Advanced Metal-Free Synthesis of 3-Selenoindole Intermediates for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for bioactive scaffolds, particularly those containing selenium, due to their significant therapeutic potential in treating inflammation and cancer. Patent CN107778210B introduces a groundbreaking methodology for the synthesis of 3-selenoindole compounds, addressing critical limitations found in traditional approaches. This innovation leverages a metal-free strategy that utilizes elemental selenium as a direct selenium source, coupled with isonitrile compounds and indoles under remarkably mild conditions. By operating at temperatures ranging from 25-70°C and employing air as the terminal oxidant, this process eliminates the need for hazardous reagents and complex catalytic systems. The technical breakthrough lies in its ability to generate diverse 3-selenoindole derivatives with high atom economy and excellent functional group tolerance. For R&D directors and procurement specialists, this represents a pivotal shift towards greener, more cost-effective manufacturing protocols that do not compromise on purity or yield. The method's reliance on commercially available starting materials further enhances its viability for immediate industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of selenium-carbon bonds within indole frameworks has relied heavily on transition metal catalysis, which introduces significant economic and environmental burdens. Traditional protocols often necessitate the use of expensive palladium or copper catalysts, requiring stringent removal steps to meet pharmaceutical purity standards regarding heavy metal residues. Furthermore, many existing methods demand harsh reaction conditions, including high temperatures and inert atmospheres, which increase energy consumption and operational complexity. The substrate scope in these conventional routes is frequently narrow, limiting the structural diversity accessible for drug discovery programs. Additionally, the use of pre-functionalized selenium reagents, such as diselenides, adds extra synthetic steps and cost to the overall supply chain. These factors collectively hinder the scalability and commercial feasibility of producing selenium-containing intermediates, creating bottlenecks for supply chain managers aiming for consistent and cost-efficient production lines.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a direct coupling strategy that bypasses the need for transition metals entirely. By employing elemental selenium powder, a cheap and abundant raw material, the process drastically reduces raw material costs and simplifies the supply chain logistics. The reaction proceeds smoothly in common organic solvents like tetrahydrofuran under an air atmosphere, leveraging oxygen as a green oxidant with water as the theoretical by-product. This metal-free condition not only ensures high purity of the final product without extensive purification but also aligns with modern green chemistry principles. The method demonstrates exceptional versatility, accommodating a wide range of substituents on the isonitrile, indole, and amine components, thereby enabling the rapid generation of diverse chemical libraries. This operational simplicity and broad applicability make it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
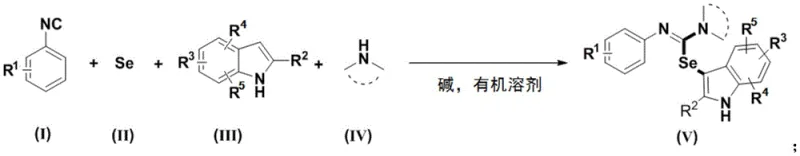
Mechanistic Insights into Metal-Free Selenylation Cyclization
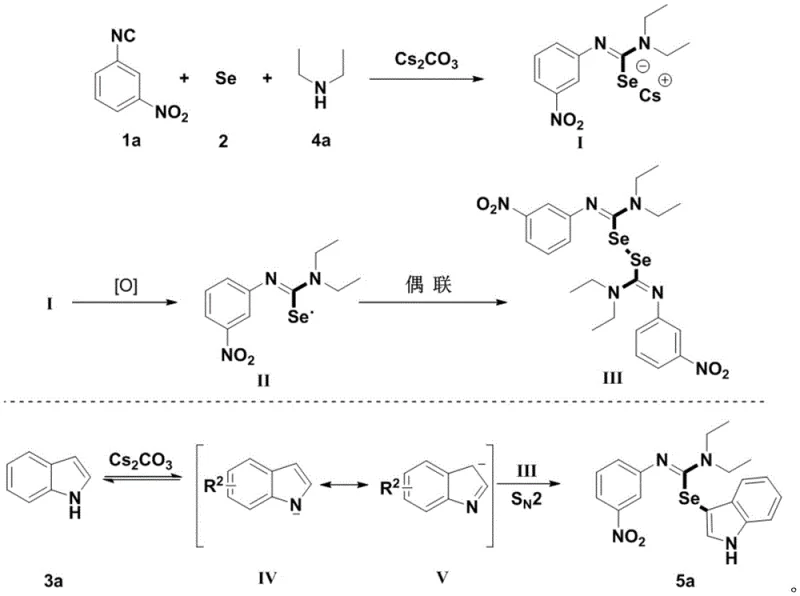
The mechanistic pathway of this transformation involves a sophisticated radical-mediated sequence initiated by the interaction of elemental selenium with the isonitrile and secondary amine components. Under the influence of a base, such as cesium carbonate, the reactants form a selenourea intermediate which serves as the key selenium donor. In the presence of an oxidant like TEMPO or simply air, this intermediate undergoes oxidation to generate a reactive selenium radical species. This radical species then participates in a coupling event that ultimately delivers the selenium moiety to the indole core. The precise control over radical generation ensures high selectivity for the C3 position of the indole ring, minimizing the formation of regioisomers or side products. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters for specific substrates, as it highlights the importance of base strength and oxidant availability in driving the reaction to completion efficiently.
Impurity control is inherently managed through the clean nature of the radical mechanism and the absence of metal catalysts that often lead to complex decomposition pathways. The reaction conditions are mild enough to preserve sensitive functional groups such as esters, nitriles, and halides, which are commonly found in advanced pharmaceutical intermediates. The use of air as the oxidant prevents the accumulation of hazardous waste streams associated with stoichiometric chemical oxidants. Moreover, the by-product profile is simple, primarily consisting of water and benign salts, which facilitates downstream processing and waste treatment. For quality assurance teams, this translates to a more predictable impurity profile and easier validation of the cleaning processes. The robustness of the mechanism across various substituted indoles and isonitriles confirms its reliability for producing high-purity pharmaceutical intermediates consistently.
How to Synthesize 3-Selenoindole Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and reaction monitoring to ensure optimal yields and purity. The process begins with the precise weighing of isonitrile, elemental selenium, indole, and secondary amine, typically in a molar ratio of 1:1.2:1.2:1.2 to drive the reaction forward effectively. These components are suspended in a dry organic solvent, with tetrahydrofuran being the preferred medium due to its solubility profile and boiling point. A base like cesium carbonate is added to activate the nucleophiles, and the mixture is stirred at a controlled temperature of 40°C for approximately 12 to 16 hours. Detailed standardized synthesis steps see the guide below.
- Mix isonitrile compound, elemental selenium, indole compound, and secondary amine in an organic solvent like THF.
- Add an inorganic base such as cesium carbonate and optionally a catalyst like TEMPO to the reaction mixture.
- Stir the reaction at 25-70°C under air atmosphere for 8-20 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers profound advantages that directly impact the bottom line and supply chain resilience for chemical manufacturers. The elimination of transition metal catalysts removes a significant cost center associated with both the purchase of precious metals and the subsequent removal processes required to meet regulatory limits. This simplification of the workflow reduces the number of unit operations, leading to lower labor costs and shorter production cycles. Furthermore, the use of elemental selenium, a commodity chemical, ensures stable pricing and availability compared to specialized organoselenium reagents. The mild reaction conditions also reduce energy consumption, contributing to lower utility costs and a smaller carbon footprint. These factors combine to create a highly competitive manufacturing process that enhances margin potential while maintaining rigorous quality standards.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the complete avoidance of expensive transition metal catalysts such as palladium or copper, which are subject to volatile market pricing. By substituting these with inexpensive elemental selenium and organic bases, the raw material cost per kilogram of product is significantly lowered. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary chemicals and filtration media. This streamlined approach minimizes waste generation and disposal costs, further enhancing the overall economic viability of the production line. The cumulative effect of these savings allows for a more aggressive pricing strategy in the competitive pharmaceutical intermediate market.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly improved by relying on commodity chemicals that are widely available from multiple global suppliers. Elemental selenium, indoles, and isonitriles are established industrial chemicals with robust supply networks, reducing the risk of shortages that often plague specialized reagents. The mild reaction conditions also mean that the process can be executed in standard glass-lined or stainless steel reactors without requiring exotic materials of construction. This flexibility allows for easier technology transfer between manufacturing sites and reduces dependency on single-source vendors for critical equipment. Consequently, procurement managers can secure long-term supply agreements with greater confidence and reduced risk of disruption.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of air as the oxidant, eliminating the need for handling hazardous high-pressure oxygen or toxic chemical oxidants. The reaction exotherm is manageable under the specified mild temperatures, ensuring safe operation in large-scale reactors. From an environmental standpoint, the generation of water as the primary by-product aligns with increasingly stringent environmental regulations and corporate sustainability goals. This green profile simplifies the permitting process for new manufacturing lines and reduces the liability associated with hazardous waste disposal. The combination of safety, scalability, and environmental compliance makes this technology a future-proof choice for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-free synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy and relevance. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing production portfolios. The responses cover aspects ranging from raw material compatibility to process safety and regulatory compliance. This information serves as a foundational resource for technical discussions between suppliers and potential manufacturing partners.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN107778210B operates under metal-free conditions, utilizing elemental selenium and air oxidation instead of costly transition metals.
Q: What are the typical reaction conditions for this process?
A: The reaction proceeds under mild conditions, typically between 25-70°C, often optimized around 40°C, using common organic solvents like tetrahydrofuran.
Q: Is this method suitable for large-scale production?
A: Yes, the use of readily available raw materials, mild temperatures, and air as a green oxidant makes this process highly scalable and environmentally compliant for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Selenoindole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating innovative academic research into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to adapt the metal-free synthesis of 3-selenoindole compounds to meet your specific volume and purity requirements. We maintain stringent purity specifications and operate rigorous QC labs to ensure every batch meets the highest international standards for pharmaceutical intermediates. Our commitment to quality and consistency makes us the ideal partner for companies seeking to secure a stable supply of these valuable selenium-containing scaffolds. We understand the critical nature of supply chain continuity in the pharmaceutical sector and are dedicated to supporting your long-term growth.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your current sourcing strategy. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this metal-free protocol for your specific projects. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Our experts are ready to collaborate with you to solve complex synthesis challenges and drive value through chemical innovation. Let us help you accelerate your development timelines with our reliable and efficient manufacturing capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
