Advanced Metal-Free Synthesis of Indanone Compounds for Commercial Scale-Up
Advanced Metal-Free Synthesis of Indanone Compounds for Commercial Scale-Up
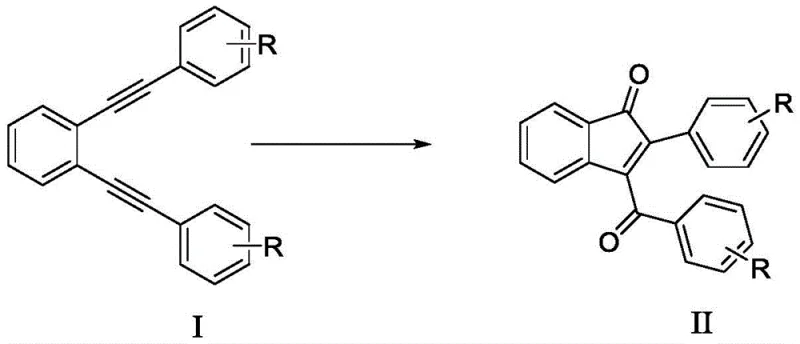
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign pathways to access privileged scaffolds like indanones. Patent CN109422631B introduces a groundbreaking synthetic methodology that transforms 1,2-bis(phenylethynyl)benzene derivatives into valuable indanone compounds through a novel oxidative cyclization process. This technology represents a significant departure from classical approaches by utilizing diiodine pentoxide as a stoichiometric oxidant in a mixed solvent system containing water. By operating under mild thermal conditions, specifically around 50°C, this process mitigates the safety risks associated with high-temperature reactions while maintaining high product selectivity. For R&D directors and process chemists, this patent offers a compelling alternative that aligns with modern green chemistry principles, ensuring that the production of these critical intermediates is both efficient and sustainable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indanone core has relied heavily on Friedel-Crafts acylation or cyclization strategies, which are fraught with significant operational and environmental drawbacks. These traditional methods typically necessitate the use of strong Lewis acids or protic acids, such as aluminum chloride or polyphosphoric acid, which generate substantial quantities of corrosive waste acid and acidic wastewater containing organic residues. Furthermore, the electrophilic nature of these reactions imposes severe limitations on substrate scope; aromatic rings bearing strong electron-withdrawing groups, such as nitro or carbonyl functionalities, often fail to undergo cyclization or result in prohibitively low yields. The reliance on harsh acidic conditions also complicates downstream processing, requiring extensive neutralization and purification steps that increase both the cost of goods sold and the environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the methodology disclosed in CN109422631B employs a metal-free oxidative cyclization strategy that circumvents the pitfalls of acid-catalyzed routes. By utilizing diiodine pentoxide in conjunction with water and an organic co-solvent like acetonitrile, the reaction proceeds efficiently at moderate temperatures without the need for transition metal catalysts. This approach not only eliminates the generation of heavy metal waste but also demonstrates remarkable tolerance for various substituents on the aromatic rings, including methyl, bromo, and propyl groups. The inclusion of water as a reactant serves as a hydroxyl source, facilitating the formation of the carbonyl moiety directly within the cyclization step. This streamlined process significantly simplifies the workup procedure, as it avoids the complex quenching and extraction protocols associated with strong acid methodologies, thereby enhancing overall process efficiency.
Mechanistic Insights into I2O5-Mediated Oxidative Cyclization
The mechanistic pathway of this transformation is driven by the unique oxidizing capability of diiodine pentoxide in the presence of water, which acts as a nucleophilic oxygen source. Unlike transition metal-catalyzed cycles that often involve complex coordination spheres and ligand exchanges, this reaction likely proceeds through an iodine-mediated activation of the alkyne moieties. The water molecules participate directly in the bond-forming events, providing the oxygen atom required to establish the ketone functionality at the 1-position of the indanone ring. This direct incorporation of oxygen from water is a distinct advantage, as it removes the need for pre-functionalized oxygen donors or hazardous peroxides. The absence of metal catalysts ensures that the final product is free from trace metal impurities, a critical quality attribute for pharmaceutical intermediates intended for subsequent coupling reactions or biological testing.
Furthermore, the reaction conditions are meticulously optimized to balance reactivity and selectivity, preventing over-oxidation or polymerization of the sensitive diyne starting materials. The use of a mixed solvent system, preferably acetonitrile and water in a 4:1 volume ratio, creates a homogeneous medium that solubilizes both the organic substrate and the inorganic oxidant effectively. This solubility profile is crucial for maintaining consistent reaction kinetics throughout the 12-hour reaction period. The mild thermal profile, centered at 50°C, provides sufficient activation energy to drive the cyclization forward while minimizing thermal degradation of the product. Such precise control over the reaction environment results in high purity crude mixtures, reducing the burden on purification columns and improving the overall mass balance of the synthesis.
How to Synthesize 3-Benzoyl-2-phenyl-1H-inden-1-one Efficiently
The practical execution of this synthesis involves charging a pressure-resistant reactor with the specific diyne precursor and the oxidant, followed by the addition of the solvent mixture. The protocol emphasizes the importance of maintaining the correct stoichiometric ratio between the oxidant and the substrate, typically using a slight excess of diiodine pentoxide to ensure complete conversion. Following the reaction period, the workup involves a straightforward adsorption of the product onto silica gel followed by solvent removal, which streamlines the isolation process compared to traditional liquid-liquid extractions.
- Charge a pressure-resistant reactor with the diyne starting material (Formula I) and diiodine pentoxide oxidant.
- Add a mixed solvent system comprising an organic solvent such as acetonitrile and water in a specific volume ratio.
- Stir the mixture at 50°C for 12 hours, then purify the resulting reaction liquid via column chromatography to isolate the target indanone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this metal-free methodology offers transformative benefits that directly impact the bottom line and operational reliability. The elimination of expensive transition metal catalysts, such as palladium or rhodium complexes, removes a significant cost driver from the raw material bill of materials. Moreover, the absence of heavy metals simplifies the regulatory compliance landscape, as there is no need for specialized resin treatments or rigorous ICP-MS testing to meet stringent ppm limits for residual metals in API intermediates. This simplification translates into faster batch release times and reduced analytical costs, allowing for a more agile response to market demands. The use of commodity chemicals like acetonitrile and water further stabilizes the supply chain against volatility in specialty reagent pricing.
- Cost Reduction in Manufacturing: The economic advantages of this process are multifaceted, stemming primarily from the drastic simplification of the downstream processing workflow. By avoiding the use of strong mineral acids and Lewis acids, the facility saves significantly on waste disposal costs associated with neutralizing and treating hazardous acidic effluents. The mild reaction conditions also contribute to energy savings, as maintaining a temperature of 50°C requires far less utility consumption than the high-temperature reflux conditions often needed for traditional cyclizations. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, leading to higher isolated yields and better utilization of starting materials, which collectively drive down the cost per kilogram of the final indanone product.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing dependency on scarce or geopolitically sensitive catalytic metals. The starting diyne compounds are accessible through established Sonogashira coupling protocols, and the oxidant, diiodine pentoxide, is a stable and commercially available reagent. This reliance on stable, non-proprietary reagents mitigates the risk of supply disruptions that can occur with specialized catalysts. Furthermore, the tolerance of the reaction to various functional groups means that a single platform technology can be applied to synthesize a wide range of indanone derivatives, allowing manufacturers to pivot quickly between different product SKUs without retooling or extensive process re-validation.
- Scalability and Environmental Compliance: Scaling this process to multi-ton production is facilitated by its inherent safety profile and lack of exothermic hazards associated with strong acid additions. The generation of waste is significantly reduced, aligning with increasingly strict global environmental regulations regarding industrial effluent. The aqueous component of the solvent system reduces the overall load of volatile organic compounds (VOCs) emitted during the process. This environmental compatibility not only lowers the cost of environmental permits and waste treatment but also positions the manufacturer as a sustainable partner for multinational corporations with aggressive carbon reduction and green chemistry goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the practical application of the method. Understanding these nuances is essential for process engineers and technical buyers evaluating the feasibility of adopting this route for their specific supply chains.
Q: What are the advantages of this metal-free method over traditional Friedel-Crafts cyclization?
A: Unlike traditional Friedel-Crafts methods which require harsh Lewis acids and generate significant acidic waste, this novel process utilizes diiodine pentoxide in a mild aqueous-organic system. It eliminates the need for transition metal catalysts, thereby avoiding costly heavy metal removal steps and allowing for substrates with electron-withdrawing groups that typically fail in acid-catalyzed routes.
Q: Can this synthesis tolerate diverse functional groups on the aromatic rings?
A: Yes, the method demonstrates excellent substrate universality. The patent data confirms successful cyclization for substrates containing methyl, bromo, and propyl substituents. This tolerance is critical for synthesizing complex pharmaceutical intermediates where specific functionalization is required for downstream biological activity.
Q: What is the role of water in this oxidative cyclization reaction?
A: Water plays a dual role as both a co-solvent and a reactant. It acts as the hydroxyl source necessary for the formation of the carbonyl functionality in the indanone ring. Its participation allows for a greener solvent system and contributes to the high selectivity observed in the transformation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes like the metal-free oxidative cyclization described in CN109422631B for the production of high-value indanone intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality assurance ensures that our clients receive intermediates that are ready for immediate use in downstream API synthesis without additional purification burdens.
We invite potential partners to engage with our technical procurement team to discuss how this advanced methodology can be tailored to your specific project requirements. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient process. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our indanone compounds in your own synthesis workflows. Let us collaborate to optimize your supply chain and accelerate the development of your next-generation therapeutic candidates.
