies and environmental drawbacks. The standard industrial practice typically involves an aldol condensation reaction under alkaline conditions, which imposes severe requirements for anhydrous environments to prevent side reactions. To maintain these dry conditions, manufacturers are forced to use excessive amounts of tri-n-butyl borate as a water scavenger, a reagent that is not only costly but also complicates the downstream processing. Furthermore, the reaction degree in these alkaline systems is often incomplete, leading to a mixture of products that are difficult to separate from reactants and solvents. The post-treatment steps are notoriously complicated, requiring rigorous purification to remove residual boron species and byproducts, resulting in substantial three-waste generation (wastewater, waste gas, and solid waste) that fails to meet modern environmental protection trends. Consequently, the overall yield rarely exceeds 60%, making the process economically suboptimal for large-scale cost reduction in food additive manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN111116339B introduces a paradigm shift by operating under weak acidic conditions. This approach leverages the formation of a stable complex between acetylacetone and boron oxide (or other boron-containing compounds like boric acid), which effectively protects the active methylene group between the two ketocarbonyls. Once this complex is formed, it reacts with various benzaldehyde derivatives—such as vanillin—in the presence of a specific catalyst to generate a curcumin derivative intermediate. The most striking advantage of this route is the dramatic improvement in selectivity; the acidic environment suppresses unwanted side reactions that are prevalent in alkaline media. Moreover, the hydrolysis of the intermediate to obtain the final curcumin derivative can be achieved completely at normal temperature, eliminating the need for the energy-intensive heating (60-80°C) required by older methods. This results in a crude product with almost zero insoluble substances, ensuring that the obtained product is inherently purer and easier to recrystallize to pharmaceutical grades.
Mechanistic Insights into Boron-Mediated Acidic Aldol Condensation
To fully appreciate the technical superiority of this synthesis, one must understand the mechanistic role of the boron complex. In the initial stage, acetylacetone reacts with boron oxide to form a cyclic boron chelate. This chelation serves a dual purpose: it activates the methylene protons for nucleophilic attack while simultaneously protecting the carbonyl oxygens from undesirable interactions. When a catalyst, such as 1,2,3,4-tetrahydroquinoline or piperidine, is introduced along with an organic acid like acetic acid, the system maintains a precise pH balance that favors the condensation with the aldehyde substrate. The reaction proceeds through a controlled aldol-type mechanism where the boron-stabilized enol equivalent attacks the carbonyl carbon of the benzaldehyde derivative. This pathway ensures that the condensation occurs specifically at the desired positions, minimizing the formation of mono-condensed byproducts or polymeric tars that often contaminate alkaline reactions. The result is the formation of Intermediate I, a bis-boron complex that retains the structural integrity of the heptadiene-dione backbone essential for curcumin's bioactivity.
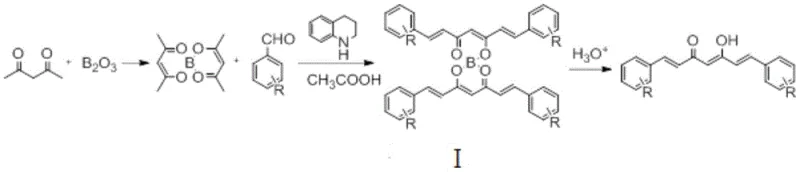
Following the condensation, the hydrolysis step is critical for releasing the free curcumin molecule from the boron scaffold. In traditional processes, this step often requires harsh acidic conditions and elevated temperatures, which can degrade the sensitive conjugated double bond system of curcumin. However, the novel method described allows for hydrolysis using a phosphoric acid aqueous solution at room temperature. The mildness of this hydrolysis is attributed to the specific lability of the boron-oxygen bonds in the intermediate under these optimized acidic conditions. This gentle release mechanism prevents thermal degradation and isomerization, thereby preserving the stereochemistry and purity of the final product. The ability to perform this cleavage without generating significant insoluble impurities means that the crude product obtained after filtration is already of high quality (often exceeding 90% content), significantly reducing the burden on the final recrystallization step and enhancing the overall commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Curcumin Efficiently
The implementation of this synthesis route requires careful attention to reagent ratios and reaction parameters to maximize the benefits of the boron-complex strategy. The process begins by dissolving acetylacetone, a boron-containing complexing agent (preferably boron oxide), and the chosen benzaldehyde derivative in a polar aprotic solvent such as dimethyl sulfoxide (DMSO) or sulfolane. An organic acid is added to establish the necessary weak acidic environment, followed by the dropwise addition of a nitrogen-containing catalyst. The reaction is typically conducted at temperatures between 60-120°C for a duration of 3-8 hours. Upon completion, the mixture is poured into an acidic aqueous solution to precipitate the product, which is then filtered and dried. For detailed operational parameters, stoichiometry, and safety protocols, please refer to the standardized synthesis guide below.
- Dissolve acetylacetone, boron oxide (complexing agent), and a benzaldehyde derivative (e.g., vanillin) in a polar aprotic solvent like DMSO.
- Add an organic acid (e.g., acetic acid) and a catalyst (e.g., 1,2,3,4-tetrahydroquinoline) to maintain weak acidic conditions and initiate the condensation.
- React at 60-120°C for 3-8 hours, then hydrolyze the intermediate at room temperature using phosphoric acid solution to isolate the pure curcumin derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this acidic boron-complex synthesis offers tangible strategic advantages that extend beyond mere chemical yield. The elimination of strict anhydrous requirements fundamentally alters the cost structure of production. By removing the necessity for expensive water scavengers like tri-n-butyl borate and the associated drying agents, the raw material costs are significantly reduced. Furthermore, the simplified workup procedure—characterized by room temperature hydrolysis and easy filtration—drastically cuts down on processing time and energy consumption. This streamlining of the manufacturing workflow translates directly into improved throughput and lower operational expenditures, providing a compelling case for cost reduction in fine chemical manufacturing without compromising on product quality.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of costly reagents with inexpensive alternatives like boron oxide and acetic acid. Traditional methods rely heavily on tri-n-butyl borate, which acts as a dehydrating agent but adds substantial material costs and waste disposal fees. By shifting to a system where water management is less critical due to the acidic mechanism, manufacturers can eliminate this expense entirely. Additionally, the higher reaction selectivity means less raw material is wasted on byproducts, improving the atom economy of the process. The reduction in purification steps further lowers the consumption of solvents and adsorbents, creating a leaner, more cost-efficient production model that enhances margin potential for bulk buyers.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex synthesis routes that are prone to batch failures or long cycle times. The robustness of the acidic boron-complex method mitigates these risks by offering a wider operating window and forgiving reaction conditions. The ability to use common solvents like DMSO and readily available catalysts reduces dependency on specialized or scarce reagents. Moreover, the shortened reaction and workup times allow for faster turnover of production batches, enabling suppliers to respond more agilely to market demand fluctuations. This reliability is crucial for maintaining steady inventory levels of high-purity curcumin and ensuring that downstream pharmaceutical or nutraceutical production schedules are not disrupted by raw material shortages.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial waste intensify, the environmental profile of a synthesis route becomes a key factor in vendor selection. This novel method generates significantly less boric acid wastewater compared to the traditional two-pot alkaline process, simplifying effluent treatment and reducing the environmental footprint. The absence of harsh hydrolysis conditions also lowers energy usage, contributing to a greener manufacturing profile. These factors make the process highly scalable, as the engineering challenges associated with heat removal and waste neutralization are minimized. Companies adopting this technology can confidently scale from pilot plants to multi-ton production facilities, assured that they are meeting both economic and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of curcumin derivatives using the patented acidic boron-complex method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this technology outperforms legacy systems. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing supply chains.
Q: What is the primary advantage of the acidic boron-complex method over traditional alkaline synthesis?
A: The acidic method eliminates the need for strict anhydrous conditions and expensive water scavengers like tri-n-butyl borate. It achieves significantly higher selectivity and yields (80-90% vs. ~60%) while allowing hydrolysis at room temperature, which simplifies purification.
Q: Which benzaldehyde derivatives are compatible with this synthesis route?
A: The process is highly versatile and supports a wide range of substrates including vanillin, isovanillin, p-hydroxybenzaldehyde, p-methoxybenzaldehyde, salicylaldehyde, and 3,4,5-trihydroxybenzaldehyde, enabling the production of various curcumin analogues.
Q: How does this method impact environmental compliance and waste generation?
A: By avoiding excessive tri-n-butyl borate and utilizing a one-pot hydrolysis strategy, the method drastically reduces boric acid wastewater and organic solvent consumption, aligning better with modern green chemistry standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Curcumin Supplier
The technological advancements detailed in patent CN111116339B underscore the immense potential for producing high-quality curcumin and its analogues with superior efficiency. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of such chemical innovations, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to excellence is reflected in our stringent purity specifications and rigorous QC labs, which ensure that every batch of curcumin or derivative meets the highest international standards for pharmaceutical and food-grade applications. We possess the technical expertise to navigate the complexities of boron-mediated chemistry, guaranteeing a consistent supply of materials that support your R&D and commercial needs.
We invite you to explore how our advanced manufacturing capabilities can optimize your supply chain and reduce your overall procurement costs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us partner with you to deliver reliable, high-performance curcumin solutions that drive your business forward.
