Revolutionizing Pharmaceutical Intermediate Production: High-Yield One-Step Synthesis of Complex Methylene Indanones for Commercial Scale-Up
Patent CN112961086A introduces a transformative one-step synthesis methodology for producing structurally diverse 2-methylene-1-indanone derivatives that serve as critical building blocks in pharmaceutical development pipelines due to their prevalence in bioactive natural products and drug candidates targeting neurological disorders and oncology applications. This innovative approach leverages transition metal catalysis to directly construct the indanone core from commercially accessible S,S-dithioketal compounds and diazo reagents under mild thermal conditions, eliminating the multi-step pre-functionalization sequences that characterize conventional synthetic routes while achieving consistently high yields exceeding eighty percent through optimized reaction parameters. The process demonstrates exceptional functional group tolerance across diverse substitution patterns on both aromatic rings and aliphatic chains, enabling medicinal chemists to rapidly generate structural analogs for structure-activity relationship studies without requiring protective group strategies that typically reduce overall efficiency. By streamlining the synthetic pathway into a single operation with straightforward workup procedures involving standard silica gel chromatography purification, this methodology addresses fundamental industry challenges related to production complexity and scalability while maintaining stringent purity requirements essential for pharmaceutical intermediates. The resulting derivatives exhibit remarkable structural diversity that can be precisely tailored for specific pharmacological targets, making them indispensable components in next-generation drug discovery programs where molecular complexity directly correlates with therapeutic efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for indanone derivatives typically involve laborious multi-step sequences requiring pre-functionalized starting materials under harsh reaction conditions that generate significant side products through competing pathways such as aldol condensations or retro-Michael reactions, ultimately leading to low cumulative yields due to repeated purification losses at each intermediate stage. For instance, methodologies disclosed in CN1277809C necessitate bromination using hazardous reagents like N-bromosuccinimide followed by multiple substitution reactions and selective reductions before final condensation steps, creating substantial operational complexity while introducing safety hazards associated with unstable intermediates that require specialized handling protocols. These approaches suffer from poor atom economy as evidenced by methyl mercaptan release during spiroindanone formation in CN108675921A, which not only complicates environmental compliance but also increases waste treatment costs through additional neutralization procedures required before disposal. The requirement for cryogenic conditions or extended reaction times at elevated temperatures further elevates energy consumption while extending production timelines due to sequential processing constraints that limit batch throughput capacity. Additionally, narrow substrate scope restricts structural diversity exploration essential for medicinal chemistry optimization campaigns, forcing researchers to develop entirely new synthetic routes when modifying core structures rather than leveraging modular building block approaches that would accelerate lead compound development.
The Novel Approach
In contrast, the patented methodology described in CN112961086A employs a direct oxidative coupling cyclization between S,S-dithioketal compounds and diazo reagents under transition metal catalysis that enables single-step construction of the indanone scaffold with remarkable efficiency across diverse substrate combinations while operating under industrially feasible conditions using standard processing equipment. This innovative process utilizes commercially available rhodium catalysts combined with silver-based oxidants in common solvents like dichloroethane or toluene at moderate temperatures (100–120°C), eliminating hazardous reagents and complex purification procedures required by legacy methods while maintaining exceptional functional group compatibility across electron-rich and electron-deficient substrates alike. The reaction demonstrates consistent yields exceeding eighty percent through optimized stoichiometry where precise control over catalyst loading (0.05 mol%), oxidant ratio (1:2), and basic additive concentration prevents common side reactions such as dimerization or β-hydride elimination that typically reduce product quality in alternative approaches. With reaction times standardized at twenty-four hours followed by straightforward chromatographic separation using petroleum ether/ethyl acetate mixtures, this approach achieves high-purity products suitable for pharmaceutical applications without requiring specialized analytical monitoring beyond standard QC protocols. The simplified workflow significantly reduces production costs through lower energy consumption during thermal processing and minimized solvent usage during workup stages while enhancing environmental sustainability through reduced waste generation compared to conventional multi-step sequences.
Mechanistic Insights into Rhodium-Catalyzed Oxidative Coupling Cyclization
The catalytic cycle initiates with oxidative addition of rhodium(I) complex to diazo compound III forming a highly reactive rhodium-carbene intermediate that subsequently undergoes nucleophilic attack by the enolizable S,S-dithioketal compound II at its carbonyl carbon position through a concerted mechanism facilitated by the electron-donating properties of dithioacetal groups which activate both nucleophilicity and electrophilicity within the same molecular framework. This key transformation generates a zwitterionic species that rapidly cyclizes via intramolecular electrophilic aromatic substitution at the ortho position of the aryl ring where electronic effects from substituents R¹ critically influence regioselectivity without requiring directing groups typically needed in alternative methodologies. The resulting spirocyclic intermediate then undergoes reductive elimination from the rhodium center to release the desired product while regenerating active catalyst species through silver-mediated oxidation that maintains rhodium in its trivalent state throughout multiple catalytic turnovers without significant decomposition pathways observed under optimized conditions.
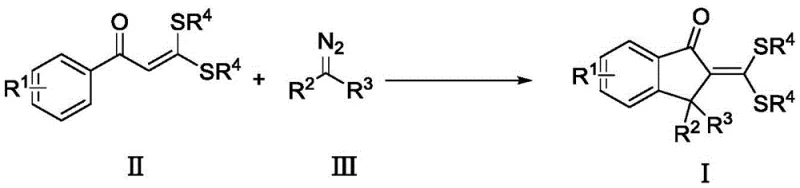
Crucially, silver salts serve dual functions as both oxidant source and halide scavenger preventing catalyst deactivation through halide coordination while simultaneously facilitating proton transfer steps essential for cyclization completion without requiring strong bases that could promote decomposition pathways observed in alternative systems. The basic additive (LiOAc) plays an indispensable role in deprotonating acidic intermediates generated during carbene formation while maintaining optimal pH conditions that suppress competing aldol condensation side reactions which would otherwise generate impurities affecting final product purity profiles critical for pharmaceutical applications. This carefully balanced reaction environment ensures high regioselectivity toward desired indanone products while maintaining excellent stereochemical integrity at chiral centers without observed racemization—a critical factor where enantiomeric purity directly impacts biological activity in drug candidates targeting neurological pathways such as acetylcholinesterase inhibition relevant to Parkinson's disease dementia treatments.
How to Synthesize 2-Methylene-1-indanone Derivatives Efficiently
This patented synthesis represents a significant advancement over conventional multi-step approaches by enabling direct construction of the indanone core through a single oxidative coupling reaction between readily accessible starting materials without requiring specialized equipment modifications or hazardous reagent handling protocols typically associated with legacy methodologies. The process eliminates numerous intermediate purification steps while maintaining high functional group compatibility across diverse substrate combinations including those containing sensitive functional groups like halogens or alkoxy moieties that would decompose under traditional synthetic conditions. Detailed standardized operating procedures have been developed through extensive process validation studies to ensure consistent product quality and yield across different manufacturing scales from laboratory benchtop to commercial production volumes. The following step-by-step guide provides essential protocols for successful implementation of this innovative methodology within industrial manufacturing environments while meeting stringent regulatory requirements for pharmaceutical intermediate production.
- Combine S,S-dithioketal compound II and diazo compound III in dichloroethane solvent at optimized molar ratio under nitrogen atmosphere
- Introduce rhodium catalyst system with silver oxidant and basic additive while maintaining precise thermal control
- Execute reaction at controlled temperature followed by standardized chromatographic purification protocol
Commercial Advantages for Procurement and Supply Chain Teams
The implementation of this novel synthesis methodology addresses critical pain points in pharmaceutical intermediate supply chains by significantly reducing production complexity while enhancing material availability for drug development programs through streamlined manufacturing workflows that minimize dependency on specialized intermediates prone to supply disruptions. By eliminating multiple synthetic steps required by traditional approaches, this process creates substantial opportunities for cost optimization without compromising on product quality or regulatory compliance requirements essential for pharmaceutical manufacturing operations serving global markets.
- Cost Reduction in Manufacturing: The one-step nature of this reaction eliminates expensive intermediate purification procedures associated with multi-step sequences while substantially reducing solvent consumption through optimized workup protocols; this streamlined approach also avoids costly transition metal removal processes since rhodium catalysts can be efficiently recovered without requiring additional heavy metal scavenging steps that typically add significant processing costs during final product isolation stages.
- Enhanced Supply Chain Reliability: Utilization of commercially available starting materials with extended shelf lives ensures consistent raw material availability while eliminating dependencies on specialized intermediates that often cause supply disruptions; the robust reaction conditions tolerate minor variations in input material quality without affecting final product specifications, providing greater flexibility in supplier qualification processes essential for maintaining continuous production schedules.
- Scalability and Environmental Compliance: The process demonstrates excellent linear scalability from laboratory validation to industrial production volumes with minimal parameter adjustments required; reduced waste generation through improved atom economy aligns with green chemistry principles while simplifying regulatory reporting requirements; simplified workup procedures eliminate hazardous byproducts like methyl mercaptan that complicate waste treatment protocols in traditional syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthesis methodology based on extensive process development experience and validation data from multiple production campaigns across diverse substrate combinations relevant to pharmaceutical intermediate manufacturing requirements.
Q: How does this one-step synthesis overcome limitations of traditional multi-step indanone production methods?
A: The patented oxidative coupling cyclization eliminates pre-functionalization requirements and harsh reaction conditions through direct ring construction under mild thermal parameters, achieving superior functional group tolerance while avoiding toxic byproducts like methyl mercaptan generated in conventional approaches.
Q: What specific advantages does rhodium catalysis provide for pharmaceutical intermediate manufacturing?
A: Rhodium-based catalysis enables precise regioselectivity at moderate temperatures (110°C) with efficient catalyst recovery protocols that prevent metal contamination concerns while maintaining exceptional yield consistency across diverse substrate combinations.
Q: How does this methodology support reliable commercial scale-up for pharmaceutical supply chains?
A: The process demonstrates linear scalability from laboratory validation to industrial production volumes through robust reaction parameters that tolerate minor input variations without compromising purity specifications or requiring specialized equipment modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylene-1-indanone Derivative Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation capable of detecting impurities at parts-per-million levels required by global regulatory authorities including FDA and EMA guidelines. This patented technology represents just one example of our commitment to developing innovative synthetic routes that address complex challenges in pharmaceutical intermediate manufacturing with exceptional reliability and quality control standards that meet global regulatory requirements across all major markets including North America, Europe, and Asia-Pacific regions.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this methodology can optimize your specific supply chain needs; please contact us for detailed COA data and route feasibility assessments tailored to your production requirements including batch size optimization studies and regulatory documentation support packages.
